

Author byline as per print journal: Brian Godman1,2,3,4, BSc, PhD; Andrew Hill5; Professor Steven Simoens6, MSc, PhD; Amanj Kurdi1,7, BSc, PhD; Jolanta Gulbinovič8, MD, PhD; Antony P Martin2,9; Angela Timoney1,10; Dzintars Gotham11, MBBS; Janet Wale12; Tomasz Bochenek13, MD, PhD; Celia C Rothe13; Iris Hoxha14; Admir Malaj15; Christian Hierländer16; Robert Sauermann16, MD; Wouter Hamelinck17; Zornitza Mitkova18; Guenka Petrova18, MPharm, MEcon, PhD, DSci; Ott Laius19; Catherine Sermet20; Irene Langer21; Gisbert Selke21; John Yfantopoulos22; Roberta Joppi23; Arianit Jakupi24; Elita Poplavska25, PhD; Ieva Greiciute-Kuprijanov26; Patricia Vella Bonanno1, BPharm, MSc, PhD; JF (Hans) Piepenbrink27; Vincent de Valk27; Carolin Hagen28; Anne Marthe Ringerud28; Robert Plisko29; Magdalene Wladysiuk29; Vanda Marković-Peković30,31, PhD; Nataša Grubiša32; Tatjana Ponorac33; Ileana Mardare34; Tanja Novakovic35; Mark Parker35; Jurij Fürst36; Dominik Tomek37, PharmD, MSc, PhD; Mercè Obach Cortadellas38; Corinne Zara38; Maria Juhasz-Haverinen39, MScPharm; Peter Skiold40; Stuart McTaggart41; Alan Haycox1, PhD
|
Introduction: There are appreciable concerns among European health authorities with growing expenditure on cancer medicines and issues of sustainability. The enhanced use of low-cost generics could help. |
Submitted: 5 February 2019; Revised: 14 March 2019; Accepted: 20 March 2019; Published online first: 2 April 2019
Despite the limited health gain for most new cancer medicines, their prices have increased appreciably in recent years [1–7]. This is reflected in the price per life year gained for new cancer medicines rising fourfold during the past twenty years after adjusting for inflation [2, 8]. Typically, prices for new cancer medicines are now approximately 6,000 to 9,000 Euros/patient/month and growing [1, 5, 9], with typically higher prices for new cancer medicines in the US versus Europe. As a result of rising prices for new cancer medicines, as well as increased prices for patented cancer medicines [5, 10–12], coupled with the increasing prevalence of cancer [1, 13], spending on cancer medicines in Europe has more than doubled in recent years, rising from Euros 8 billion in 2005 to Euros 19.1 billion in 2014 (at current prices) [14].
As a result, medicines for oncology now dominate pharmaceutical expenditures in developed markets, with projected worldwide sales for oncology medicines in 2017 ranging from US$74 to US$84 billion [15]. Overall, total worldwide sales of oncology medicines are expected to reach US$112 billion (Euros 91 billion) per year by 2020 [16]. Further growth is expected after this with cases of cancer worldwide likely to rise to 21.4 million per year by 2030 [1, 13] coupled more with than 500 companies actively pursuing new oncology medicines in over 600 indications, and looking to benefit from their investment [17]. The cost of cancer care also currently accounts for up to 30% of total hospital expenditure across diseases among European countries, and this is also rising with the launch of new premium-priced cancer medicines [14, 18].
Increasing prices for new cancer medicines, combined with increasing prevalence rates, is putting considerable strain on patients to access cancer treatments and healthcare systems to fund them [19–23]. High reimbursed prices for new cancer medicines certainly in Europe have been enhanced by the emotive nature of cancer, capturing public, physician, and political attention [13, 24–27]. If these trends with new cancer medicines continue, there is a real risk that universal healthcare across Europe will become unsustainable, which will not be in the best interest of any key stakeholder group. Initiatives among European health authorities to help fund future cancer treatments, including new valued high-cost medicines, include encouraging greater use of generic cancer medicines when they become available [12, 26]. This is especially important given the increasing number of cancer medicines that are now out of patent [28]. Initiatives to enhance greater use of generic cancer medicines include encouraging International Nonproprietary Name (INN) prescribing, financial incentives to enhance the prescribing and dispensing of generics versus originators, as well as compulsory generics substitution [29–38]. INN prescribing is supported by multiple publications showing no difference in effectiveness and safety between generics and originators across a range of disease areas including oncology medicines [28, 39–44]. There have though been concerns with generic imatinib due to different polymorphic forms between the originator and generics as early small scale studies suggested differences [45]. However, later studies involving more patients showed no difference in outcomes between generics and originators of imatinib [45, 46], and, typically, no substantial evidence that generic imatinib is less effective than the originator [47].
In general, prices for generic medicines in Europe are 20% to 80% below originator prices; however, some generics can be priced as low as 2% to 4% of the originator price before the patent was lost [48, 49]. As a result, substantial differences can occur in the prices of generics across Europe [37, 50, 51]. Overall, low prices can potentially be achieved for generic cancer medicines because of the low cost of goods that have been reported at just 1% or more of originator prices for some new cancer medicines [52, 53]. These low cost of goods have already resulted in considerable discounts for generic imatinib across countries versus pre-patent loss prices [54, 55]. However, this is not universal. For instance, in China, generic imatinib is only 10%–20% below originator prices although generic capecitabine is 50% lower than originator prices [56]. There have also been low prices for generic versions of paclitaxel in Europe at just over 1% of originator prices [57]. Docetaxel also has a low price in some European markets enhanced by an appreciable number of generic versions available [26]. Having said this, changes in the manufacturer have resulted in the prices of some low volume old anticancer medicines rising appreciably among European countries including Italy and the UK [58]. However, pharmaceutical companies are now being fined for such behaviour, e.g. Euros 5.2 million for Aspen in Italy for price increases for some of its anticancer medicines, with ongoing investigations and initiatives across Europe to address this [59, 60].
In some European countries, there have also been issues regarding the prescribing of certain generic medicines where there are different indications, one of which is still patent protected. This was the case with pregabalin for the treatment of neuropathic pain as opposed to epilepsy [61].
There have also been concerns with drug shortages if the prices of medicines including generics become too low, which is already happening for certain parenteral medicines [62]. However, these concerns have to be balanced against the increasing availability and use of low-cost generic medicines to release resources to fund increased cancer care including new innovative medicines, reducing patient co-payments where these exist, and stimulating innovation [13, 48, 63–65].
Consequently, there is a need to document current regulations surrounding the pricing of generic oral anticancer medicines across Europe, and their impact on subsequent prices, as well as issues regarding prices of medicines once one indication loses its patent but not the others. As a result, the aims of this paper are multiple. The primary aim is to document current arrangements for the pricing of oral generic medicines across Europe and whether there are any differences in pricing policies for generic cancer medicines versus those for other disease areas. This is important to maximize savings following generics availability as we do see differences in reimbursement decisions for new medicines for cancer versus those for other disease areas [27, 66]. Secondly, to investigate what happens to the prices of oral cancer medicines if one indication loses its patent but not the other indication(s) since indication-specific pricing will undoubtedly reduce potential savings following generics availability, and, as mentioned, there were issues with the prescribing of pregabalin across indications when the first indication lost its patent [61]. Thirdly, to investigate how payers and their advisers envisage developments in the pricing of oral generic medicines for cancer as more cancer medicines lose their patents. Fourthly, to assess whether health authorities have any concerns with patient safety when prescribing oral generic cancer medicines similar to the situation with medicines with a narrow therapeutic index, such as lithium and certain medicines for epilepsy [67–69]. Fifthly, to review current prices for a number of oral cancer medicines across Europe where generic versions are available in all or some of the countries. This will also include an evaluation of prices over time to fully assess the impact of the different pricing arrangements for generic cancer medicines across Europe since we are aware that prices of generics can vary substantially across Europe due to different policies [26, 37, 48, 49], and that Central and Eastern European (CEE) countries are likely to have generics earlier [70]. Sixthly, to assess whether there are any differences in the prices of generics by population size and geography (CEE versus Western European countries) as there have been concerns that countries with small populations, and hence lower economies of scale, may have difficulty negotiating low prices for medicines [71] ; however, published literature has not found to be the case [31, 72]. Countries with greater economic power may potentially be able to negotiate lower prices for generics, and we will also investigate this. Lastly, as mentioned, prices of some old low volume anticancer medicines have risen among European countries in recent years with changes in the manufacturer [58], and we will assess the impact of any changes in manufacturers on subsequent prices, and the likely future direction.
We developed a number of hypotheses for the different aims. These include:
We believe that this is the first time that such comprehensive research regarding the regulations surrounding the pricing of oral cancer medicines, and their influence on subsequent prices, has been undertaken across Europe. Consequently, we believe the findings will inform future discussions within and between European and other countries regarding the pricing of oral generic cancer medicines now and into the future. In addition, we aim to stimulate discussions regarding subsequent prices and/or discounts for still patented oral cancer medicines that used off-patented products as their comparator during pricing negotiations. As a result, help with issues of affordability and sustainability of medicine use in the future in this high priority disease area.
When we refer to ‘generics’, we mean the chemical entity (INN). This study also includes imatinib, a comparatively newer anticancer agent, which is treated similarly to other oral generic cancer medicines. Biosimilars of originator biological medicines are regulated by a different framework, and incentives to increase their use are different [73]. Consequently, we will not be discussing biosimilars.
We have chosen to concentrate on Europe in view of the high use of generic medicines in this region. There are also different approaches to the pricing of generics among the various countries, as well as an appreciable number of existing initiatives to enhance the prescribing of generic medicines.
A mixed method approach was employed including both qualitative and quantitative research to meet the study objectives, and included 25 European countries, see Box 1.
This appreciable number of European countries fully encompasses differences in geography, population size, gross domestic product (GDP) per capita, pricing approaches towards generics as well as different approaches to the financing of healthcare [30, 70, 74]. Countries were also broken down into CEE and Western European countries based on the Organisation for Economic Co-operation and Development (OECD) definition [75] to ascertain any differences in the pricing of oral generic cancer medicines based on population sizes and market power.
Qualitative interviews were undertaken with senior personnel within health authorities including heads of pricing and reimbursement of medicines as well as their advisers and a limited number of academics with expertise on national pharmaceutical issues and other key issues among European countries. The involvement of these senior level personnel in the qualitative interviews, and their involvement as co-authors, is seen as very important to enhance the accuracy, robustness and insight of the replies as there have been concerns when such approaches are not used [76, 77]. The co-authors were either identified via the Piperska group [78, 79] – a multidisciplinary network of professionals with interest in the quality use of medicines – or had been previous co-authors on similar pan-European projects involving generic medicinal products [29, 80–82]. We have successfully used this approach in other key areas [30, 61, 70, 73, 74, 80, 81, 83].
The qualitative questions to address the identified objectives included:
If applicable, pricing policies for generics were broken down into three principal groups to aid comparisons. This builds on previous publications that have used this approach [84–87], and include:
Health authority and health insurance company databases were used for the quantitative research apart from Greece and Serbia where commercial sources were used. The information within these databases is robust as they are typically used to trigger payment to pharmacists. In addition, the databases are regularly audited, and cover the whole country rather than a sample of pharmacies as seen with commercial databases. We have used this approach before when conducting research on prices and utilization of generics, originators and patented products within a class or related class [30, 49, 70, 72, 80–82, 88].
The oral cancer medicines selected for the quantitative research were busulfan (L01AB01), capecitabine (L01BC06), chlorambucil (L01AA02), cyclophosphamide (L01AA01), flutamide (L02BB01), imatinib (L01XE01), melphalan (L01AA03), methotrexate (L01BA01), and temozolomide (L01AX03) [89]. These oral cancer medicines were chosen to provide a range of medicines for use across different cancer types, with typically multiple indications, covering the oral cancer medicines listed in the World Health Organization (WHO) essential medicines list, and exhibiting varying timings regarding the loss of patent, building on previous publications [55]. Oral medicines, such as gefitinib, sorafenib, sunitinib and tioguanine were not included as there appeared to be no generics yet available for these oral cancer medicines even among the selected CEE countries at the time of the study. The (non-) tyrosine kinase inhibitors are a particular case as there are typically multiple indications as well as potentially different dates for patent loss across countries [90].
Since the perspective of this study is that of health authorities, reimbursed prices were principally chosen for comparative purposes rather than total prices, which include patient co-payments. Reimbursed prices can include prices after deducting legally mandated discounts; however, this is unlikely for generic medicines. In a minority of countries, e.g. Kosovo, procured and total prices were used as reimbursed prices were unavailable. In Italy and Norway, reimbursed prices were also listed but the medicines are typically dispensed in hospitals where further confidential discounts are provided. However, since these discounts are confidential, we could only report ambulatory care prices. Furthermore, in some countries, prices did not include VAT and/or pharmacy margins, e.g. Malta, The Netherlands and the Republic of Srpska. However, this was not seen to have an appreciable impact on the analyses since percentage reductions were typically used for comparative purposes to assess the influence of different policies. In addition, the prices of generics at least initially in a number of European countries are based on percentage reductions from pre-patent loss originator prices [30, 70]. We have successfully used this approach in a number of previous publications [30, 70, 74, 80, 84].
We are conscious that we used prices from Scotland rather than the UK as a whole. However, there are no differences in prices across the UK with reimbursed prices in ambulatory care based on the tariff price coupled with free-market competition, which leads to further price reductions over time. Similarly, no differences are envisaged in generic and originator drug prices in ambulatory care in Catalonia versus Spain as a whole. However, we acknowledge that there will be differences in hospital prices across Spain through variations in discounts and managed entry agreements between the different regions [91].
Prices were collected over a period of time between 2013 and 2017, based on the unit, e.g. tablet strength, for the different originators and the cheapest generics substitute unit available, especially for branded generics, as opposed to defined daily doses (DDDs). Unit strength was chosen as generally there are no DDDs for oral cancer medicines in view of typically a number of indications for each molecule [89]. The documented prices for each year between 2013 and 2017 were the last available price for that year, e.g. October to December if prices are adjusted every three months or December if prices are adjusted monthly. The years were chosen to assess the extent of any price changes in recent years especially given recent price increases for some off-patent medicines.
The actual unit strength chosen for comparative purposes reflects the most used strength in a number of European countries, although we recognize that there may be different strengths available in some countries. In addition:
Initially, prices were documented in the country’s currency if this was not Euros. Subsequently where relevant, prices were converted to Euros for the purposes of comparison based on current exchange rates and validated with the co-authors to enhance the robustness of the findings, see Table 1A in the Appendix.
Table 2A in the Appendix contains details of the population sizes and the breakdown of countries into three groups for analysis purposes (small, medium and large). Countries with populations under three million were described as small, those with populations between three and 16 million as medium, and the remainder as large to produce three roughly equal groups although recognizing that more countries were in the ‘small’ category than in the others.
Statistical tests were performed to ascertain any trends in prices across countries as well as any difference in price reductions over time as a result of the different approaches towards the pricing of generics across Europe. The significance threshold was set at p > 0.05. Statistical tests were also run to check whether prices were influenced by population size as this has been a concern in the past. However, we did not have enough variables to perform any multivariate analyses as there are not enough specific measures in each European country that had been initiated to control the price of generic cancer medicines which could be used as a yes/no variable. We have though collated the impact of the different pricing approaches for generic medicines across Europe as one of the principal objectives of this paper was to document these and their influence on subsequent prices for oral generic cancer medicines.
No analysis was made regarding the impact of volumes on generic drug prices as seen in other studies because this was not a focus of the paper [92]. This will be followed up in future studies. Potential savings from the increased utilization of generic medicines for cancer over time will also form a separate project building on previous calculations [93].
In line with the objectives of this paper, we will first report the findings from the qualitative research. This will be followed by the findings from the quantitative research.
Regulations surrounding the pricing of generics including those for cancer across Europe
Regulations for the pricing of generics generally and for oral cancer medicines including likely changes in the short term
As previously seen, there were differences in the approaches to the pricing of generics among the various European countries, see Table 1, which could again be categorized under three broad headings. These are: (i) prescriptive pricing approaches; (ii) market forces, and (iii) and a mixture of the approaches.
As shown in Table 1, there were no differences in the pricing approaches for generic oral medicines for oncology compared with those for other disease areas, see Table 1, apart from the fact that in some countries these cancer medicines were dispensed in hospitals, which could include additional discounts that are typically confidential. This is important given the considerable savings that can be achieved with the introduction of generics, see Table 2.
Pricing of oral cancer medicines if one indication has lost its patent
There were generally no issues with the pricing and usage of oral generic cancer medicines once one indication had lost its patent but not the other indication(s), see Table 1; however, this was not universal. There is similar to the situation in Germany once the first indication for a multiple-sourced product has lost its patent [61], i.e. the generic medicine can be prescribed across all indications at the discounted price versus the originator. However, this is different to the situation that existed with pregabalin among a number of European countries where the generics could only be prescribed for epilepsy and the originator must be prescribed and reimbursed accordingly for neuropathic pain [61].
This situation is encouraging for maximiẕing savings following generic drug availability given the considerable savings that can be achieved with the introduction of generics, see Table 2, and will be closely monitored in the future. This includes procured medicines for distribution in hospitals.
Substitution of oral cancer medicines and initiatives to encourage their prescribing
It was also encouraging to see that once generics became available, there were no concerns with substitution with the selected oral generic cancer medicines, see Table 1. This is similar to the general situation for cancer medicines [28]. Consequently, originators can be substituted with generics without compromising care. This is again important for maximizing savings once generics become available.
Initiatives to encourage the prescribing of generics in preference to originator cancer medicines among the various European countries, see Box 2, were similar to those for generic medicines in general.
Price reductions with generics over time and with originators, as well as the influence of population size and market forces
Prices for originator 400 mg imatinib were similar in Western European countries in 2015 before generic drug availability, see Figure 1. Generic imatinib was already available in CEE countries, e.g. in Albania, Estonia, Latvia, Lithuania, Romania, Serbia and Slovakia, on or before 2013, and in Poland and Slovenia in 2014. Generic capecitabine and flutamide were already available in a number of Western European countries in 2013. The other three oral anticancer medicines were already available as generics among Western European countries before 2013.
As expected, there were differences in the prices of the generics for capecitabine (500 mg), flutamide (250 mg), imatinib (400 mg) and temozolomide (20 mg and 250 mg) in 2017 among the European countries where these generics were available, see Table 3, reflecting differences in the approaches to the pricing of oral generics among the various European countries, see Table 1.
There were no differences in the prices of capecitabine (500 mg), flutamide (250 mg), imatinib (400 mg) and temozolomide (20 mg and 250 mg) when the countries were broken down into small, medium or large populations, see Table 2A, apart from generic imatinib for European countries with large populations, i.e. capecitabine (p = 0.372 for medium and p = 0.100 for large), flutamide (p = 0.700 for medium and 0.385 for large), imatinib (p = 0.249 for medium and p = 0.037 for large), temozolomide 20 mg (p = 0.764 for medium and p = 0.085 for large) and temozolomide 250 mg (p = 0.951 for medium and p = 0.105 for large). Prices for generic capecitabine (p = 0.007), generic imatinib (p = 0.015) and temozolomide 20 mg (p = 0.015) were though typically appreciably higher when combined among Western European countries compared with CEE countries. However, this was not the case for flutamide 250 mg or temozolomide 250 mg. This will change for generic imatinib as prices fall among the large Western European countries over time, e.g. generic imatinib in Scotland by March 2018 was already 89.1% below pre-patent loss prices having only been made available in 2016.
Overall in 2017, the differences in the prices of generics of capecitabine (500 mg), flutamide (250 mg), imatinib (400 mg) and temozolomide (20 mg and 250 mg) among the different European countries, see Table 3, appear to be a reflection of the differences in the country’s pricing approaches towards generics, see Table 1, rather than population size when broken down into small, medium or large, as well as the timing of generic drug availability. These findings are similar to those with generic busulfan, chlorambucil, cyclophosphamide and melphalan, see Table 4, and confirm this trend certainly for oral generic cancer medicines.
There also appears to be no correlation in terms of the price reductions seen for generic capecitabine, flutamide, imatinib or temozolomide in 2017 versus 2013 originator prices with population sizes, i.e. appreciable price reductions over time were not confined to European countries with higher populations, see Table 2. There was also no significant difference in the percentage reduction between Western and CEE countries for capecitabine, flutamide, imatinib or temozolomide.
If anything, there was a greater reduction in the prices of generic capecitabine, see Figure 2, in countries with a mixed approach to the pricing of generics versus those with market forces (p = 0.035) and prescriptive (p = 0.041) approaches.
This was not the case for generic imatinib, see Figure 3, although there was a trend (p = 0.079 for market forces and p = 0.119 for prescriptive pricing approaches). However, this may change as prices of generic imatinib fall in The Netherlands, Spain, Sweden and the UK, e.g. as mentioned generic imatinib in Scotland was already 89.1% below pre-patent loss prices by March 2018 having only been made available in 2016.
Price changes for generics with changes in the licence holder
In recent years, the generic drug manufacturer Aspen has taken over the licence of busulfan, chlorambucil, and melphalan and enjoyed market monopoly, with typically no generics competition, despite the loss of patent protection. Another pharmaceutical company, Baxter, has used a similar strategy with cyclophosphamide. Countries have typically been faced with having to either accept these higher prices or no longer procuring and/or reimbursing these medicines.
Figure 4 depicts current prices per tablet for these four oral generic anticancer medicines (2017) where these are currently listed and reimbursed since not all European countries currently reimburse these four medicines. There were no significant differences in the mean prices for these four anticancer medicines between Western and CEE countries. In the case of busulfan, there was a difference in current prices among European countries, with Western European countries typically having higher prices but this did not reach significance (p = 0.08). Population size again did not appear to influence subsequent prices.
There were appreciable price rises in a number of other European countries following Aspen’s purchase of busulfan, chlorambucil and melphalan from GlaxoSmithKline, see Table 4.
Similar price rises were seen in Germany during this period, for example:
Overall there again appears to be no link between prices, geographic region and population size for these four medicines despite previous concerns. If anything, countries with smaller populations, such as Estonia had lower prices than Belgium, Italy and the UK. This appears to be in contrast with previous publications [71], although this is in accordance with previous findings in Lithuania and the Republic of Srpska [31, 72].
The difference in prices of these four molecules between the countries in 2017 may well reflect differences in negotiations when the new company took over the molecules and relaunched them as a new originator at a higher price. Countries such as Estonia appeared successful in negotiating lower price increases for the relaunched medicines versus for instance Belgium, Italy and the UK. However, as mentioned earlier, the company was subsequently fined Euros 5.2 million in Italy for their activities [59]. Currently, Germany has a moratorium law stating that for any price increase, there will be an increase in the mandatory rebate of exactly the same amount as the increase. Effectively, this means that the price paid by statutory heathcare funds for ambulatory care medicines does not change [114, 115]. Legislation is also being introduced in the UK to reduce the price of generic medicines where competition fails to reduce prices and companies are seen to charge the National Health Service (NHS) unreasonably high prices [113]. The Competition and Markets Authority (CMA) in the UK has also provisionally found that two companies broke the law by agreeing not to compete to supply generic hydrocortisone to the NHS driving up annual costs by two million GBP [116]. There are also ongoing deliberations in Europe regarding concerns with appreciable price increases for generics and whether this breaks EU competition rules [60, 117]. In addition, we may see countries combining to form regional co-operations between Member States to further reduce prices where there are concerns with unjustified price rises building on current consortia [118–120]. However, this has to be balanced against issues of future profitability and potential drug shortages with for instance only 75 melphalan tablets dispensed in ambulatory care in Scotland during the study period (SM personal communication), and with just 11 boxes of generic melphalan dispensed in Serbia in 2015.
We believe this is the first study to comprehensively research the situation regarding the pricing of oral generic cancer medicines in this high priority disease area. We believe our findings again highlight the differences that are seen in the various approaches to the pricing of generics across Europe, see Table 1, and their subsequent influence on the prices of generics and discounts obtained. These considerable differences are in direct contrast with the more limited differences in prices for on-patent medicines across Europe, especially high cost ones [121], although these can still be considerable [122].
Prices for generic capecitabine, imatinib, and temozolomide 20 mg, were significantly higher when combined among Western European countries as compared with CEE countries. This may reflect greater availability of resources to spend on medicines among Western countries. However, this is not universal as there were no differences in prices between Western European and CEE countries for flutamide 250 mg or temozolomide 250 mg. Alternatively, generics may be available earlier in CEE countries with associated earlier falls in their price as seen with imatinib. As mentioned, generic imatinib in Scotland was already 89.1% below pre-patent loss prices in March 2018 having only been made available in 2016, see Figure 1. Having said this, there were already some appreciable price reductions for these various molecules and strengths among Western European countries comparing generic drug prices in 2017 versus originator prices in 2013, Table 3 and Figure 3. In addition, no overall difference in the price reductions for these molecules and strengths was found for Western European countries combined versus CEE countries combined.
The picture regarding methotrexate is complicated by the use of this medicine for immunological conditions, such as rheumatoid arthritis and psoriasis. This meant that different originator and tablet strengths (2.5 mg or 5 mg) were available amongst the European countries making comparisons difficult. In addition, the considerable time that both the originator and generic methotrexate have been available across Europe meant there were limited price reductions in reality between 2017 compared with 2013 among the various European countries. Consequently, no detailed analysis was undertaken with methotrexate.
The price reductions for capecitabine, flutamide, imatinib and temozolomide that have been achieved in practice, see Table 3, confirm the findings of Hill et al. that the costs of goods for cancer medicines can be very low in reality [52, 53]. This fuels the debate for greater transparency in the pricing of new medicines for cancer when pharmaceutical companies request premium prices especially where there is limited health gain for their new cancer medicines versus current standards.
In view of our findings, we also believe European health authorities, as well as health authorities from other countries, can use these results to reassess their pricing approaches for generics, and the subsequent implications for oral generic cancer medicines, given concerns with the increasing costs of medicines to treat patients with cancer and issues of sustainability [3, 9, 28]. This is already happening, and may well accelerate, especially if issues of access and sustainability of cancer care continue to be priority issues for all key stakeholder groups. For instance, Austria, Lithuania, and Sweden, have recently introduced measures to further lower the prices they pay for generics and this trend is likely to continue, see Table 1. However, this has to be balanced against issues of parallel exports and associated drug shortages if the prices of generics become too low [37, 123], which is depicted in the unavailability of some oral generic cancer medicines in the countries studied. This will be the subject of ongoing research.
Based on our findings combined with the continual pressure on oncology budgets, we would also likely to see countries increasingly reassess the prices of on-patent cancer medicines in their country once the comparator medicine used for pricing and reimbursement negotiations loses its patent [124]. Such activities will be enhanced by the low prices that have been achieved for oral cancer medicines once their patent is lost, see Tables 2 and 3, which will continue.
Encouragingly, we saw no differences in the pricing approaches for generic cancer medicines versus those for other disease areas despite the emotive nature of cancer, and this will continue. This is essential to maximize savings from the availability of generic cancer medicines once available, with ongoing initiatives across Europe to encourage their use where pertinent. Likely, current and future initiatives to enhance the prescribing of oral generic cancer medicines include additional demand-side measures especially among European countries with currently low use of generics versus originators. This could be alongside continued educational initiatives among key stakeholder groups, including patients, where pertinent about the lack of problems with oral generic cancer medicines given there were no concerns with substitution among the European countries surveyed, see Table 1.
We are also unlikely to see major changes in pricing approaches once one indication loses its patent. The situation seen in Germany, as well as initiatives to encourage INN prescribing, helps in this regard. These initiatives are essential in oncology given the growing burden of the cost of medicines to treat patients with cancer across Europe combined with the need to continue to provide universal healthcare. Encouraging greater prescribing of generic medicines will be helped by limited or no fears with substituting generic cancer medicines for originators among physicians and pharmacists. This is unlike a limited number of other disease areas including some medicines for patients with epilepsy as well as lithium for patients with certain mental health conditions [67–69].
If companies continue to purchase the patent for old cancer medicines and other products, and relaunch them at considerably higher prices, there is likely to be co-ordinated activities across Europe to try and address this. We are already seeing companies being fined, e.g. Italy, as well as countries instigating measures to reduce any burden from such approaches as seen currently in Germany and the UK. Such punitive actions are likely to grow in the future if this trend continues. However, this has to be balanced against potential availability especially if only small volumes are being used. This will again be the subject of continuing research.
We have again seen appreciable differences in the regulations surrounding the pricing of generics across Europe, reflected in different reimbursed prices for oral generic cancer medicines. Welcomed from an equity and resource perspective were no differences in the pricing approaches for medicines for cancer as opposed to other disease areas to help maximize savings once generics become available. In addition, the prices of generics, and any difference in the prices of generics in 2017, or price reductions versus 2013 originator prices, did not appear to be influenced by population size. This is also encouraging to maximize savings from the availability of oral generic cancer medicines. The prices of some generics were higher among Western European countries in 2017; however, this could have been influenced by generics being available earlier among CEE countries. There are concerns with some off-patented medicines being relaunched by alternative companies at appreciably higher prices, although there are ongoing steps across Europe to try to address this. These initiatives are likely to grow if this trend continues.
Reassuringly, there are no concerns with substitution of oral generic medicines. In addition, prices were typically similar across indications. Both results are important to maximiẕe savings from generic drug availability once at least one indication loses its patent.
We have tried to reduce limitations with this study by using senior level personnel for the qualitative research as well as health authority and health insurance company databases for the quantitative research. However, we are aware that we did not use reimbursed prices throughout the countries, some prices did not include VAT, and we cannot be sure that the medicines chosen for the research are entirely used for cancer indications. Despite these concerns, we believe the findings of our comprehensive research are robust providing direction not only to key stakeholder groups across Europe but also wider.
There was no funding for this research and no assistance with the write-up.
Competing interests: Most of the authors work directly for health authorities or health insurance companies or are advisers to them. Professor Steven Simoens has previously held the EGA Chair ‘European policy towards generic medicines’. All the authors have no other conflicts of interest to declare.
Provenance and peer review: Not commissioned; externally peer reviewed.
1Strathclyde Institute of Pharmacy and Biomedical Sciences, University of Strathclyde, Glasgow G4 0RE, UK
2Health Economics Centre, University of Liverpool Management School, Liverpool, UK
3Division of Clinical Pharmacology, Karolinska Institute, Karolinska University Hospital Huddinge, SE-141 86, Stockholm, Sweden
4School of Pharmaceutical Sciences, Universiti Sains Malaysia, Penang, Malaysia
5Institute of Translational Medicine, University of Liverpool, UK
6KU Leuven Department of Pharmaceutical and Pharmacological Sciences, Leuven, Belgium
7Department of Pharmacology, College of Pharmacy, Hawler Medical University, Erbil, Iraq
8Department of Pathology, Forensic Medicine and Pharmacology, Institute of Biomedical Sciences, Faculty of Medicine, Vilnius University, Vilnius, Lithuania
9HCD Economics, The Innovation Centre, Daresbury, WA4 4FS, UK
10NHS Lothian, Edinburgh, UK
11Independent researcher, Boston, MA, USA
12Independent consumer advocate, 11a Lydia Street, Brunswick, Victoria 3056, Australia
13Department of Drug Management, Faculty of Health Sciences, Jagiellonian University Medical College, Krakow, Poland
14Department of Pharmacy, Faculty of Medicine, University of Medicine, Tirana, Albania
15University of Medicine, Tirana, Albania
16Main Association of Austrian Social Security Institutions, Department of Pharmaceutical Affairs, 1 Haidingergasse, AT-1030 Vienna, Austria
17Statistics Department, APB, 11 Rue Archimède, BE-1000 Bruxelles, Belgium
18Faculty of Pharmacy, Department of Social Pharmacy and Pharmacoeconomics, Medical University of Sofia, Sofia, Bulgaria
19State Agency of Medicines, 1 Nooruse, EE-50411 Tartu, Estonia
20IRDES, 117 bis rue Manin, FR-75019 Paris, France
21Wissenschaftliches Institut der AOK (WidO), 31 Rosenthaler Straße, DE-10178 Berlin, Germany
22School of Economics and Political Science, University of Athens, Athens
23Pharmaceutical Drug Department, Azienda Sanitaria Locale of Verona, Verona, Italy
24UBT – Higher Education Institute Prishtina, Kosovo
25Institute of Public Health & Faculty of Pharmacy, Riga Stradins University, Latvia
26Department of Pharmacy, Ministry of Health of the Republic of Lithuania, Vilnius, Lithuania
27National Health Care Institute (ZIN), 4 Eekholt, NL-1112 XH Diemen, The Netherlands
28HTA and Reimbursement, Norwegian Medicines Agency, Oslo, Norway
29HTA Consulting, 17/3 Starowis′lna Str, PL-31-038 Cracow, Poland
30Ministry of Health and Social Welfare, Banja Luka, Republic of Srpska, Bosnia and Herzegovina
31University of Banja Luka, Faculty of Medicine, Department of Social Pharmacy, Banja Luka, Republic of Srpska, Bosnia and Herzegovina
32Health Insurance Fund of Republika Srpska, 8 Zdrave Korde, 78000 Banja Luka, Republic of Srpska, Bosnia and Herzegovina
33Agency for medicines and medical devices of Bosnia and Herzegovina, Veljka Mladjenovica bb, 78000 Banja Luka, Bosnia and Herzegovina
34Faculty of Medicine, Public Health and Management Department, ‘Carol Davila’ University of Medicine and Pharmacy Bucharest, RO-050463 Bucharest, Romania
35ZEM Solutions, 9 Mosorska, RS-11 000 Belgrade, Serbia
36Health Insurance Institute, 24 Miklosiceva, SI-1507 Ljubljana, Slovenia
37Faculty of Medicine, Slovak Medical University in Bratislava, Bratislava, Slovakia
38591 Gran Via de les Corts Catalanes, 4a place, ES-08007 Barcelona, Spain
39Stockholm County Council, Stockholm, Sweden
40TLV (Dental and Pharmaceutical Benefits Agency), 18 Fleminggatan, SE-10422, Stockholm, Sweden
41NHS National Services Scotland, Gyle Square, 1 South Gyle Crescent, Edinburgh EH12 9EB, UK
References
1. Kelly RJ, Smith TJ. Delivering maximum clinical benefit at an affordable price: engaging stakeholders in cancer care. Lancet Oncol. 2014;15(3):e112-8.
2. Howard DH, Bach PB, Berndt ER, Conti RM. Pricing in the market for anticancer drugs. J Econ Perspect. 2015;29(1):139-62.
3. Godman B, Wild C, Haycox A. Patent expiry and costs for anticancer medicines for clinical use. Generics and Biosimilars Initiative Journal (GaBI Journal). 2017;6(3):105-6. doi:10.5639/gabij.2017.0603.021
4. Prasad V, Wang R, Afifi SH, Mailankody S. The rising price of cancer drugs-a new old problem? JAMA Oncol. 2016. doi:10.1001/jamaoncol.2016.4275. [Epub ahead of print]
5. Kantarjian HM, Fojo T, Mathisen M, Zwelling LA. Cancer drugs in the United States: Justum Pretium–the just price. J Clin Oncol. 2013;31(28):3600-4.
6. Cohen D. Cancer drugs: high price, uncertain value. BMJ. 2017;359:j4543.
7. Grössmann N, Wild C. Between January 2009 and April 2016, 134 novel anticancer therapies were approved: what is the level of knowledge concerning the clinical benefit at the time of approval? ESMO Open. 2017;1(6):e000125. doi:10.1136/esmoopen-2016-000125.
8. Bach PB, Saltz LB. Raising the dose and raising the cost: the case of pembrolizumab in lung cancer. J Natl Cancer Inst. 2017;109(11).
9. Godman B, Oortwijn W, de Waure C, Mosca I, Puggina A, Specchia ML, et al. Links between pharmaceutical R&D models and access to affordable medicines. A study for the ENVI Committee.
10. Dusetzina SB. Drug pricing trends for orally administered anticancer medications reimbursed by commercial health plans, 2000-2014. JAMA Oncol. 2016;2(7):960-1.
11. Gordon N, Stemmer SM, Greenberg D, Goldstein DA. Trajectories of injectable cancer drug costs after launch in the United States. Journal of clinical oncology. 2017:Jco2016722124.
12. Gyawali B, Sullivan R. Economics of cancer medicines: for whose benefit? New Bioeth. 2017;23(1):95-104.
13. Chalkidou K, Marquez P, Dhillon PK, Teerawattananon Y, Anothaisintawee T, Gadelha CA, et al. Evidence-informed frameworks for cost-effective cancer care and prevention in low, middle, and high-income countries. Lancet Oncol. 2014;15(3):e119-31.
14. Wilking N, Lopes G, Meier K, Simoens S, van Harten W, Vulto AG. Can we continue to afford access to cancer treatment? Eur Oncol Haematol. 2017;13(2):114-9.
15. Lindsley CW. New 2016 data and statistics for global pharmaceutical products and projections through 2017. ACS Chem Neurosci. 2017;8(8):1635-6.
16. Allied Market Research. Oncology/cancer drugs market by therapeutic modalities (chemotherapy, targeted therapy, immunotherapy, hormonal), cancer types (blood, breast, gastrointestinal, prostate, skin, respiratory/lung cancer) – global opportunity analysis and industry forecast, 2013-2020 [homepage on the Internet]. [cited 2019 Mar 14]. Available from: https://www.alliedmarketresearch.com/oncology-cancer-drugs-market?oncology-cancer-drugs-market
17. IMSH Institute for Healthcare Informatics. Global Oncology Trend Report. A review of 2015 and outlook to 2020. June 2016. Available from: https://www.scribd.com/document/323179495/IMSH-Institute-Global-Oncology-Trend-2015-2020-Report
18. Simoens S, van Harten W, Lopes G, Vulto A, Meier K, Wilking N. What Happens when the cost of cancer care becomes unsustainable. Eur Oncol Haematol. 2017;13(2):108-13.
19. Tefferi A, Kantarjian H, Rajkumar SV, Baker LH, Abkowitz JL, Adamson JW, et al. In support of a patient-driven initiative and petition to lower the high price of cancer drugs. Mayo Clin Proc. 2015;90(8):996-1000.
20. Sarwar MR, Iftikhar S, Saqib A. Availability of anticancer medicines in public and private sectors, and their affordability by low, middle and high-income class patients in Pakistan. BMC Cancer. 2018;18(1):14.
21. Atieno OM, Opanga S, Martin A, Kurdi A, Godman B. Pilot study assessing the direct medical cost of treating patients with cancer in Kenya; findings and implications for the future. J Med Econ. 2018;21(9):878-87.
22. Jakupi A, Godman B, Martin A, Haycox A, Baholli I. Utilization and expenditure of anti-cancer medicines in Kosovo: findings and implications. Pharmacoecon Open. 2018;2(4):423-32.
23. Ghinea H, Kerridge I, Lipworth W. If we don’t talk about value, cancer drugs will become terminal for health systems. The Conversation. 2015 Jul 26.
24. Prasad V, De Jesus K, Mailankody S. The high price of anticancer drugs: origins, implications, barriers, solutions. Nat Rev Clin Oncol. 2017;14(6):381-90.
25. Goldstein DA, Clark J, Tu Y, Zhang J, Fang F, Goldstein R, et al. A global comparison of the cost of patented cancer drugs in relation to global differences in wealth. Oncotarget. 2017;8(42):71548-55.
26. World Health Organization. Hoen E’t. Access to cancer treatment: a study of medicine pricing issues with recommendations for improving access to cancer medication [homepage on the Internet]. [cited 2019 Mar 14]. Available from: http://apps.who.int/medicinedocs/documents/s21758en/s21758en.pdf
27. Haycox A. Why cancer? Pharmacoecon. 2016;34(7):625-7.
28. Yang YT, Nagai S, Chen BK, Qureshi ZP, Lebby AA, Kessler S, et al. Generic oncology drugs: are they all safe? Lancet Oncol. 2016;17(11):e493-e501.
29. Godman B, Wettermark B, van Woerkom M, Fraeyman J, Alvarez-Madrazo S, Berg C, et al. Multiple policies to enhance prescribing efficiency for established medicines in Europe with a particular focus on demand-side measures: findings and future implications. Front Pharmacol. 2014;5:106.
30. Godman B, Shrank W, Andersen M, Berg C, Bishop I, Burkhardt T, et al. Comparing policies to enhance prescribing efficiency in Europe through increasing generic utilization: changes seen and global implications. Expert Rev Pharmacoecon Outcomes Res. 2010;10(6):707-22.
31. Garuoliene K, Godman B, Gulbinovic J, Wettermark B, Haycox A. European countries with small populations can obtain low prices for drugs: Lithuania as a case history. Expert Rev Pharmacoecon Outcomes Res. 2011;11(3):343-9.
32. Dylst P, Vulto A, Simoens S. Demand-side policies to encourage the use of generic medicines: an overview. Expert Rev Pharmacoecon Outcomes Res. 2013;13(1):59-72.
33. Dylst P, Vulto A, Godman B, Simoens S. Generic medicines: solutions for a sustainable drug market? Appl Health Econ Health Pol. 2013;11(5):437-43.
34. Babar ZU, Kan SW, Scahill S. Interventions promoting the acceptance and uptake of generic medicines: a narrative review of the literature. Health Policy. 2014;117(3):285-96.
35. Moe-Byrne T, Chambers D, Harden M, McDaid C. Behaviour change interventions to promote prescribing of generic drugs: a rapid evidence synthesis and systematic review. BMJ Open. 2014;4(5):e004623.
36. Kaplan WA, Ritz LS, Vitello M, Wirtz VJ. Policies to promote use of generic medicines in low and middle income countries: a review of published literature, 2000-2010. Health Policy. 2012;106(3):211-24.
37. Wouters OJ, Kanavos P, McKEE M. Comparing generic drug markets in Europe and the United States: prices, volumes, and spending. Milbank Q. 2017;95(3):554-601.
38. Bucek Psenkova M, Visnansky M, Mackovicova S, Tomek D. Drug policy in Slovakia. Value Health Reg Issues. 2017;13:44-9.
39. Kesselheim AS, Misono AS, Lee JL, Stedman MR, Brookhart MA, Choudhry NK, et al. Clinical equivalence of generic and brand-name drugs used in cardiovascular disease: a systematic review and meta-analysis. JAMA. 2008;300(21):2514-26.
40. Lessing C, Ashton T, Davis PB. The impact on health outcome measures of switching to generic medicines consequent to reference pricing: the case of olanzapine in New Zealand. J Prim Health Care. 2015;7(2):94-101.
41. Veronin M. Should we have concerns with generic versus brand antimicrobial drugs? A review of issues. JPHSR. 2011;2:135-50.
42. Paton C. Generic clozapine: outcomes after switching formulations. Br J Psychiatry. 2006;189:184-5.
43. Corrao G, Soranna D, Arfe A, Casula M, Tragni E, Merlino L, et al. Are generic and brand-name statins clinically equivalent? Evidence from a real data-base. Eur J Internal Med. 2014;25(8):745-50.
44. Corrao G, Soranna D, Merlino L, Mancia G. Similarity between generic and brand-name antihypertensive drugs for primary prevention of cardiovascular disease: evidence from a large population-based study. Eur J Clin Invest. 2014;44(10):933-9.
45. Mathews V. Generic imatinib: the real-deal or just a deal? Leuk Lymphoma. 2014;55(12):2678-80.
46. Malhotra H, Sharma P, Bhargava S, Rathore OS, Malhotra B, Kumar M. Correlation of plasma trough levels of imatinib with molecular response in patients with chronic myeloid leukemia. Leuk Lymphoma. 2014;55(11):2614-9.
47. Canadian Association of Pharmacy in Oncology. de Lemos M, Kletas V. Generic imatinib: review of the literature on clinical efficacy [homepage on the Internet]. [cited 2019 Mar 14]. Available from: http://www.capho.org/sites/default/files/nops/Generic%20Imatinib%20Review%20of%20the%20Literature%20on%20Clinical%20Efficacy%20-%20Mario%20de%20 Lemos.pdf
48. Dylst P, Vulto A, Simoens S. Societal value of generic medicines beyond cost-saving through reduced prices. Expert Rev Pharmacoecon Outcomes Res. 2015;15(4):701-11.
49. Woerkom M, Piepenbrink H, Godman B, Metz J, Campbell S, Bennie M, et al. Ongoing measures to enhance the efficiency of prescribing of proton pump inhibitors and statins in The Netherlands: influence and future implications. J Comp Eff Res. 2012;1(6):527-38.
50. Simoens S. International comparison of generic medicine prices. Curr Med Res Opin. 2007;23(11):2647-54.
51. Wouters OJ, Kanavos PG. A comparison of generic drug prices in seven European countries: a methodological analysis. BMC Health Serv Res. 2017;17(1):242.
52. Hill A, Gotham D, Fortunak J, Meldrum J, Erbacher I, Martin M, et al. Target prices for mass production of tyrosine kinase inhibitors for global cancer treatment. BMJ Open. 2016;6(1):e009586.
53. Hill A, Redd C, Gotham D, Erbacher I, Meldrum J, Harada R. Estimated generic prices of cancer medicines deemed cost-ineffective in England: a cost estimation analysis. BMJ Open. 2017;7(1):e011965.
54. Chen CT, Kesselheim AS. Journey of generic imatinib: a case study in oncology drug pricing. J Oncol Pract. 2017;13(6):352-5.
55. Barber M, Gotham D, Hill A. Potential price reductions for cancer medicines on the WHO Essential Medicines List [homepage on the Internet]. [cited 2019 Mar 14]. Available from: http://apps.who.int/medicinedocs/documents/s23154en/s23154en.pdf
56. Guan X, Tian Y, Ross-Degnan D, Man C, Shi L. Interrupted time-series analysis of the impact of generic market entry of antineoplastic products in China. BMJ Open. 2018;8(7):e022328.
57. Matusewicz W, Godman B, Pedersen HB, Furst J, Gulbinovic J, Mack A, et al. Improving the managed introduction of new medicines: sharing experiences to aid authorities across Europe. Expert Rev Pharmacoecon Outcomes Res. 2015;15(5):755-8.
58. Hawkes N. Drug company Aspen faces probe over hiking generic prices. BMJ. 2017;357:j2417.
59. Kahn T. Aspen loses Italy appeal over cancer drug prices. Business Day. 2017 Jun 15.
60. Pagliarulo N. EU investigating generic drugmaker over cancer drug price hikes. Biopharma Dive. 2017 May 15.
61. Godman B, Wilcock M, Martin A, Bryson S, Baumgärtel C, Bochenek T, de Bruyn M. Generic pregabalin; current situation and implications for health authorities, generics and biosimilars manufacturers in the future. Generics and Biosimilars Initiative Journal (GaBI Journal). 2015;4(3):125-35. doi:10.5639/gabij.2015.0403.028
62. McLaughlin M, Kotis D, Thomson K, Harrison M, Fennessy G, Postelnick M, et al. Empty shelves, full of frustration: consequences of drug shortages and the need for action. Hosp Pharm. 2013;48(8):617-8.
63. Putrik P, Ramiro S, Kvien TK, Sokka T, Pavlova M, Uhlig T, et al. Inequities in access to biologic and synthetic DMARDs across 46 European countries. Ann Rheum Dis. 2014;73(1):198-206.
64. Kosti M, Djakovic L, Šuji R, Godman B, Jankovi SM. Inflammatory Bowel Diseases (Crohn’s disease and Ulcerative Colitis): cost of treatment in Serbia and the implications. Appl Health Econ Health Policy. 2017;15(1):85-93.
65. Cameron A, Mantel-Teeuwisse AK, Leufkens HG, Laing RO. Switching from originator brand medicines to generic equivalents in selected developing countries: how much could be saved? Value Health. 2012;15(5):664-73.
66. Wild C, Grössmann N, Bonanno PV, Bucsics A, Furst J, Garuoliene K, et al. Utilisation of the ESMO-MCBS in practice of HTA. Ann Oncol. 2016;27(11):2134-6.
67. Duerden MG, Hughes DA. Generic and therapeutic substitutions in the UK: are they a good thing? Br Clin Pharmacol. 2010;70(3):335-41.
68. Ferner RE, Lenney W, Marriott JF. Controversy over generic substitution. BMJ. 2010;340:c2548.
69. Medicines and Healthcare products Regulatory Agency. Antiepileptic drugs: updated advice on switching between different manufacturers’ products. 2017 [homepage on the Internet]. [cited 2019 Mar 14]. Available from: https://www.gov.uk/drug-safety-update/antiepileptic-drugs-updated-advice-on-switching-between-different-manufacturers-products#chm-review-and-update
70. Godman B, Shrank W, Andersen M, Berg C, Bishop I, Burkhardt T, et al. Policies to enhance prescribing efficiency in Europe: findings and future implications. Front Pharmacol. 2010;1:141.
71. McKee M, Stuckler D, Martin-Moreno JM. Protecting health in hard times. BMJ. 2010;341:c5308.
72. Markovic-Pekovic V, Skrbic R, Godman B, Gustafsson LL. Ongoing initiatives in the Republic of Srpska to enhance prescribing efficiency: influence and future directions. Expert Rev Pharmacoecon Outcomes Res. 2012;12(5):661-71.
73. Moorkens E, Vulto AG, Huys I, Dylst P, Godman B, Keuerleber S, et al. Policies for biosimilar uptake in Europe: an overview. PloS One. 2017;12(12):e0190147.
74. Baumgärtel C, Godman B, Malmström R, Andersen M, Abuelkhair M, Abdu S, et al. What lessons can be learned from the launch of generic clopidogrel? Generics and Biosimilars Initiative Journal (GaBI Journal). 2012;1(2):58-68. doi:10.5639/gabij.2012.0102.016
75. Organisation for Economic Co-operation and Development. OECD Glossary of statistical terms [homepage on the Internet]. [cited 2019 Mar 14]. Available from: http://www.oecd-ilibrary.org/docserver/download/3008121e.pdf?expires=1521443673&id=id&accname=guest&checksum=EDB68C374A8FE629D5A2C1FAFB0AEB6C
76. Coma A, Zara C, Godman B, Agusti A, Diogene E, Wettermark B, et al. Policies to enhance the efficiency of prescribing in the Spanish Catalan region: impact and future direction. Expert Rev Pharmacoecon Outcomes Res. 2009;9(6):569-81.
77. Kanavos P. Generic policies: rhetoric vs. reality. Euro Observer. 2008;10(2):1-6.
78. Piperska Group. Piperska 2017 [homepage on the Internet]. [cited 2019 Mar 14]. Available from: http://www.piperska.org/
79. Garattini S, Bertele V, Godman B, Haycox A, Wettermark B, Gustafsson LL. Enhancing the rational use of new medicines across European health care systems. Eur J Clin Pharmacol. 2008;64(12):1137-8.
80. Moon JC, Godman B, Petzold M, Alvarez-Madrazo S, Bennett K, Bishop I, et al. Different initiatives across Europe to enhance losartan utilization post generics: impact and implications. Front Pharmacol. 2014;5:219.
81. Godman B, Petzold M, Bennett K, Bennie M, Bucsics A, Finlayson AE, et al. Can authorities appreciably enhance the prescribing of oral generic risperidone to conserve resources? Findings from across Europe and their implications. BMC Med. 2014;12:98.
82. Vonina L, Strizrep T, Godman B, Bennie M, Bishop I, Campbell S, et al. Influence of demand-side measures to enhance renin-angiotensin prescribing efficiency in Europe: implications for the future. Expert Rev Pharmacoecon Outcomes Res. 2011;11(4):469-79.
83. Ferrario A, Arja D, Bochenek T, ati T, Dankó D, Dimitrova M, et al. The implementation of managed entry agreements in Central and Eastern Europe: findings and implications. Pharmacoecon. 2017;35(12):1271-85.
84. Godman B, Shrank W, Wettermark B, Andersen M, Bishop I, Burkhardt T, et al. Use of generics – a critical cost containment measure for all healthcare professionals in Europe? Pharmaceuticals. 2010;3(8):2470-94.
85. Godman B, Bucsics A, Burkhardt T, Piessnegger J, Schmitzer M, Barbui C, et al. Potential to enhance the prescribing of generic drugs in patients with mental health problems in Austria; implications for the future. Front Pharmacol. 2012;3:198.
86. Simoens S. A review of generic medicine pricing in Europe. Generics and Biosimilars Initiative Journal (GaBI Journal). 2012;1(1):8-12. doi:10.5639/gabij.2012.0101.004
87. Leopold C, Vogler S, Mantel-Teeuwisse AK, de Joncheere K, Leufkens HG, Laing R. Differences in external price referencing in Europe: a descriptive overview. Health Policy. 2012;104(1):50-60.
88. Leporowski A, Godman B, Kurdi A, MacBride-Stewart S, Ryan M, Hurding S, et al. Ongoing activities to optimize the quality and efficiency of lipid-lowering agents in the Scottish national health service: influence and implications. Expert Rev Pharmacoecon Outcomes Res. 2018;18(6):655-66.
89. WHO Collaborating Centre for Drug Statistics Methodology. ATC/DDD Index [homepage on the Internet]. [cited 2019 Mar 14]. Available from: https://www.whocc.no/
90. Venkatesan S, Lamfers M, Leenstra S, Vulto AG. Overview of the patent expiry of (non-) tyrosine kinase inhibitors approved for clinical use in the EU and the US. Generics and Biosimilars Initiative Journal (GaBI Journal). 2017;6(2):89-96. doi:10.5639/gabij.2017.0602.016
91. Clopes A, Gasol M, Cajal R, Segu L, Crespo R, Mora R, et al. Financial consequences of a payment-by-results scheme in Catalonia: gefitinib in advanced EGFR-mutation positive non-small-cell lung cancer. J Med Econ. 2017;20(1):1-7.
92. Dylst P, Simoens S. Does the market share of generic medicines influence the price level?: a European analysis. Pharmacoecon. 2011;29(10):875-82.
93. BBC News – Health. Cancer drugs price rise ‘costing NHS millions’. 2017 Jan 29.
94. Vella O. Further reductions in medicine prices. Times of Malta. 2017 Jan 15. Available from: https://www.timesofmalta.com/articles/view/20170115/consumer-affairs/Further-reductions-in-medicine-prices.636591
95. Petrova G. Pricing and reimbursement of medicines in Central and East European countries. J Pharmaceut Res Health Care. 2014;2(6);4311-28.
96. Kawalec P, Stawowczyk E, Tesar T, Skoupa J, Turcu-Stiolica A, Dimitrova M, et al. Pricing and reimbursement of biosimilars in Central and Eastern European countries. Front Pharmacol. 2017;8:288.
97. Baltic Statistics on Medicines 2010-2012 [homepage on the Internet]. [cited 2019 Mar 14]. Available from: http://apps.who.int/medicinedocs/documents/s21301en/s21301en.pdf
98. Marcheva M T-KL, Petrova G. Pricing and reimbursement of medicines in Central and East European countries. World Journal of Pharmacy and Pharmaceutical Sciences. 2013;2(6):4311-28.
99. WHO Collaborating Centre for Pharmaceutical Pricing and Reimbursement Policies. Festøy H, Aanes T, Ognøy AE. PPRI Pharma Profile Norway June 2015 [homepage on the Internet]. [cited 2019 Mar 14]. Available from: https://ppri.goeg.at/sites/ppri.goeg.at/files/inline-files/PPRI%20Norway%202018.pdf
100. Godman B, Sakshaug S, Berg C, Wettermark B, Haycox A. Combination of prescribing restrictions and policies to engineer low prices to reduce reimbursement costs. Expert Rev Pharmacoecon Outcomes Res. 2011;11(1):121-9.
101. Kalaba M, Godman B, Vuksanovi A, Bennie M, Malmström RE. Possible ways to enhance renin-angiotensin prescribing efficiency: Republic of Serbia as a case history? J Comp Eff Res. 2012;1(6):539-49.
102. Vogler S, Zimmermann N, Gombocz M, Mörtenhuber S, Skala R, Lichtenecker J, et al. Short PPRI Pharma Profile Austria. January 2018 [homepage on the Internet]. [cited 2019 Mar 14]. Available from: https://ppri.goeg.at/sites/ppri.goeg.at/files/inline-files/Short_PPRI_Pharma_Profile_AT_2017_final_neu_1.pdf
103. Bucsics A, Godman B, Burkhardt T, Schmitzer M, Malmström RE. Influence of lifting prescribing restrictions for losartan on subsequent sartan utilization patterns in Austria: implications for other countries. Expert Rev Pharmacoecon Outcomes Res. 2012;12(6):809-19.
104. Godman B, Abuelkhair M, Vitry A, Abdu S, Bennie M, Bishop I, et al. Payers endorse generics to enhance prescribing efficiency: impact and future implications, a case history approach. Generics and Biosimilars Initiative Journal (GaBI Journal). 2012;1(2):69-83. doi:10.5639/gabij.2012.0102.017
105. Pontén J, Rönnholm G, Skiöld P. PPRI Pharma Profile Sweden. April 2017 [homepage on the Internet]. [cited 2019 Mar 14]. Available from: https://ppri.goeg.at/sites/ppri.goeg.at/files/inline-files/PPRI_Pharma_Profile_Sweden_2017_5.pdf
106. Godman B, Persson M, Miranda J, Skiold P, Wettermark B, Barbui C, et al. Changes in the utilization of venlafaxine after the introduction of generics in Sweden. Appl Health Econ Health Pol. 2013;11(4):383-93.
107. GaBI Online – Generics and Biosimilars Initiative. The Greek problem of generics pricing [www.gabionline.net]. Mol, Belgium: Pro Pharma Communications International; [cited 2019 Mar 14]. Available from: www.gabionline.net/Generics/Research/The-Greek-problem-of-generics-pricing.
108. Vogler S. The impact of pharmaceutical pricing and reimbursement policies on generics uptake: implementation of policy options on generics in 29 European countries–an overview. Generics and Biosimilar Journal. 2012;1(2):93-100. doi:10.5639/gabij.2012.0102.020
109. Dylst P, Vulto A, Simoens S. Analysis of the Italian generic medicines retail market: recommendations to enhance long-term sustainability. Expert Rev Pharmacoecon Outcomes Res. 2015;15(1):33-42.
110. Godman B, Bishop I, Finlayson AE, Campbell S, Kwon HY, Bennie M. Reforms and initiatives in Scotland in recent years to encourage the prescribing of generic drugs, their influence and implications for other countries. Expert Rev Pharmacoecon Outcomes Res. 2013;13(4):469-82.
111. McGinn D, Godman B, Lonsdale J, Way R, Wettermark B, Haycox A. Initiatives to enhance the quality and efficiency of statin and PPI prescribing in the UK: impact and implications. Expert Rev Pharmacoecon Outcomes Res. 2010;10(1):73-85.
112. Bennie M, Godman B, Bishop I, Campbell S. Multiple initiatives continue to enhance the prescribing efficiency for the proton pump inhibitors and statins in Scotland. Expert Rev Pharmacoecon Outcomes Res. 2012;12(1):125-30.
113. UK Departemnt of Health and Social Care. Health service medical supplies (costs) bill factsheet. Updated 8 November 2016 [homepage on the Internet]. [cited 2019 Mar 14]. Available from: https://www.gov.uk/government/publications/health-service-medical-supplies-costs/health-service-medical-supplies-costs-bill-factsheet
114. Bundesministerium für Gesundheit. Preismoratorium für Arzneimittel [homepage on the Internet]. [cited 2019 Mar 14]. Available from: https://www.bundesgesundheitsministerium.de/themen/krankenversicherung/arzneimittelversorgung/preismoratorium.html
115. Bundesministerium für Gesundheit. Sozialgesetzbuch (SGB) Fünftes Buch (V) – Gesetzliche Krankenversicherung – (Artikel 1 des Gesetzes v. 20. Dezember 1988, BGBl. I S. 2477). § 130a Rabatte der pharmazeutischen Unternehmer [homepage on the Internet]. [cited 2019 Mar 14]. Available from: https://www.gesetze-im-internet.de/sgb_5/__130a.html.
116. UK Government. Pharma firms accused of illegal agreements over life-saving drug. 28 February 2019 [homepage on the Internet]. [cited 2019 Mar 14]. Available from: https://www.gov.uk/government/news/pharma-firms-accused-of-illegal-agreements-over-life-saving-drug
117. European Commission. Antitrust: Commission opens formal investigation into Aspen Pharma’s pricing practices for cancer medicines. 2017. Available at URL: file:///C:/Users/mail/Downloads/IP-17-1323_EN.pdf
118. Department of Health. Press Release. Ireland to open negotiations with Belgium, the Netherlands, Luxembourg and Austria on drug pricing and supply – Minister Harris [homepage on the Internet]. [cited 2019 Mar 14]. Available from:http://health.gov.ie/blog/press-release/ireland-to-open-negotiations-with-belgium-the-netherlands-luxembourg-and-austria-on-drug-pricing-and-supply-minister-harris/
119. European Observatory. How can voluntary cross-border collaboration in public procurement improve access to health technologies in Europe? [homepage on the Internet]. [cited 2019 Mar 14]. Available from: http://www.euro.who.int/__data/assets/pdf_file/0009/331992/PB21.pdf?ua=1
120. Ferrario A, Humbert T, Kanavos P, Pedersen HB. Strategic procurement and international collaboration to improve access to medicines. Bull World Health Organ. 2017;95(10):720-2.
121. Vogler S, Zimmermann N, Babar ZU. Price comparison of high-cost originator medicines in European countries. Expert Rev Pharmacoecon Outcomes Res.2017;17(2):221-30.
122. Vogler S, Kilpatrick K, Babar ZU. Analysis of medicine prices in New Zealand and 16 European countries. Value in Health. 2015;18(4):484-92.
123. Bochenek T, Abilova V, Alkan A, Asanin B, de Miguel Beriain I, Besovic Z, et al. Systemic Measures and Legislative and Organizational frameworks aimed at preventing or mitigating drug shortages in 28 European and Western Asian countries. Frontiers in pharmacology. 2017;8:942.
124. Godman B, Bucsics A, Vella Bonanno P, Oortwijn W, Rothe CC, Ferrario A, et al. Barriers for Access to new medicines: searching for the balance between rising costs and limited budgets. Front Public Health. 2018;6:328.
|
Author for correspondence: Brian Godman, BSc, PhD, Strathclyde Institute of Pharmacy and Biomedical Sciences, University of Strathclyde, Glasgow G4 0RE, UK |
Disclosure of Conflict of Interest Statement is available upon request.
Copyright © 2019 Pro Pharma Communications International
Permission granted to reproduce for personal and non-commercial use only. All other reproduction, copy or reprinting of all or part of any ‘Content’ found on this website is strictly prohibited without the prior consent of the publisher. Contact the publisher to obtain permission before redistributing.
Source URL: https://gabi-journal.net/pricing-of-oral-generic-cancer-medicines-in-25-european-countries-findings-and-implications.html
Author byline as per print journal: Steven Simoens, PhD; Claude Le Pen, PhD; Niels Boone, PharmD; Ferdinand Breedveld, MD, PhD; Antonella Celano; Antonio Llombart-Cussac, MD, PhD; Frank Jorgensen, MPharm, MM; Andras Süle, PhD; Ad A van Bodegraven, MD, PhD; Rene Westhovens, MD, PhD; Jo De Cock
|
Introduction/Objectives: This manuscript aims to provide guidance to policymakers with a view to fostering a fair, competitive and sustainable market for off-patent biologicals and biosimilars in Europe. |
Submitted: 6 June 2018; Revised: 13 July 2018; Accepted: 13 July 2018; Published online first: 20 July 2018
Reference biologicals are originator medicinal products made by or derived from living organisms using biotechnology. When the patent and exclusivity rights on a reference biological expire, biosimilar medicines can enter the market. A biosimilar is a biological product that contains a version of the active substance of an already authorized reference biological medicinal product [1]. On the one hand, the European Medicines Agency (EMA) and regulatory authorities guarantee the quality, safety and efficacy of registered biosimilars. After a decade of experience with biosimilar products, no unexpected concerns have emerged [2, 3]. Also, health economists of international organizations refer to the important savings potential of a competitive off-patent biologicals and biosimilars market and to the opportunity to improve patient access. On the other hand, the market access, uptake and price evolution of off-patent biologicals and biosimilars stay heterogeneous between countries and between therapeutic classes, e.g. erythropoiesis-stimulating agents, granulocyte colony-stimulating factors, human growth hormones, antitumour necrosis factors, follitropin alfa and insulins [4].
This implies that European countries are not realizing the full potential of the off-patent biologicals and biosimilars market. In this respect, the European Commission and the Organisation for Economic Co-operation and Development (OECD) have argued that competition in the off-patent biologicals and biosimilars market could yield substantial savings to healthcare systems [5, 6]. To capture these savings, the European Commission (Directorate-General for Internal Market, Industry, Entrepreneurship and small and medium-sized enterprises [SMEs]) has supported a multi-stakeholder approach and has hosted multiple workshops with a view to facilitating access to and uptake of biosimilars [7, 8]. The European Commission also issued a consensus information paper in 2013, an information document for patients in 2016, and an information guide for healthcare professionals in 2017 [8].
However, the development of a competitive market for off-patent biologicals and biosimilars is not certain because of numerous factors including the risk of non-recognition of the difference between biosimilars and generics, physician and patient lack of confidence, and unbalanced payer pricing and procurement policies. In particular, after a decade of experience with these products, it seems clear that a simple logic whereby price reductions multiplied by prescribed volumes of reference biologicals will lead to potential savings is misleading and inappropriate. Price reductions alone do not appear to be the key factor guaranteeing greater market penetration of biosimilars [4]. On the contrary, price reductions can lead to a race to the bottom, preventing manufacturers to enter the market.
It is also important that policymakers keep in mind that biosimilars (where the reference product is a biological medicine) are inherently different from generics (where the reference product is a chemically synthesized medicine), due to their more elaborate size and structure of the molecule, higher risks and costs of research and development, more complex manufacturing processes, extended development times, and the need to institute post-marketing pharmacovigilance programmes [9]. Therefore, policy must be adapted to the specific needs of the off-patent biologicals and biosimilars market.
Developing a competitive market for off-patent biologicals and biosimilars in Europe is a necessary condition for stakeholders to reap the benefits that such competition may create. These benefits include more control of drug expenditure for healthcare payers, expanded access to health care for patients, increased treatment choices for physicians, and headroom for innovation for industry. The development of such a market requires the implementation of a long-term, sustainable and specific policy framework based on a multi-stakeholder approach. Thus, the aim of this manuscript is to provide guidance to policymakers with a view to fostering a fair, competitive and sustainable market for off-patent biologicals and biosimilars in Europe, taking into account the role of all stakeholders.
This manuscript has been commissioned by the Belgian National Institute for Health and Disability Insurance. Individuals and stakeholder representatives from patient groups, clinicians, healthcare professional organizations, government bodies, and industry have participated in a series of roundtable discussions in 2016–2017 which have contributed to the development of the manuscript. These discussions were held under Chatham House Rules. This manuscript represents the authors’ views on the topic which have been informed by their participation in the roundtable discussions and do not represent the position of their respective organization/institution.
When developing guidance to policymakers, the focus of this manuscript is specifically on those product classes for which patent expiry and loss of exclusivity has recently occurred or is imminent, such as monoclonal antibodies for the treatment of inflammatory diseases (rheumatoid arthritis, inflammatory bowel diseases, psoriasis) and cancers.
Supply-side incentives
A building block of a fair, competitive and sustainable market for off-patent biologicals and biosimilars in Europe, see Box 1, relates to the need to put in place appropriate supply-side incentives. In particular, policymakers need to design smart procurement and reimbursement mechanisms with a view to allowing physicians to prescribe off-patent biologicals and biosimilars based on scientific evidence and clinical experience. The design of such mechanisms needs to be anchored in good clinical practice, which will evolve with knowledge, and needs to respect the European legislative framework on public procurement [10, 11]. Physician involvement in procurement and reimbursement mechanisms is vital to ensure that physicians maintain the freedom to prescribe. Also, there is a need to build up technical and practical expertise, and exchange experiences between countries with respect to designing smart procurement and reimbursement mechanisms.
Box 1: Recommendations for developing policy on off-patent biologicals and biosimilars in Europe
In the hospital setting, tendering mechanisms, i.e. public procurement mechanisms for medicines based on competition between pharmaceutical suppliers [12], can be applied to off-patent biologicals and biosimilars nationally or locally, although future studies need to provide guidance to policymakers on how to optimize the features of these mechanisms, such as the frequency of tenders, the criteria to grant the tender, the reward for the winner(s), and the number of winners. For example, tendering may lead to a market where only one medicine is available, i.e. the ‘winner takes all’ principle, and shortages may occur if that supply fails. Thus, tendering mechanisms need to be monitored to ensure that several pharmaceutical suppliers participate and that the market does not fail [13].
In the ambulatory care setting, off-patent biologicals and biosimilars can be included in a reference pricing system, which sets a common reimbursement level for a group of medicines. Policymakers need to appreciate that the application of a reference pricing system could imply that off-patent biologicals and biosimilars included in the same reference group could be used interchangeably (which indicates that a patient can be alternated between products whilst expecting the same clinical outcomes in respect to efficacy and safety as if no alternation were to occur) [14]. In such cases, the relevant policymaker should clarify the status of the medicine.
If implemented, smart tendering mechanisms and/or reference pricing systems need to be sufficiently flexible to permit the prescribing physician to act in the best interests of the patient, including the availability of choice between products. In our opinion, this also implies that physicians are in charge of any switching protocols and that no forced switching occurs. Although procurement and reimbursement mechanisms may generate price competition and produce short-term savings, policymakers also need to consider the possible impact of these mechanisms on the sustainability and the level of competition in the market for off-patent biologicals and biosimilars in the long run.
Demand-side incentives
In our opinion, a key challenge relates to how physicians should use off-patent biologicals and biosimilars in real-life, clinical practice. It is clear that biosimilars can be administered to treatment-naïve patients in all approved indications. However, there is debate and concern about whether it is appropriate to introduce incentives that encourage patients to be switched from a reference biological product to a biosimilar; from one biosimilar to another biosimilar; and about switching on multiple occasions. Although such incentives purport to encourage price competition between manufacturers, there remains residual uncertainty and resistance from different stakeholders to these various forms of switching.
Therefore, we believe that the clinical profession, academia and patients need to be involved in the policy framework for off-patent biologicals and biosimilars, see Box 1. Due to the complex nature of off-patent biologicals and biosimilars, it is clear that the appropriate use of these products needs to be a clinical decision made by a treating physician for an individual patient on the basis of shared decision-making with that patient [15]. The decision needs to balance the physician’s freedom to prescribe with his/her therapeutic and budgetary accountability based on scientific evidence, clinical experience and expert judgement. In order to make an informed and documented decision, physicians need to be supported by developing the evidence base around switching, including real-world data, pharmacovigilance data, switching data and outcome data [16]. Scientific and medical societies have a particular responsibility to provide detailed guidance on appropriate use through position statements [17]. Furthermore, the role of hospital pharmacists is important as they provide a ‘hub of information’ in respect of the uptake, good use and evidence base (monitoring of outcomes, real-world use and reporting of adverse events).
A number of European countries have implemented other physician incentives, such as quota [18]. From those experiences applied in countries with social health insurance systems clearly results that sufficient success will only be realized as far as a formal involvement of the medical profession and other relevant stakeholders is put in place.
In Europe, pharmacist substitution, i.e. the practice of a pharmacist dispensing a different, but similar biological medicine other than that which was prescribed, is a Member State responsibility. Today, the majority of European countries do not favour pharmacist substitution of a biosimilar for a reference biological medicine, although several countries are experimenting with various pharmacist substitution policies [18]. Pharmacist substitution should only be considered in well-defined circumstances, motivated by specific needs and informed by high quality evidence. If a country would consider pharmacist substitution, policymakers need to ensure that any substitution policies guarantee that the prescribing physician is aware of and approves which specific product is dispensed, that pharmacists are trained to provide unbiased information about off-patent biologicals and biosimilars, and that patients are fully informed and agree with substitution.
Gainsharing
A third building block that can align different supply- and demand-side stakeholders in promoting a competitive and sustainable market for off-patent biologicals and biosimilars relates to the recent trend of ‘gainsharing’, see Box 1. Several European countries are experimenting with gainsharing arrangements, which share the savings generated from off-patent biological and biosimilar competition between stakeholders, e.g. healthcare payers, hospitals, physicians and patients [19]. For instance, savings can be invested in supporting physicians and nurses when switching patients or in providing additional services to patients. Today, the experience with gainsharing arrangements is limited and future studies need to investigate the optimal design and impact of such arrangements.
In our opinion, policymakers need to introduce a long-term, sustainable and specific policy framework based on a multi-stakeholder approach with a view to fostering a fair, competitive and sustainable market for off-patent biologicals and biosimilars in Europe. Although there exists residual uncertainty regarding the appropriate terms for switching off-patent biologicals and biosimilars, we believe that such issues will be clarified and resolved over the coming years with the development of new studies, data and experience with these products. Additionally, we advocate that a policy framework for the off-patent biological and biosimilar market needs to be founded on multiple building blocks including the implementation of supply- and demand-side incentives, and the prospective evaluation of gainsharing arrangements.
Competition among off-patent biologicals and biosimilars in Europe benefits patients as it may help to control drug expenditure, expand access to health care, increase treatment choices, and encourage pharmaceutical innovation. However, there remains uncertainty about switching patients from a reference biological product to a biosimilar; from one biosimilar to another biosimilar; and about switching on multiple occasions. Therefore, we believe that the clinical profession, academia and patients need to be involved in developing a policy framework for off-patent biologicals and biosimilars. Due to the complex nature of off-patent biologicals and biosimilars, it is clear that the appropriate use of these products needs to be a clinical decision made by a treating physician for an individual patient on the basis of shared decision-making with that patient.
The authors would like to thank John Bowis, Laura Batchelor and Johan Van Calster from FIPRA for facilitating and chairing the roundtable discussions.
This work was enabled by Amgen, MSD and Pfizer.
Competing interests: SS is one of the founders of the KU Leuven Fund on Market Analysis of Biologics and Biosimilars following Loss of Exclusivity. SS has previously conducted biosimilar research sponsored by Hospira (now Pfizer Inc); and has participated in an advisory board meeting on biosimilars for Pfizer Inc. NB and AvB declare no competing interests. RW is currently principal investigator for Galapagos/Gilead and Celltrion; and has received unrestricted research grants to the KU Leuven from BMS, Janssen and Roche.
Provenance and peer review: Not commissioned; externally peer reviewed.
Professor Steven Simoens, PhD
KU Leuven, Faculteit Farmaceutische Wetenschappen
Department of Pharmaceutical and Pharmacological Sciences
Onderwijs en Navorsing 2, bus 521 49 Herestraat
BE-3000 Leuven, Belgium
Professor Claude Le Pen, PhD
Paris Dauphine University LEGOS Laboratory of Economics and Management of Health Organizations
Place du Maréchal de Lattre de Tassigny FR-75775 Paris Cedex 16, France
Niels Boone, PharmD
Orbis Medical Center
Department of Clinical Pharmacy and Toxicology
PO Box 5500
NL-6130 MB Sittard-Geleen, The Netherlands
Professor Ferdinand Breedveld, MD, PhD
Professor of Rheumatology
European League Against Rheumatism Leiden University Medical Center EULAR/EMA Liaison
83a Rapenburg
NL-2311 GK Leiden, The Netherlands
Antonella Celano, President
Italian National Association of People with Rheumatological and Rare Diseases 16 Via Molise
IT-73100 Lecce (LE), Italy
Antonio Llombart-Cussac, MD, PhD
Medica Scientia Innovation Research (MedSIR ARO)
Barcelona, Spain
Hospital Arnau de Vilanova de Valencia Valencia, Spain
Frank Jorgensen, MPharm, MM
The Hospital Pharmacy Bergen
NO-5085 Bergen, Norway
Andras Süle, PhD
Chief Pharmacist
Péterfy Hospital and Trauma Center 8-20 Peterfy Sandor u
HU-1076 Budapest, Hungary
Ad A van Bodegraven, MD, PhD Gastroenterologist
Department of Gastroenterology, Geriatrics, Internal and Intensive Care Medicine (COMIK)
Zuyderland Medical Centre
PO Box 5500
NL-6130 MB Sittard-Geleen, The Netherlands
Department Gastroenterology
Amsterdam University Medical Centres (AUMC), location Vrije Universiteit
1117 De Boelelaan
NL-1081 HV Amsterdam, The Netherlands
Professor Rene Westhovens, MD, PhD
Professor of Rheumatology
University Hospital Leuven – Rheumatology
49 Herestraat
BE-3000 Leuven, Belgium
Rijksinstituut voor ziekte- en invaliditeitsverzekering
Jo De Cock, Administrator-General 211 Tervurenlaan
BE-1150 Brussels, Belgium
References
1. European Medicines Agency. Guideline on similar biological medicinal products. 2014 [homepage on the Internet]. [cited 2018 Jul 13]. Available from: http://www.ema.europa.eu/docs/en_GB/document_library/Scientific_guideline/2014/10/WC500176768.pdf
2. Madsen S. Improving access to modern therapies: what can we learn from gainsharing practices? The Scandinavian experience. 15th Biosimilar Medicines Conference. 23-24 March 2017, London, UK.
3. Bosco JLF, Bryant A, Dreyer NA. The current biosimilars landscape and methodological considerations for real-world evidence generation. Value Outcomes Spotlight. 2017;3:15-8.
4. QuintilesIMS. The impact of biosimilar competition in Europe. 2017 [homepage on the Internet]. [cited 2018 Jul 13]. Available from: http://www.medicinesforeurope.com/wp-content/uploads/2017/05/IMS-Biosimilar-2017_V9.pdf
5. European Commission. Joint report on health care and long-term care systems & fiscal sustainability: volume 1. Institutional paper 037. 2016 [homepage on the Internet]. [cited 2018 Jul 13]. Available from: https://publications.europa.eu/en/publication-detail/-/publication/d6042a45-b535-11e6-9e3c-01aa75ed71a1/language-en
6. OECD. Tackling wasteful spending on health. Paris: OECD Publishing; 2017.
7. European Commission. Second multi-stakeholder workshop on biosimilar medicinal products. 20 June 2016 [homepage on the Internet]. [cited 2018 Jul 13]. Available from: http://ec.europa.eu/growth/content/second-multi-stakeholder-workshop-biosimilar-medicinal-products_en
8. European Commission. Third stakeholder conference on biosimilar medicines. 5 May 2017 [homepage on the Internet]. [cited 2018 Jul 13]. Available from: http://ec.europa.eu/growth/content/third-stakeholder-conference-biosimilar-medicines-0_is
9. Declerck P, Simoens S. A European perspective on the market accessibility of biosimilars. Biosimilars. 2012;2:33-40.
10. EUR-Lex. Directive 2014/24/EU of the European Parliament and of the Council of 26 February 2014 on public procurement and repealing Directive 2004/18/EC Text with EEA relevance [homepage on the Internet]. [cited 2018 Jul 13]. Available from: http://eur-lex.europa.eu/legal-content/EN/TXT/?uri=celex%3A32014L0024
11. EUR-Lex. Directive 2014/25/EU on procurement by entities operating in the water, energy, transport and postal services sectors [homepage on the Internet]. [cited 2018 Jul 13]. Available from: http://eur-lex.europa.eu/legal-content/EN/TXT/?uri=celex:32014L0025
12. Dylst P, Vulto A, Simoens S. Tendering for outpatient prescription pharmaceuticals: what can be learned from current practices in Europe? Health Policy. 2011;101(2):146-52.
13. Dranitsaris G, Jacobs I, Kirchhoff C, Popovian R, Shane LG. Drug tendering: drug supply and shortage implications for the uptake of biosimilars. Clinicoecon Outcomes Res. 2017;9:573-84.
14. McCamish M, Pakulski J, Sattler C, Woollett G. Toward interchangeable biologics. Clin Pharmacol Ther. 2015;97(3):215-7.
15. Boone NW, Liu L, Romberg-Camps MJ, Duijsens L, Houwen C, van der Kuy PHM, et al. The nocebo effect challenges the non-medical infliximab switch in practice. Eur J Clin Pharmacol. 2018;74(5):655-61.
16. Moots R, Azevedo V, Coindreau JL, Dörner T, Mahgoub E, Mysler E, et al. Switching between reference biologics and biosimilars for the treatment of rheumatology, gastroenterology, and dermatology inflammatory conditions: considerations for the clinician. Curr Rheumatol Rep. 2017;19(6):37.
17. Medicines for Europe. Positioning statements on physician-led switching for biosimilar medicines. [homepage on the Internet]. [updated on April 2018; cited 2018 Jul 13]. Available from: https://www.medicinesforeurope.com/wp-content/uploads/2017/03/M-Biosimilars-Overview-of-positions-on-physician-led-switching.pdf
18. Reiland JB, Freischem B, Roediger A. What pricing and reimbursement policies to use for off-patent biologicals in Europe? – Results from the second EBE biological medicines policy survey. Generics and Biosimilars Initiative Journal (GaBI Journal). 2017;6(2):61-78. doi:10.5639/gabij.2017.0602.014
19. Simon Kucher & Partners. Payers’ price & market access policies supporting a sustainable biosimilar medicines market. Simon Kucher & Partners; 2016.
|
Author for correspondence: Professor Steven Simoens, PhD, KU Leuven, Faculteit Farmaceutische Wetenschappen, Department of Pharmaceutical and Pharmacological Sciences, Onderwijs en Navorsing 2, bus 521, 49 Herestraat, BE-3000 Leuven, Belgium |
Disclosure of Conflict of Interest Statement is available upon request.
Copyright © 2018 Pro Pharma Communications International
Permission granted to reproduce for personal and non-commercial use only. All other reproduction, copy or reprinting of all or part of any ‘Content’ found on this website is strictly prohibited without the prior consent of the publisher. Contact the publisher to obtain permission before redistributing.
Source URL: https://gabi-journal.net/how-to-realize-the-potential-of-off-patent-biologicals-and-biosimilars-in-europe-guidance-to-policymakers.html
|
Abstract: |
Submitted: 29 October 2013; Revised: 13 November 2013; Accepted: 14 November 2013; Published online first: 27 November 2013
Generic drugs are cheaper than originator drugs but of comparable quality to their branded counterparts, and can therefore yield substantial savings. We conducted a comparative analysis of policies on generic medicines in retail markets in Europe, and proposed tools to continue developing them. Here we provide an overview of our research.
European governments have implemented a variety of generic medicine policies, encompassing supply-side policies, i.e. market access, pricing and reimbursement; and demand-side policies, i.e. incentives for physicians, pharmacists and patients. On the supply-side, we analysed market access to generic medicines [1], and identified factors that might delay entry. These included defensive patenting strategies, patent litigations, patent linkage, third-party interventions during the process of obtaining marketing authorization, pricing and reimbursement decisions, and backlogs in national approval systems.
First, we examined the status and effect of generic medicine pricing policies in Europe [2]. Competition from Indian generic medicine manufacturers, European variation in generic medicine prices, and competition between generic medicine manufacturers by discount indicated that the potential savings from generic medicines to healthcare payers and patients were not fully realized in Europe. The European experience also suggests a fragmented approach towards developing generic medicine pricing policies in Europe. We identified a relationship between the market share of generic medicines and the change of their price level [3]. The average price level of generic medicines decreased more in countries with a high generics market share than in those with a low generics market share. In addition, we assessed the experience of tendering programmes for outpatient prescription pharmaceuticals in Europe [4]. Only seven countries had applied this policy for pharmaceuticals in ambulatory care in 2011. Tendering led to drastic reductions of the price level of medicines and generated significant short-term savings in some countries. Several negative long-term consequences were associated with this policy, including drug shortages, closure of domestic manufacturing plants, and closure of pharmacies.
We also analysed reference pricing systems in Europe [5, 6]. We found that reference pricing reduced medicines prices, usually only to the level of the reference price, and increased the use of medicines priced at or below the reference price. No negative effects on health outcomes of patients were reported in the literature. The effect on pharmaceutical expenditures was limited, as no, or only marginal, savings were attained, which also tended to be short term. Reference pricing, however, caused a once only setback of expenditure, whereupon the growth rate returned to its former levels. Although long-term growth of pharmaceutical expenditures was not affected, the setback of expenditures created headroom to finance innovative, expensive medicines.
On the demand side, we conducted a literature review of policies implemented by European governments to encourage the use of generic medicines [7]. We found that positive knowledge and perceptions of generic medicines by all stakeholders are necessary prerequisites to increasing the use of generic medicines. Therefore, governments must initiate appropriate policies to achieve this, e.g. independent academic detailing programmes, continuous medical education events for healthcare professionals, and information campaigns for all stakeholders. These policies should be combined with those designed to facilitate prescribing and dispensing of generic medicines and to increase all stakeholders’ financial responsibility within the healthcare system.
We then analysed the effect of prescribing quota for cheap medicines in Belgium [8]. A cheap medicine was defined in this case as a generic medicine, an original medicine whose price has dropped to the reference price level, or a prescription by International Nonproprietary Name (INN). The policy was successful, as most groups of physicians reached their minimum annual percentages between 2006 and 2009. The percentage of cheap medicines (in defined daily doses) increased from 22.9% in January 2005 to 44.2% of all prescribed medicines in ambulatory care in December 2009. The percentage of generic medicines increased from 12.10% in 2004 to 24.03% of all prescribed medicines in ambulatory care in 2008. Despite the success of this policy, it could be even more successful. The definition of cheap medicines in Belgium currently also includes any prescription by INN, whether or not this actually leads to the dispensing of a lower priced medicine.
We investigated pharmacist remuneration systems, as it is important that pharmacists are not financially penalized for dispensing generic medicines, which has been the case in some European countries [9]. Pharmacist remuneration should move towards a fee-for-performance model instead of a price-dependent model; the latter has been the model of choice in many European countries.
We also reviewed the Spanish generic medicines market, and identified hurdles that impede its development. We found that the market share of generic medicines varied among autonomous communities, demonstrating the importance of demand-side policies. Limited use of generic medicines, drastic reductions of the price level of both innovator and generic medicines, and the erosion of price differences between originator and generic medicines have undermined the economic viability of the Spanish generic medicines market. We make recommendations for increasing the sustainability of the Spanish generic medicines market and the efficiency of the pharmaceutical use within the healthcare system. These include accelerating market entrance of generic medicines, creating a price difference between originator and generic medicines, differentiating between patient co-payment rates between originator and generic medicines, improving physicians’ and patients’ trust in generic medicines, increasing prescribing by INN, deploying electronic prescribing systems, making physicians financially responsible for their prescribing behaviour, and making pharmacists’ remuneration independent of the prices of medicines.
On the basis of our research, we propose recommendations to enhance market access of generic medicines in the European Union. These include establishing a unitary EU patent and a unified and specialized litigation system at European level; faster approval of pricing and reimbursement decisions upon marketing authorization; and a reduction of backlogs in national approval systems.
We make recommendations for governments so that they can continue developing their generic medicine retail markets. These proposed policies have been shown to increase the use of generic medicines effectively, independent of any country-specific characteristics. The recommendations concentrate mainly on demand-side issues, such as increasing prescribing by INN, improving physicians’ training and education in drug selection, implementing electronic prescribing systems for physicians, introducing fee-for-performance systems for pharmacists, and improving patients’ perception of generic medicines. The effect of hospital policies on generic medicines is discussed, as this may affect future medication schemes in ambulatory care.
We conclude by forecasting the future of the generic medicines industry. The industrial landscape for pharmaceutical companies is changing, with a tendency for pharmaceutical companies to combine both originator and generics divisions, and the development of biopharmaceuticals and biosimilars [10]. Future research should focus on the development of generic medicines in emerging markets and country-specific analyses of other European countries with limited use of generic medicines.
Competing interests: Professor Steven Simoens holds the EGA Chair ‘European policy towards generic medicines’. The authors have no conflicts of interest that are directly relevant to the content of this manuscript. This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Provenance and peer review: Commissioned; internally peer reviewed.
Professor Arnold G Vulto, PharmD, PhD, Deputy Head Hospital Pharmacy, Professor of Hospital Pharmacy and Practical Therapeutics, Erasmus University Medical Center, PO Box 2040, 230 Gravendijkwal, NL-3015 CE Rotterdam, The Netherlands.
Professor Steven Simoens, MSc, PhD, Department of Pharmaceutical and Pharmacological Sciences, KU Leuven, Onderwijs en Navorsing 2, PO Box 521, 49 Herestraat, BE-3000 Leuven, Belgium.
References
1. Dylst P, Vulto A, Simoens S. Overcoming challenges in market access of generic medicines in the European Union. J Generic Med. 2012;9(1):21-8.
2. Dylst P, Simoens S. Generic medicine pricing policies in Europe: current status and impact. Pharmaceuticals. 2010;3(3):471-81.
3. Dylst P, Simoens S. Does the market share of generic medicines influence the price level?: a European analysis. Pharmacoeconomics. 2011;29(10):875-82.
4. Dylst P, Vulto A, Simoens S. Tendering for outpatient prescription pharmaceuticals: what can be learned from current practices in Europe? Health Policy. 2011;101(2):146-52.
5. Dylst P, Vulto A, Simoens S. The impact of reference-pricing systems in Europe: a literature review and case studies. Expert Rev Pharmacoecon Outcomes Res. 2011;11(6):729-37.
6. Dylst P, Vulto A, Simoens S. Reference pricing systems in Europe: characteristics and consequences. Generics and Biosimilars Initiative Journal (GaBI Journal). 2012;1(3-4):127-31. doi:10.5639/gabij.2012.0103-4.028
7. Dylst P, Vulto A, Simoens S. Demand-side policies to encourage the use of generic medicines: an overview. Expert Rev Pharmacoecon Outcomes Res. 2013;13(1):59-72.
8. Dylst P, Vulto A, Simoens S. Where a cheap medicine is not the same as a generic medicine: the Belgian case. Journal of Pharmaceutical Health Services Research. 2011;2:185-89.
9. Dylst P, Vulto A, Simoens S. How can pharmacist remuneration systems in Europe contribute to generic medicine dispensing? Pharmacy Pract. 2012;10(1):3-8.
10. Dylst P, Vulto A, Godman B, Simoens S. Generic medicines: solutions for a sustainable drug market? Applied Health Economics and Health Policy 2013; doi:10.1007/s40258-013-0043-z
|
Author for correspondence: Pieter Dylst, PharmD, PhD, Department of Pharmaceutical and Pharmacological Sciences, KU Leuven, Onderwijs en Navorsing 2, PO Box 521, 49 Herestraat, BE-3000 Leuven, Belgium |
|
Call for national news and manuscripts proposals:The Editor of GaBI Journal would like to invite readers to contribute relevant national news from their respective countries for publication in GaBI Journal, as well as proposals for new manuscripts. Please contact the editorial office (editorial@gabi-journal.net) or the Publisher (LT@gabi-journal.net). |
Disclosure of Conflict of Interest Statement is available upon request.
Copyright © 2014 Pro Pharma Communications International
Permission granted to reproduce for personal and non-commercial use only. All other reproduction, copy or reprinting of all or part of any ‘Content’ found on this website is strictly prohibited without the prior consent of the publisher. Contact the publisher to obtain permission before redistributing.
Source URL: https://gabi-journal.net/analysis-of-european-policy-towards-generic-medicines.html
|
Background: Due to the declining innovativeness of the classic R & D model in the original pharmaceutical industry, the generic pharmaceutical industry is aiming to become an innovation generator itself. |
Submitted: 17 December 2012; Revised: 4 February 2013; Accepted: 11 March 2013; Published online first: 15 March 2013
Innovation is widely regarded as an instrument to create competitive advantage. Different types of innovation exist, including incremental innovation, re-innovation and radical innovation. Incremental innovation deals with creating minor improvements or simple adjustments in a product’s current state [1, 2]. Re-innovation has been defined as: ‘the process of innovation and product development that occurs after a new product is launched, building upon early success but improving the next generation with revised and refined features’ [3]. Finally, radical innovation refers to radical, new inventions that produce milestones, new products or services, and as a result lead to the development of new industries [4]. Today there is less radical product innovation in the original pharmaceutical industry. Moreover, the concept of ‘new product’ has also evolved by the application of strategies such as incremental innovation and re-innovation. In the past, radical or disruptive innovation changed the pharmaceutical market, whereas today generic pharmaceutical firms attempt to innovate in a less costly way in a shorter time with less regulatory obstacles due to the substantial R & D costs to achieve a radical new product. Incremental innovation and re-innovation meet these objectives.
The generic pharmaceutical industry is now evolving in an innovative way. Some firms are applying strategic changes in their management systems and business models and creating new product portfolios fortified with ‘super generics’, new chemical entities and novel drug delivery systems. A super generic drug is an improved version of an original drug which has lost product patent protection. The product patent for the original drug will have expired or have been circumvented by the company developing the super generics. The nature of the improvement may include drug delivery, manufacturing or reformulation technology. This kind of value-added version is manufactured in a re-innovation framework. This innovative design is between incremental and radical innovation. Companies producing super generics have a greater regulatory risk in gaining marketing approval compared to strict generics manufacturers [5]. Without getting into the details, there are three regulatory pathways for drug approval in Europe and the US.
The US Food and Drug Administration (FDA) does not recognize the term ‘super generics’. These products are also referred to as ‘added value generics, new therapeutic entities or hybrids’. These products differ from the original product in formulation or method of delivery. These products are improved formulation of a known product.
This group of generics needs a completely New Drug Application (NDA) in order to gain FDA approval. The regulatory pathway in Europe appears to be very similar to that in the US and was introduced within the Directive 2001/83/EC in November 2001 and in the Regulation (EC) No 726/2004. These products are not interchangeable with the brand-name drugs. Those regulatory pathways are summarized in Table 1.
With a NDA, innovative drug therapies are reaching the market in a specific dosage form for one or more clinically proven indications of which, after expiration of the patent or the data exclusivity, copies are launched using Abbreviated New Drug Applications (ANDA). Advanced therapies that emerged from launched molecules during their product life cycle have gained considerable attention as clinical practice provides evidence for additional therapeutic values; patient centric delivery systems show improved therapeutic outcomes or emerging technologies offer efficiency gains in manufacturing or access to emerging markets. The US and European regulatory framework has set reasonable regulations in place for these super generics or hybrid applications. While these regulations are relatively recent the pharmaceutical industry is just starting to use this route for its product development [6, 7].
However, super generics take an average of three to four years development time to registration, and enjoy reduced development and regulatory risks compared to new chemical entities. The end product may gain a significant price premium to conventional generics once marketing approval is received. Depending on the type of modification to the original formulation and whether the super generic drug is being developed for the same or a different indication will also have an impact on the level of additional research that is needed to gain approval for the reformulated product [8]. The quantity of issued patents highlights the technical knowledge and skill sets that are available in generic pharmaceutical firms. The success of these pharmaceutical firms has illustrated the possibility of changing from the classic model of ‘copy maker’ towards a model of creating new value-added products, manufacturing strategies and new business models [9, 10].
Meanwhile, the demand side for pharmaceutical treatments has also evolved. ‘New’ customers have emerged, i.e. a better informed, web data empowered generation of patients searching for cost-effective treatments. The generic pharmaceutical industry is reacting to this by applying new business models.
By applying a patient-centred and quality-based perspective into their business models, the generic pharmaceutical industry is attempting to offer new less risky and cost-effective products. The most important aspect is that innovation is no longer just about the product itself, it is also centred on how a company contributes to improving the health of patients. This process has required the out-licensing of innovative generic drug products and has also involved the establishment of new partnerships and alliances to better utilize technological platforms and manufacturing facilities [11]. As an example, Teva Pharmaceuticals acquired Ivax in 2006, Barr Laboratories in 2008, and Ratiopharm in 2010.
The aim of this article is to gain insight into re-innovation in the generic pharmaceutical industry by focusing on product innovation, and a business model based on value proposition employed by some of the innovative generic pharmaceutical firms. This is an alternative model between hybrid and classic R & D companies.
This research complies with the procedure of Paris Dauphine University not to require consent from an institutional review board when subjects cannot be identified. Also, there are no personal identifiers in the data files or in the results.
We applied a qualitative approach. Semi-structured interviews [12] were conducted because they offer the opportunity to ask experts about their views and experiences of the recent changes. In the absence of studies and documentation on this topic due to its novelty, we conducted interviews with managers, industry consultants, lawyers, physicians, pharmacists, patent attorneys, and researchers to gather more views and share their experiences in this area. We prepared two questionnaires; the original questionnaire was more focused on the intellectual aspect of innovation, trying to investigate about the type of innovation in this industry. In Table 2 we have presented a list of our sources in this qualitative research.
The questionnaire had three parts: the first part was about innovation strategy and how it has influenced the generic drug form; the second part was about innovation in their business model, and how do they boost their model by value proposition to customers; the third part was about innovation in the product portfolio and the reasons of product selection and the use of new technology platforms and new statistical methods to reduce risks and optimize product manufacturing in a shorter time to a quicker access to market. We checked the questions with two researchers who were specialists in survey design and we consulted with an American economist who conducts this type of research in order to validate our questionnaire, some of the questions were added during or after some interviews.
Some new topics emerged during the interviews.From April to October of 2011, a total of 20 interviews were conducted in Basel,Budapest, Paris, at the forum on ‘Biopharmaceuticals and Supergenerics’, and in Frankfurt. There are also some interviews that were conducted by telephone calls to Australia, India, UK and US.
We have also followed relevant forums and conferences in France, Hungary, and International Fairs like the CPhI Worldwide in Frankfurt, Germany, to get up-to-date information.
During the first forum on super generics in Budapest, Hungary, we discussed the regulatory aspect of this innovation in the companies manufacturing innovative products known as super generics, hybrid products and value added generics. In Frankfurt, we met for the first time the specialists we contacted via LinkedIn and by email. It was a unique occasion to meet and discuss with the representatives of the super generics manufacturers worldwide.
We studied almost every day every piece of news related directly or indirectly to our research coming from reliable references. This shows that quality by design (QbD) is a very important concept and that innovative generic drug firms may apply QbD not only to reduce the risk of product failure but also to respond to the demands of FDA.
Several economic and financial reports from Business Insight, Data Monitor, IMS, Ernest & Young, Markets and Research were also reviewed before and during the interviews. The companies that accepted participation in this research are: Mayne Pharma (Australia), Capsugel (Belgium), Biogaran (France), Gedeon Richter (Hungary), Dr Reddy (India) and Hanmi (Korea). The other participants were from drug development companies, consultancy companies, and formulation scientists. Other information was collected using the websites of the associations of generic drugs, such as the European Generic medicines Association (EGA); the generic drug industry association in France (GEMME, Association des professionnels du médicament générique); the American Generic Pharmaceutical Association (GPhA); and the International Generic Pharmaceutical Alliance (IGPA) as part of their insights into 2010 on the generics markets in Europe.
Data analysis
We have constructed a database of our data collected from the interviews. Interviews were recorded, transcribed verbatim and analyzed using the software NVivo 9.2 software according to the Matrix Framework approach. We used NVivo Dataset and survey to explore our findings. In practice we began by coding the ‘raw’ data at nodes representing themes in our text-data. Alternatively, we ran ‘Text’ search query or ‘Word’ frequency to identify common themes in survey responses before coding them. Matrix coding analysis helped us to associate the main results to the three main axes of our research work: Innovation in management system, Business model innovation and Product portfolio innovation. Framework matrices provided a way to summarize or condense the source materials in a grid. Subsequently, we launched questions and found patterns based on our coding and checked for coding consistency among interviewees. This method helped us to compare results, and to identify new perspectives of the survey results that could not be acquired without running the queries and coding the results.
Producing novel products is defined as the part of new product development strategy which explores the extension of existing innovations, which can only happen after the first generation of a new product is launched [13]. This is, for example, the case with the development of super generics and bio-superior products that follow on from reference biopharmaceutical products. Being built upon early successful products, re-innovative products are created through applying new platforms, new components, or new configurations with breakthrough technologies to previous products or manufacturing processes [14, 15]. The new re-innovated medicines are focusing on improving health outcomes for patients.
‘… In the past, successful pharmaceuticals stemmed from having good clinical trial data which companies owned and controlled. In the future, their success in the market will instead be evaluated by post marketing data resulting from patients’ satisfaction, of which they will no longer have sole possession …’ (Pharma Researcher, UK)
At the industrial level, through re-innovation attempts, generic pharmaceutical firms aim to minimize the new product failure rate [7], reduce the cost of developing a new product and decrease the lead time in bringing it to market. A pharmaceutical product developed and manufactured with less excipients and unit operation, while maintaining the product therapeutic performance compared to the originator, could be considered as an improved therapeutic entity as it reduces the overall costs of manufacturing that could lead to reduced healthcare spending [16].
Innovative generic pharmaceutical firms may apply QbD and design of experiment methods to optimize their production outcome and minimize the risk. Quality by design means designing and developing a product and associated manufacturing processes that will be used during product development to ensure that the product consistently attains a predefined quality at the end of the manufacturing process [17]. Statistical methods are becoming increasingly vital for pharmaceutical firms. Design of experiments is a tool for determining the relationship between the factors that have an effect on a process and the response of that process [18].
The re-innovative product (as compared to an incremental new product) can be defined as a product that provides new features, benefits, or improvements through existing technology. As such, re-innovation and incremental innovation are different in two aspects: 1) incremental products are improved only by incremental technologies while breakthrough technologies can be used in re-innovative products; and 2) incremental products must be based on the current platform but re-innovative products are either (mostly) based on a new platform or (occasionally) based on an existing platform [19].
‘… As to technology platforms, if for example you consider aerosolization as a platform, then using such a platform to create new, better forms of an existing entity are part of re-innovation …’ (US Manager, 2012)
Re-innovation by the generic pharmaceutical industry can be observed in drug product design, formulation, process development and manufacturing processes going back to the early stages of the product development cycle.
Some product examples are:
1-Abraxane, super generic form of Taxol (FDA, 2005), which uses albumin to deliver the chemotherapy, not Cremophor, and so avoids hypersensitivity and claims a greater tumour response rate than Taxol. The drug Abraxane (nanoparticle albumin bound paclitaxel) uses the approach of coating Taxol with albumin to reduce the side effects associated with standard Taxol (paclitaxel), making it possible to give it without steroids (which can be a rather bothersome issue for many patients, causing problems from severe insomnia to very high blood sugars and more) and also reducing some other Taxol-associated side effects like joint and muscle aches [20, 21].
2-SUBACAP is an improved version of the conventional itraconazole formulation used to treat fungal infections. In June 2012, Mayne Pharma announced that the UK Medicines and Healthcare products Regulatory Agency (MHRA) had reversed its previous decision on SUBACAP and advised that the SUBACAP marketing authorization application was approvable in the UK. Mayne Pharma is in the process of submitting the response to re-activate the ‘Decentralized Procedure’ to seek approval in Germany, Spain and Sweden. Following approval in these countries, the company will seek a second round of approvals in other European countries, including Belgium, Italy, Greece, Portugal and The Netherlands. The total European market sales of itraconazole in 2011 were US$85 million (companies communication and annual report 2012). SUBACAP provides enhancements to patients and prescribers with reduced inter- and intra-patient variability and therefore a more predictable clinical response enabling a reduction in active drug quantity to deliver therapeutic blood levels. Itraconazole is one of the broadest spectrum antifungal drugs on the market and can be used to treat both superficial fungal infections such as onychomycosis (nail infection) and systemic fungal infections such as histoplasmosis, aspergillosis and candidiasis which can be life threatening to immunocompromised patients [22].
Another example of novel technology platform used in super generic drug manufacturing is the application of nanoparticle technology to address challenges associated with the delivery of poorly soluble compounds. Re-innovation has, for example, led to the development of a tablet dosage form that incorporated candesartan cilexetil nanoparticles [23–28] to reduce dosage, reduce toxicity, improve bioavailability and enhance solubility. The original candesartan cilexetil is used for the treatment of hypertension. The major drawback in the therapeutic efficacy of candesartan cilexetil is its very low aqueous solubility leading to low and variable bioavailability. Low bioavailability may lead to variability in therapeutic response. The formulation change resulting from Design of Experiments and nanoparticle technology resulted in better solubility. Using Design of Experiments for process optimization resulted in a robust scalable manufacturing process with design space established for critical process parameters that can balance milling time, particles size and yield. Design of Experiments studies indicated that, out of the three parameters tested in the experimental design, disc speed, pump speed and bead volume were found to affect the critical product attributes either through non-linear, quadratic or interaction effects [29, 30].
The robustness of the model was validated based on confirmatory trials that indicated statistically no difference between predicted and experimental values. The rate and extent of drug dissolution from tablet dosage form incorporating drug nanoparticles was significantly higher than in the tablet containing micronized drug and marketed product.
The increase in drug dissolution resulted in significant enhancement in rate (Cmax) and extent of drug absorption (AUC).
The manufacturing process used is simple and scalable indicating general applicability of the approach to develop oral dosage forms of sparingly soluble drug.
The formulation approach used provides a viable approach to enhance dissolution and bioavailability of sparingly soluble compounds (BCS class II) that may translate into improved therapeutic outcome [23].
This innovative change is also illustrated by the following quote:
‘Super-generic [drug] products, mostly nano- and micro-sized drug delivery systems, focus on improving active principles which were previously commercialized in another formulation. These new formulations are certainly not bioequivalent in the generic [drug] industry’s sense of the term, they are therefore not generics. They are new, i.e. innovative, drugs, which can replace treatment with the previous entity.’ (Drug Delivery Manager, USA)
Another example of re-innovation in super generic drugs relates to the development of a per oral [29] dosage form for a sparingly soluble camptothecin analogue. This was achieved by formulating it as a drug complex [30]. This formulation approach addressed limitations of the currently marketed product that is only amenable for intravenous administration. The drug complex following oral administration demonstrated safety and efficacy comparable to marketed product in athymic mice with implanted tumours. The manufacturing process used is simple and scalable indicating general applicability of the approach to develop oral dosage forms of sparingly soluble drugs. An oral dosage form should result in lower treatment cost, better patient compliance and improved therapeutic outcome for better disease management [23].
In a recent compliance review for antihypertensive drug treatments it was found that some drug classes have significantly poorer adherence performance by patients than other drug classes. Only one third of patients were adherent to b-blockers and diuretics, while two thirds of patients were adherent to angiotension converting enzyme inhibitors and angiotensin II Receptor blockers [31]. Even an adherence of two-thirds of patients still remains at an unsatisfactory low level and leaves considerable room for improvement.
Modifying the release of drugs that have a short biological half-life by extending their release, circumvents high plasma peaks, reduces fluctuations in plasma levels and allows for a once-daily intake that can optimize therapy. This can avoid the daily oral intake for people with dysphagia or dementia. In this new business model, therapy is moving away from a clinical parameter oriented treatment to an outcome oriented disease management programme [32].
The low price of generic drugs threatens to undermine the sustainability of the generic pharmaceutical industry in regards to its low margins, number of competitors, increased requirements for pharmacovigilance, the mature markets in developed countries, and the post-patent cliff arena after 2015 [8]. Meanwhile, medical and technological changes push the pharmaceutical industry to implement new business models. These changes coincide with a growing demand from ageing populations, and better-informed patients who have a substantial need for individualized cost-effective treatments.
Several generic pharmaceutical firms have evolved their traditional business models into innovative models. These models are key to maintaining market position. They are focused on patients’ unmet medical needs and a high quality approach to the manufacturing process.
The innovative business models emerge from new management systems. The challenge of new management systems in these innovative generic pharmaceutical firms is on product innovation: how to manage a better organization to achieve a maximum product differentiation through value proposition to patients? How to optimize product quality? How to reduce manufacturing costs? How to reduce the time to market?
The generic pharmaceutical industry is evolving into a less generic, but more innovative format. In this respect, it should be noted that many generic pharmaceutical firms have the capacity to re-innovate. They have experience, good knowledge and the technical possibility to re-innovate. Alternatively, new alliances can provide the necessary financial resources for technical and marketing requirements.
Implementing re-innovation as a strategy strives to convert price-focused competition into product quality competition, this is central to an innovative business model. Some generic pharmaceutical firms are re-innovating their product portfolio by using new technology platforms, new components and new configurations. These attempts have mainly resulted in super generic drugs; value added products or hybrid products and biosimilars. These super generic drugs and improved therapeutic entities are an important source for innovation in drug therapy in the coming decades.
The so-called super generics are a promising alternative. The value added products resulting from re-innovation strategy by using new ‘technology platforms’, new components and new services will be a strategic element for affordable and individualized medicines.
According to our findings:
This research may be followed by further investigation in innovative business models adapted by an evolving generic pharmaceutical industry that has not yet been studied. A quantitative study of R & D investment and strategic alliances in an innovative generic pharmaceutical industry will reveal more.
Due to evolution in the pharmaceutical industry landscape, some generic pharmaceutical companies are restructuring their business models. In this new industrial design, some of the generics manufactures are re-inventing their product portfolio through a re-innovation strategy. New technology platforms, new components and new configurations are adopted to provide patient compliance and increase patient quality of life. Super generics, biosimilars, bio-superiors and value added versions are some of the new product alternatives resulting from this innovative evolution. The product itself is not the only target; the conversion of competition from price to product quality ensures the value proposition and provides product differentiation. New innovative product portfolios are the evidence that innovative generics companies are not only mastering incremental innovation but are also adopting re-innovation in their new strategies. In this perspective, biotechnology, nanosciences and nanotechnology are ‘strategic’ areas for its scientific and commercial development.
For patients, the innovative changes in product portfolios struggle to improve patient’s quality of life, reduce side effects and enhance efficiency by new product alternatives. These new product alternatives are developed by applying new technology platforms like nanotechnology. Enhancing drug solubility is often key to improving a product’s formulation. New nanotechnologiesa are now being used to solubilize drugs with the aim of improving bioavailability and activity, and reducing in vivo variabilityb.
The re-innovated product portfolios propose more personalized products according to patient’s unmet medical needs. Non-compliance can be attributed to poor taste, difficulty in administration or swallowing, and the inconvenience of multiple doses per day. Non-compliance is a frequent issue (see Cap Gemini Ernest & Young, Compliance Discovery Workshop, 2003). General reasons for non-compliance include: side effects/adverse events, lack of access, financial constraints and lack of communication or information, poor taste and difficulty to swallow (inconvenience in administration). Compliance is dependent on the class of drugs. Some of the new super generics have the advantage of a lower dose, they have the same positive effect provided by the original version, and have significantly reduced adverse side effects [33]. The importance of the oral route of administration [34] from both a clinician and patient acceptance point of view means there has been a vast amount of development and research in drug delivery via this route. Pharmaceutical devices will continue to drive patient compliance and acceptability. The convergence of microelectromechanical systems and nanotechnology with biological applications offers breakthrough in drug developments [35]. As such, improved therapeutic entities could bring innovation faster and at lower risk to society and help to improve health outcomes [36].
For further reading, please refer to references 37 to 47.
Competing interests: Professor Claude Le Pen is professor of health economics at Paris Dauphine University. He has also several organizational, scientific and governmental responsibilities. Professor Steven Simoens holds the EGA Chair ‘European policy towards generic medicines’. The authors have no conflicts of interest that are directly relevant to the content of this manuscript. This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Provenance and peer review: Not commissioned; externally peer reviewed.
Professor Claude Le Pen, PhD
LEGOS, Laboratory of Economics and Management of Health Organizations, Paris Dauphine University, Place du Maréchal de Lattre de Tassigny, FR-75775 Paris Cedex 16, France
Professor Steven Simoens, MSc, PhD
Department of Pharmaceutical and Pharmacological Sciences, Research Centre for Pharmaceutical Care and Pharmacoeconomics, Katholieke Universiteit Leuven, Onderwijs en Navorsing 2, PO Box 521, 49 Herestraat, BE-3000 Leuven, Belgium
References
1. Banbury C, Mitchell W. The effect of introducing important incremental innovations on market share and business survival. Strategic Management Journal. Special Issue. 1995;16 S1:161-82. doi:10.1002/smj.4250160922
2. Henderson RM, Clark KB. Architectural innovation: the reconfiguration of existing product technologies and the failure of established firms. Adminstrative Science Quarterly. 1990;35(1):9-30.
3. Rothwell R, Gardiner P. The strategic management of re-innovation. R & D Management. 1989;19(2):147-60.
4. Cheng C, Shiu E. Preconditions for product innovation in Taiwan: evidence from high technology firms, Asia Pacific Business Review. 2008;14(2):191-211.
5. Parrek A. A cost effective clinical trial procedures to ensure the smooth development and approval of super generics. 2nd Annual NextGen 2011: Super-generics Strategies. 27–29 April 2011, Mumbai, India. Data on file.
6. Nadler HL, Degraft-Johnson D. Demystifying FDA’s 505(b)(2), drug registration process. Regulatory Affairs Professionals Society (RAPS); 2009.
7. Stegemann S, Klebovich I, Anta I, et al. Improved therapeutic entities derived from known generics as an unexplored source of innovative drug products. Eur J Pharm Sci. 2011;44(4):447-54. doi:10.1016/j.ejps.2011.09.012
8. Buggle I. Marketing opportunities for supergenerics. Business Insight. 2010;38-46. Data on file.
9. Sommerfeld T. Strategic options for mid-sized generic players. Journal of Generic Medicines. 2005;2(4):308-15.
10. Kermani F. Generic companies: aspiring to innovate. Journal of Generic Medicines. 2004;1(4):336-46.
11. The significance and apparent repercussions of 2009–2015 pharmaceutical patent cliff. Bioassociate. Innovative Consulting; 2012.
12. Pope C, Mays N. Qualitative research in health care. 3rd ed. Blackwell Publishing: Oxford; 2008. p. 22-43.
13. Cheng C, Shiu E. Re-innovation – what is it and what could influence its performance? Academy of Marketing Conference; 2006: London, UK. Conference Paper No. 254.
14. Rothwell R, Gardiner P. Invention, innovation, re-innovation and the role of the user: A case study of the British Hovercraft development. Technovation. 1985;3(3):167-86.
15. Rothwell R, Gardiner P. Re-innovation and robust designs: producer and user benefits. Journal of Marketing Management. 1988;3(3):372-87.
16. Yu LX. Pharmaceutical quality by design: product and process development, understanding, and control. Pharm Res. 2008;25(4):781-91.
17. Anderson MJ, Whitcomb P. Design of experiments. In: Kirk-Othmer Encyclopedia of Chemical Technology. Wiley; 2010. p. 1-22.
18. Dosage forms and routes of administration [homepage on the Internet]. [cited 2013 Feb 4]. Available from: scribd.com/doc/22880389/Dosage-Forms-and-Routes-of-Administration
19. Nekkanti V, Karatgi P, Paruchuri S, et al. Drug product development and pharmacological evaluation of a sparingly soluble novel camptothecin analog for peroral administration. Drug Delivery. 2011;18(4):294-303.
20. Drug delivery innovations driving improved patient compliance [homepage on the Internet]. C2011 [cited 2013 Feb 4]. Available from: http://www.marketresearch.com/product/display.asp?ProductID=6481767
21. Abraxane bests taxol in response rate for nsclc: what might this mean? [homepage on the Internet]. 2010 [cited 2013 Feb 4]. Available from: http://cancergrace.org/lung/2010/03/20/prelim-abraxane-vs-taxol-rr-results/
22. Mayne Pharma reports final FY12 result. Melbourne, Australia: Australian Securities Exchange. 2012 Aug 21 [cited 2013 Feb 4]. Available from: http://www.asx.com.au/asxpdf/20120821/pdf/4284tjk7gfxtsc.pdf
23. New formulation technologies to fast track development of super generics. Pillai R. Dr Reddy’s Laboratories, 2011. Data on file.
24. Basa S, Muniyappan T, Karatgi P, Prabhu R, Pillai R. Production and in vitro characterization of solid dosage form incorporating drug nanoparticles. Drug Dev Ind Pharm. 2008;34(11):1209-18.
25. Nekkanti V, Karatgi P, Joshi M, Pillai R. Developing nanoparticle formulations of poorly soluble drugs. Pharm Tech Europe. 2008 Nov;20(11):24.
26. Nekkanti V, Pillai R, Venkateswarlu V, Harisudhan T. Development and characterization of solid oral dosage form incorporating candesartan nanoparticles. Pharm Dev Technol. 2009;14(3):290-8.
27. Singare DS, Marella S, Gowthamrajan K, Kulkarani GT, Vooturi R, Srinivasa P, Rao PS. Optimization of formulation and process variable of nanosuspension: an industrial perspective. Int J Pharm. 2010;402(1-2):213-20.
28. Nekkanti V, Venkateswarlu V, Ansari KA, Pillai R. Development and pharmacological evaluation of a PEG based nanoparticulate camptothecin analog for oral administration. Curr Drug Deliv. 2011 Nov;8(6):661-6.
29. Nekkanti V, Muniyappan T, Karatgi P, Hari MS, Marella S, Pillai R. Spray-drying process optimization for manufacture of drug-cyclodextrin complex powder using design of experiments. Drug Dev Ind Pharm. 2009; 35(10):1219-29.
30. Nekkanti V, Karatgi P, Paruchuri S, Pillai R. Drug product development and pharmacological evaluation of a sparingly soluble camptothecin analog for peroral administration. Drug Deliv. 2011 May;18(4):294-303.
31. Kronish IM, Woodward M, Sergie Z, Ogedegbe G, Falzon L, Mann DM. Meta-analysis: impact of drug class on adherence to antihypertensives. Circulation. 2011;123(15):1611-21.
32. Stegemann et al. Improved therapeutic entities derived from known generics as an unexplored source of innovative drug products. Eur J Pharm Sci. 2011;44:447-54.
33. Cuba develops its first industrial-scale nanopharmaceutical. Latin American Herald Tribune. 2013 Feb 12. Available from: http://www.laht.com/article.asp?ArticleId=587208&CategoryId=14510
34. Pareek A. Leveraging innovation in generics: a business imperative for pharmaceutical companies. First conference on Supergeneric innovation 2010; Mumbai, India.
35. Ross MS. Innovation strategies for generic drug companies: moving into supergenerics. IDrugs. 2010 Apr;13(4):243-7.
36. Gassmann O, Reepmeyer G, Zedtwitz M. Leading pharmaceutical innovation. 2nd ed. Berlin: Springer Verlag; 2004. Chapter 2: Pharmaceutical innovation: the case of Switzerland, p. 23-52, chapter 3: The science and technology challenges, how to find new drugs? P. 53-77.
37. Kale D. Learning to innovate: the Indian pharmaceutical industry response to emerging TRIPs regime. DRUID Academy Winter 2005 PhD Conference; 2005: Aalborg, Denmark.
38. Simoens S, De Coster S. Sustaining generic medicines markets in Europe. Journal of Generic Medicines. 2006;3(4):257-68.
39. Le Pen C. Consommation pharmaceutique et indicateurs de santé publique. Leem. 2009.
40. Ring in generics in 2012 and beyond. Zacks Equity Research – 2011 Dec 29 [cited 2013 Feb 4]. Available from: http://www.zacks.com/stock/news/67042/Ring+in+Generics+in+2012+and+Beyond
41. Dingermann T. Innovator companies should focus on innovations. Generics and Biosimilars Initiative Journal (GaBI Journal). 2012;1(1):6. doi:10.5639/gabij.2012.0101.003
42. Class JN, Langis l. A patient-centred paradigm for the biosimilar market? Generics and Biosimilars Initiative Journal (GaBI Journal). 2012;1(1):17-21. doi:10.5639/gabij.2012.0101.006
43. Van Lerberghe W. The world health report 2008. Primary health care [homepage on the Internet]. 2013 [cited 2013 Feb 4]. Available from: http://www.who.int/whr/2008/en
44. Shaw B. The role of the interaction between the user and the manufacturer in medical equipment innovation. R & D Management. 1985;15(4):283-92.
45. Terziovski M. Building innovation capability in organizations: an international cross-case perspective. Imperial College Press, London; 2007. p. 157-75.
46. Cheng CJ, Shiu E. Re-innovation: the construct, measurement, and validation. Technovation. 2008;28(10):658-66.
47. Miele E, Spinelli GP, Miele E, Tomao F, Tomao S. Albuminbound formulation of paclitaxel (Abraxane ABI-007) in the treatment of cancer. Int J Nanomedicine. 2009;4:99-105.
aNanotechnology is the science of manipulating materials on a scale so small that they cannot be seen with a regular microscope. The technology could have a broad range of applications, such as increasing the effectiveness of a particular drug, improving the packaging of food, or altering the look and feel of a cosmetic. Nanotechnology could also be used in medicines designed for the detection, treatment, and prevention of disease; food production and preservation; water decontamination and purification; environmental remediation; lighter and stronger materials for construction and transportation; and energy resources such as solar cells and fuel-efficiency additives, just to name a few. Paul C Howard, PhD, Director of the Office of Scientific Coordination and Director of the Nanotechnology Core Facility at FDA’s National Center for Toxicological Research, FDA website, 2011.
bSkyepharma and Elan have developed technologies that have been used to reposition well-known drugs (fenofibrate, megestrol and sirolimus).
|
Author for correspondence: Fereshteh Barei, PhD, LEGOS – Laboratory of Economics and Management of Health Organizations, Paris Dauphine University, Place du Maréchal de Lattre de Tassigny, FR-75775 Paris Cedex 16, France |
Disclosure of Conflict of Interest Statement is available upon request.
Copyright © 2013 Pro Pharma Communications International
Permission granted to reproduce for personal and non-commercial use only. All other reproduction, copy or reprinting of all or part of any ‘Content’ found on this website is strictly prohibited without the prior consent of the publisher. Contact the publisher to obtain permission before redistributing.
Source URL: https://gabi-journal.net/the-generic-pharmaceutical-industry-moving-beyond-incremental-innovation-towards-re-innovation.html
Author byline as per print journal: Pieter Dylst, PharmD; Professor Steven Simoens, MSc, PhD; Professor Arnold G Vulto, PharmD, PhD
|
Introduction: A reference pricing system is a system that establishes a common reimbursement level or reference price for a group of interchangeable medicines, i.e. the reference group. This article provides an overview of the different characteristics of the different reference pricing systems in Europe. Additionally, the impact of reference pricing on price competition, generic medicine use, pharmaceutical expenditure and health outcome will be discussed. |
Submitted: 16 May 2012; Revised: 8 August 2012; Accepted: 15 August 2012; Published online first: 17 August 2012
A reference pricing system is a system that establishes a reimbursement level or reference price for a group of interchangeable medicines. If a medicine is priced above the reference price, the patient pays the difference between the price of the medicine and the reference price, in addition to any other co-payments, e.g. prescription fee, percentage co-payment [1].
Unlike its name suggests, a reference pricing system is not a pricing system, but in fact a reimbursement system. A reference pricing system sets a common reimbursement level, i.e. reference price, for a group of medicines, thereby generating savings for the third-party payer. Manufacturers are in principle free to set prices, although medicines priced above the reference price incur an additional patient co-payment and generic medicines in some countries, e.g. Belgium, need to be priced below the reference price in order to be reimbursed.
Reference pricing can help governments to contain public pharmaceutical expenditure as it controls the reimbursement level of medicines. A reference pricing system may also promote generic medicine use because originator medicines priced above the level of the reference price are likely to lose market share as a result of the additional patient co-payment.
Many European countries have already installed a reference pricing system, see Table 1. Sweden had adopted a reference pricing system in 1993 but abandoned this in 2002 [2]. In Norway, reference pricing applied from 1993 until the end of 2000. In 2003, the Norwegian government installed a system called ‘index pricing’ to a set of off-patent medicines, which has many resemblances with a reference pricing system [3, 4]. Reference pricing is in many European countries combined with other policies such as prescribing by international non-proprietary name or generics substitution, as this combination of policies seems to positively influence each other [5].
This article describes the characteristics of the different reference pricing systems in Europe. Also, the impact of reference pricing on price competition, generic medicine use, pharmaceutical expenditure and health outcome will be discussed. To this effect, a literature review and survey were carried out. The survey was used to document the current status of reference pricing systems in Europe. Survey data were collected from member associations of the European Generic medicines Association in the context of their 2011 survey of European drug retail markets [6].
Table 2 gives an overview of the different methods used by European countries to set reference prices. A country may employ one or a combination of method(s) to establish reference prices. The reference price is generally calculated as a function of market prices of medicines. The medicines which are taken into account for the calculation of these reference prices can differ between countries, see Table 2.
The different methods of setting reference prices need to be considered in the context of price competition and the volume of generic medicine use. Some European countries set the reference price at the average price level of (generic) medicines in the reference group. If accompanied by incentives to stimulate demand for generic medicines, generic medicine companies have an incentive to compete, thereby driving down (reference) prices of medicines.
Once the generic medicines market reaches a minimum level of development—for instance, a generic drug market share by volume of 40%—the reference price can be further reduced and set at the level of the lowest priced (generic) medicine. For instance, the high generic medicines market share in combination with reference prices based on the lowest priced medicines in Poland support price competition between companies and maximize savings from generic medicines use. However, other countries such as Italy and Spain have set the reference price based on the lowest priced medicine in the absence of strong incentives to stimulate demand for generic medicines, thereby undermining the development of the generic medicines market [9].
Conversely, in countries with developing generic medicines markets, setting the reference price at a higher level to encourage generic medicines market entry can be introduced as a temporary measure to boost the generic medicines market until it reaches a more mature level of development [9]. For instance, when the reference pricing system was first introduced in Portugal in 2003, the reference price was established at the level of the most expensive generic medicine. This approach increased the number of generics competitors and competition in the generic medicines market [10].
This recommendation reflects the approach used in Germany, where the reference price is calculated as a function of medicine prices and the number of generics competitors. Higher reference prices are awarded in reference groups with fewer generics competitors, thereby stimulating market entry of generic medicines companies. Conversely, reference prices are reduced and price competition is stimulated in established markets, but not to the extent that it becomes economically unviable for generic medicines companies to remain on the market [11].
In Belgium, the reference price is set at 69% (imposed by the government) of the price of the originator medicine on the day that the patent expires. This method has the benefit of guaranteeing savings to health insurance funds, but has in general not generated price reductions of generic medicines below the reference price [12].
In a reference pricing system, equivalent medicines are put together in a reference group as defined by:
The term ‘generic reference pricing’ refers to a level 1 reference group, whereas ‘therapeutic reference pricing’ relates to level 2 and 3 reference groups [13].
The methods for establishing reference groups in European countries are outlined in Table 3. Countries can implement a combination of these methods to group medicines in reference groups.
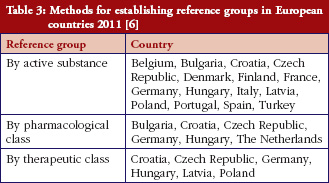 [6]
[6]
The majority of countries group medicines by active substance, see Table 2. Such a method may lead to ‘re-allocation of demand’ away from off-patent medicines included in the reference pricing system towards patented medicines with a similar therapeutic indication that do not fall under the reference pricing system [9].
Re-allocation of demand occurred in Italy in the case of ranitidine: the falling market share of originator ranitidine following the advent of generic medicines was offset by increasing sales of patented medicines with the same therapeutic indication, e.g. omeprazole and its derivatives. Estimates suggest that re-allocation of demand was responsible for an increase in public pharmaceutical expenditure by 3.1% in 2003 [14].
Following the patent expiry of omeprazole in The Netherlands, a higher percentage of patients switched to another proton pump inhibitor (such as pantoprazole and esomeprazole, which are not included in the reference pricing system) than before the patent expiry of omeprazole [15].
In the Valencian region of Spain, the inclusion of fluoxetine in the reference pricing system and the market entry of generics competitors resulted in a shift away from fluoxetine to other antidepressants, e.g. escitalopram, venlafaxine, which were not included in the reference pricing system [16].
In countries that define reference groups by pharmacological class or by therapeutic class, see Table 3, the heterogeneity of medicines within the reference group increases. As a result, physicians may be incited to prescribe a specific medicine for financial reasons, i.e. avoidance of patient co-payment, rather than for clinical reasons, i.e. effectiveness, safety, drug–drug interaction profile [9].
Differences exist between European countries with respect to the consideration of dosage equivalence when establishing reference groups [13]. If there is no differentiation between dosages, e.g. 10 mg, 20 mg and 40 mg of the same active substance are included in the same reference group; 1 mg of active substance will be the cheapest in the highest dosage formulation. Thus, patients have an incentive to switch to the highest dosage of the medicine with the lowest co-payment. Alternatively, reference groups may be defined based on the defined daily dose of medicines. However, one defined daily dose of a specific medicine may not be therapeutically equivalent with one defined daily dose of another medicine in the same reference group. Defined daily doses of medicines also might change over time. World Health Organization therefore stresses that the defined daily dose methodology should not be misused for pricing and reimbursement decisions [17].
Hungary already applied a reference pricing system by active substance since 1997 and introduced a reference pricing system by therapeutic class for, amongst other medicine classes, statins in 2003. Statins were put together in one reference group based on their mechanism of action, without taking into account differences in pharmacologic profile, safety and effectiveness between individual products. As the reference price was determined based on the price per mg, the reference pricing system financially promoted the use of higher-dosed statins such as atorvastatin 40 mg and fluvastatin 80 mg. As a result, the majority of patients switched to higher-dosed statins and the anticipated reduction in the average price of prescribed statins did not materialize [18].
What is the impact of a reference pricing system on price competition? The literature suggests that the introduction of a reference pricing system reduces prices of all medicines that are included in the system [19, 20]. Obviously, price reductions tend to be larger for originator medicines than for generic medicines. Also, greater price reductions have been witnessed in markets where generic medicine competition already occurred prior to the introduction of a reference pricing system [19].
The European experience with respect to price reductions below the reference price is mixed: such price competition has occurred in some countries, e.g. Lithuania [20], but not in others, e.g. Slovenia, Spain [21]. Conversely, some medicines that were originally priced below the reference price actually increased their prices to the level of the reference price following the implementation of a reference pricing system in The Netherlands [22] and in Spain [23]. Therefore, in addition to setting reference prices as a function of the prices of existing medicines, some countries have established a fixed minimum price difference between generic and originator medicines, e.g. Finland, Portugal, Spain [6].
The number of generic medicine competitors within a reference pricing system appears to be associated with the extent of competition, although this impact occurs mainly through discounting to the distribution chain rather than through price competition. Larger discounts have been observed when there are more generic medicine competitors [20, 24].
Finally, as compared with other mechanisms to regulate prices such as price caps i.e. a system where the regulator sets a maximum price that can be charged for a medicine, a Norwegian study reports that a reference pricing system stimulates generics competition to a greater extent and leads to lower prices than price cap regulation [3].
In Germany, medicine prices dropped by 10–26% in the first years following the introduction of the reference pricing system in 1989. This price reduction was greater for originator medicines that faced more generic medicines competition. However, price reductions were counterbalanced by an increase in prices of medicines that were not included in the reference pricing system [25, 26]. In Norway, the implementation of a system called ‘index pricing’ to a set of off-patent medicines in 2003, which has many resemblances with reference pricing, reduced prices of originator medicines by 18% and prices of generic medicines by 8% [3]. In Sweden, medicines covered by the reference pricing system saw a drop in prices by 19% following the introduction of the system [27].
In general, the implementation of a reference pricing system has been accompanied by an increase in the use of medicines priced at or below the reference price [13]. However, a literature review indicated that a reference pricing system does not aid generic medicines use if:
In Germany, therapeutic reference pricing applies to the group of statins since 2005. As the manufacturer of atorvastatin claimed that atorvastatin was superior to other statins in terms of side effects and drug interaction profile, the manufacturer kept the price of atorvastatin above the reference price. As a result, the market share of atorvastatin declined from 33.3% prior to the reference pricing system to 4.8% in 2006. This volume shifted mainly towards simvastatin to which no additional patient co-payment was applicable as its price was below the reference price [28].
A literature review has indicated that reference pricing systems generate savings in the short term, but that savings are probably limited to the one-off impact of the introduction of the reference pricing system and that savings may be substantial at the level of individual medicines, but not necessarily at the level of total pharmaceutical expenditure [19]. Figure 1 shows how the introduction of a reference pricing system may impact long-term pharmaceutical expenditure [29].
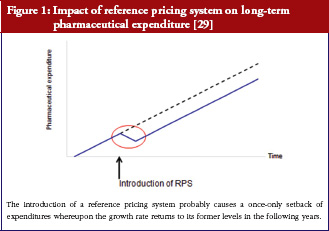 [29]
[29]
The limited scope for savings is to be expected for at least two reasons [29]. First, the coverage of reference pricing systems tends to be limited to off-patent medicines. Patented medicines are included in the reference pricing system by therapeutic class in some countries only. Second, although a reference pricing system may reduce medicine prices and may affect the substitution of new more expensive medicines for older less expensive medicines, it does not affect other drivers of increasing pharmaceutical expenditure such as medicines use by ageing populations, the introduction of expensive biotechnology medicines and orphan medicines, and the transformation from acute to chronic diseases.
A potential concern of therapeutic reference pricing is that switches in medicine use in response to the reference pricing system may adversely affect patient health outcomes and thereby increase the use (and costs) of other healthcare services. There are few studies that explore the impact of reference pricing systems on health and they suffer from methodological limitations. Nonetheless, the available evidence suggests that there is no association between reference pricing systems and health outcomes [13, 19].
A related issue is whether a reference pricing system influences access to health (care) of patient groups with a different socio-economic status. For instance, are patients with a lower socio-economic status more likely to buy costly originator medicines (incurring an additional patient co-payment) in a reference pricing system, thereby placing a financial burden on patient groups with a lower ability to pay? Very few studies have investigated this issue, but a recent Belgian study showed that patients with a lower socio-economic status tended to buy the cheapest (generic) medicines within a reference group and incurred lower medication costs than patients with a higher socio-economic status, thereby laying to rest potential socio-economic equity concerns related to reference pricing systems [8].
Reference pricing is used by many European countries as one element of governments’ strategies to contain public healthcare expenditure. It puts pressure on pharmaceutical companies to compete with the reference priced product but also reduces competition beyond this reference price. It also makes patients sensitive of drug prices, as an increased use of drugs priced at or below the reference price level was observed. The introduction of reference pricing generates a once-only setback of expenditures but does not affect the overall growth rate of health expenditure in the long term. No association between reference pricing and health outcome has been observed.
Over the last decades, European healthcare budgets have been increasing. Therefore, governments have implemented reference pricing, amongst other measures, in order to contain the pharmaceutical budget. In the reference pricing system, a common reimbursement level is set for a group of comparable and interchangeable medicines. The difference between this reimbursement level and the actual price of the medicine is paid by the patient, a so-called co-payment. This provides patients with a financial incentive to opt for the least expensive medicine. Reference pricing seems to reduce prices of medicines, increases the use of cheaper medicines, i.e. without co-payment, generates savings for healthcare budgets in the short term and does not seem to have a negative influence on the health of patients.
Competing interests: Professor Steven Simoens holds the EGA Chair ‘European policy towards generic medicines’. The authors have no conflicts of interest that are directly relevant to the content of this manuscript. This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Provenance and peer review: Commissioned; externally peer reviewed.
The paper is abstracted based on the presentation at the 24th Annual EuroMeeting, Drug Information Association, 28 March 2012, Copenhagen, Denmark.
Professor Steven Simoens, MSc, PhD, Research Centre for Pharmaceutical Care and Pharmacoeconomics, Katholieke Universiteit Leuven, Onderwijs en Navorsing 2, PO Box 521, 49 Herestraat, BE-3000 Leuven, Belgium
Professor Arnold G Vulto, PharmD, PhD, Deputy Head Hospital Pharmacy, Professor of Hospital Pharmacy and Practical Therapeutics, Erasmus University Medical Center, PO Box 2040, NL-3000 CA Rotterdam, The Netherlands
References
1. Folino-Gallo P, Muscolo L, Vogler S, Morak S. PHIS Glossary: Glossary for pharmaceutical policies/systems developed in the Pharmaceutical Health Information Systems (PHIS) Project. PHIS/AIFA/GÖG; July 2009 [update 2011 Apr].
2. Vogler S, Habl C, Leopold C, Rosian-Schikuta I, de Joncheere K, Thomsen TL. PPRI Report. Vienna, Austria: Commissioned by European Commission, Directorate-General Health and Consumer Protection and Austrian Federal Ministry of Health, Family and Youth; 2008.
3. Brekke K, Grasdal A, Holmås TH. Regulation and pricing of pharmaceuticals: Reference pricing of price cap regulation? Eur Econ Rev. 2009;53:170-85.
4. Håkonsen H, Horn AM, Toverud E-L. Price control as a strategy for pharmaceutical cost containment – What has been achieved in Norway in the period 1994–2004? Health Policy. 2009;90(2):277-85.
5. Vogler S. The impact of pharmaceutical pricing and reimbursement policies on generics uptake: implementation of policy options on generics in 29 European countries—an overview. Generics and Biosimilars Initiative Journal (GaBI Journal). 2012;1(2):93-100. doi:10.5639/gabij.2012.0102.020
6. European Generic medicines Association. 2011 Market Review. Brussels, Belgium: European Generic medicines Association; 2011.
7. Vogler S, Habl C, Bogut M, Voncina L. Comparing pharmaceutical pricing and reimbursement policies in Croatia to the European Union Member States. Croat Med J. 2011;52:183-97.
8. Vrijens F, Van de Voorde C, Farfan-Portet M-I, le Polain M, Lohest O. The reference price system and socio-economic differences in the use of cheap medicines. Brussels, Belgium: KCE; 2010. Report No.: 126A.
9. Simoens S, De Coster S. Sustaining generic medicines markets in Europe. Journal of Generic Medicines. 2006;3(4):257-68.
10. Portela C. Reference pricing system and competition: case study from Portugal. Croat Med J. 2009;50(5):429-39.
11. Stargardt T, Schreyögg J, Busse R. [Pharmaceutical reference pricing in Germany: definition of therapeutic groups, price setting through regression procedure and effect]. Gesundheitswesen. 2005;67(7):468-77.
12. Simoens S, De Bruyn K, Bogaert M, Laekeman G. Pharmaceutical policy regarding generic drugs in Belgium. Pharmacoeconomics. 2005;23(8):755-66.
13. Dylst P, Vulto A, Simoens S. The impact of reference-pricing systems in Europe: a literature review and case studies. Expert Rev Pharmacoecon Outcomes Res. 2011;11(6):729-37.
14. Garattini L, Ghislandi S. Off-patent drugs in Italy. A short-sighted view? Eur J Health Econ. 2006;7:79-83.
15. Klok RM, Boersma C, Oosterhuis I, Visser ST, De Jong-Van den Berg LTW, Postma MJ. Switch patterns before and after patent expiry of omeprazole: a case study in The Netherlands. Aliment Pharmacol Ther. 2006;23(11):1595-600.
16. Ubeda A, Cardo E, Sellés N, Broseta R, Trillo JL, Fernández-Llimós F. Antidepressant utilization in primary care in a Spanish region. Impact of generic and reference-based pricing policy (2000–2004). Soc Psychiatry Psychiatr Epidemiol. 2007;42(3):181-8.
17. World Health Organization Collaborating Centre for Drug Statistics and Methodology. Use of ATC/DDD. Oslo, Norway: WHOCC; 2012.
18. Kaló Z, Muszbek N, Bodrogi J, Bidló J. Does therapeutic reference pricing always result in cost-containment? The Hungarian evidence. Health Policy. 2007;80(3):402-12.
19. Galizzi MM, Ghislandi S, Miraldo M. Effects of reference pricing in pharmaceutical markets: a review. Pharmacoeconomics. 2011;29(1):17-33.
20. Garuolienè K, Godman B, Gulbinovic J, Wettermark B, Haycox A. European countries with small populations can obtain low prices for drugs: Lithuania as a case history. Expert Rev Pharmacoecon Outcomes Res. 2011;11(3):343-9.
21. Puig-Junoy J. Impact of European pharmaceutical price regulation on generic price competition: a review. Pharmacoeconomics. 2010;28(8):649-63.
22. Danzon P, Ketcham J. Reference pricing of pharmaceuticals for Medicare: evidence from Germany, the Netherlands and New Zealand. Cambridge, MA: National Bureau of Economic Research; 2004.
23. Puig-Junoy J. The impact of generic reference pricing interventions in the statin market. Health Policy. 2007;84(1):14-29.
24. Puig-Junoy J. Do higher-priced generic medicines enjoy a competitive advantage under reference pricing? Appl Health Econ Health Policy. 2012; in press.
25. Kanavos P, Costa-Font J, Seeley E. Competition in off-patent drug markets: Issues, regulation and evidence. Economic Policy. 2008;23(55):499-544.
26. Pavcnik N. Do pharmaceutical prices respond to potential patient out-of-pockets expenses? Rand J Econ. 2002;33(3):469-87.
27. Andersson K, Petzold M, Sonesson C, Lönnroth K, Carlsten A. Do policy changes in the pharmaceutical reimbursement schedule affect drug expenditure? Interrupted time series analysis of cost, volume and cost per volume trends in Sweden 1986–2002. Health Policy. 2006;79(2):231-43.
28. Stargardt T. The impact of reference pricing on switching behaviour and healthcare utilization: the case of statins in Germany. Eur J Health Econ. 2010;11:267-77.
29. Dylst P. The impact of reference-pricing systems in Europe. 24th Annual EuroMeeting, DIA, 2012 March 28, Copenhagen, Denmark.
|
Author for correspondence: Pieter Dylst, PharmD, Research Centre for Pharmaceutical Care and Pharmacoeconomics, Katholieke Universiteit Leuven, Onderwijs en Navorsing 2, PO Box 521, 49 Herestraat, BE-3000 Leuven, Belgium |
Disclosure of Conflict of Interest Statement is available upon request.
Permission granted to reproduce for personal and non-commercial use only. All other reproduction, copy or reprinting of all or part of any ‘Content’ found on this website is strictly prohibited without the prior consent of the publisher. Contact the publisher to obtain permission before redistributing.
Related article
Reference price systems: stakeholder dialogue and involvement
Source URL: https://gabi-journal.net/reference-pricing-systems-in-europe-characteristics-and-consequences.html
| Introduction: Switching from originator products to generic equivalents is a key method used by governments to reduce costs and keep their healthcare systems sustainable. The aim of this article is to review generic medicine pricing in Europe by analysing the factors that influence them. Methods: The literature review focused on selected studies that highlighted generic medicine pricing in ambulatory care in Europe. PubMed, the Centre for Reviews and Dissemination databases, Cochrane Database of Systematic Reviews, and EconLit were searched up to August 2011. Search terms included, ‘pharmaceuticals’, ‘generic medicines’, ‘Europe’, ‘pricing’, ‘discount’, and ‘rebate’. Bibliographies of included studies were checked for relevant studies and the status of generic medicines pricing in Europe was also documented via accessing the European Generic medicines Association market survey. Results: Ex-manufacturer prices for generic medicines were found to vary substantially between European countries, which suggests that pricing not only reflects production costs, but is also influenced by the domestic regulatory environment. The penetration of generic medicines is more successful in countries that permit free pricing of medicines than in those that have price regulation. Although tendering systems may reduce (generic) medicine prices in the short term, little is known about the overall long-term impact of such systems. Conclusion: No single approach towards developing generic medicine pricing policies and attaining cost savings was evident |
Submitted: 20 April 2011; Revised manuscript received: 7 September 2011; Accepted: 13 October 2011
Generic medicines are a key instrument for governments to sustain their healthcare systems and control pharmaceutical expenditures. This contribution of generic medicines to the sustainability of European healthcare systems has been recognised by the High Level Pharmaceutical Forums, ‘Generic medicines provide an opportunity to obtain similar treatments at lower costs for patients and payers, while liberating budgets for financing new innovative medicines’ [1]. WHO also emphasises the cost-saving potential of switching consumption from originator products to generic equivalents [2].
This underlines the importance of gaining insight into generic medicine pricing in Europe, where pricing is an area of national responsibility of the EU Member States. The aim of this brief article is to review generic medicine pricing in Europe. This will be achieved by providing evidence concerning the generic medicine price levels in Europe and the factors that influence price levels and by providing an insight into the international literature on generic medicine pricing policies and their impact. When addressing generic medicine pricing policies, this article focuses on free-pricing systems versus price-regulated systems, reference pricing, price competition and discounts, and tendering procedures.
This literature review did not focus on summarising and assessing all relevant studies, but rather drew on personally selected studies about generic medicine pricing in Europe. As such, this literature review was not systematic.
Studies were identified by searching PubMed, Centre for Reviews and Dissemination databases, Cochrane Database of Systematic Reviews, and EconLit, up to August 2011. Additionally, the bibliography of included studies was checked for other relevant studies. Search terms included pharmaceuticals, generic medicines, Europe, pricing, policies, regulation, market, competition, reference pricing claw-back, tendering, discount and rebate alone and in combination with each other.
The literature review was restricted to generic medicine pricing in ambulatory care in a European country. Studies of generic medicine pricing in the hospital setting were excluded. Studies could be published in Dutch, English, French, German or Italian. Studies published prior to January 2000 were not included as this is a rapidly changing field. Additionally, the market survey conducted by the European Generic medicines Association was accessed to document the status of generic medicine pricing in Europe in 2011.
Generic medicine pricing
Price levels
Professor Simoens conducted an international comparison of ex-manufacturer prices of generic medicines in ambulatory care in 2005 [3]. Data on generic medicine prices of 15 molecules/strengths were derived from IMS Health. The analysis was limited to medicines in immediate release, oral, and solid dosage forms. Prices were compared between nine European countries (Belgium, Denmark, France, Germany, The Netherlands, Norway, Spain, Sweden and UK) and with India. India was included because the European generic medicines industry has been facing competition from Indian firms which benefit from lower labour and production costs, and weaker patent protection laws.
Ex-manufacturer prices, i.e. the price at which a pharmaceutical company sells medicines, for generic medicines varied substantially between European countries. For instance, generic medicine prices in the Scandinavian countries were substantially lower than prices in countries such as France, Germany, The Netherlands or UK. Such European variation suggests that the prices of generic medicines not only reflect underlying production costs, but are also influenced by the domestic regulatory environment surrounding registration, pricing, reimbursement and distribution of generic (and originator) medicines. This illustrates the importance of domestic healthcare policies in the pricing strategies of manufacturers. For instance, the introduction of the ‘manufacturer’ and ‘wholesaler’ schemes in UK in 2005 (which purported to increase generic medicine pricing transparency) reduced generic medicine prices by 32% in the first year following policy implementation [4, 5].
This study also indicated that the lowest ex-manufacturer prices of generic medicines were observed in India. Imports from India are likely to put downward pressure on generic medicine price levels in Europe. Indeed, Indian generic medicine prices could give an indication of how much generic medicine prices in Europe could drop in the future.
Factors influencing price levels
Generic medicine pricing not only varies between European countries, but it can also differ between medicines within a single country. Adriaen et al. examined the evolution of the retail price of originator and generic medicines from July 2001 to December 2005 in Belgium [6]. Price levels in the Belgian off-patent market were influenced by a large number of issues such as successive reductions in reference prices and prescription status of medicines; by market incentives in the form of price competition between generic medicines, competition between originator and generic medicines or price agreements between originator and generic medicines; by medication class; and by the market power of the originator medicine.
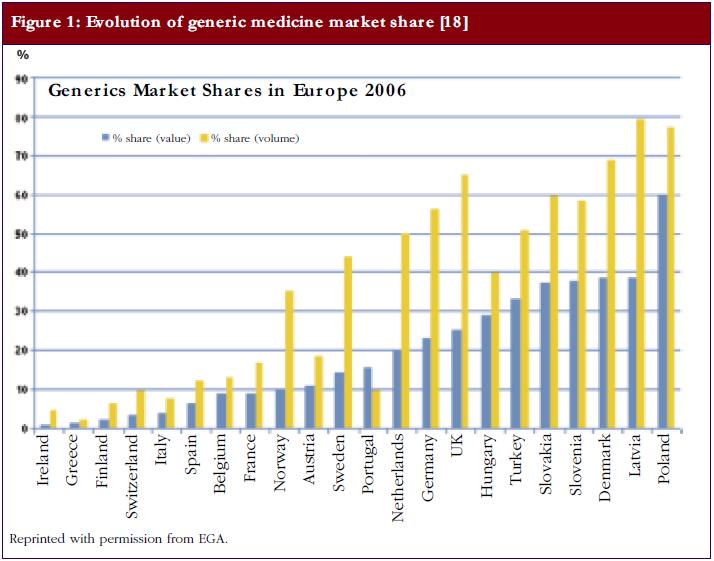
A recent study investigated whether there is a relationship between the market share of generic medicines and the price level in European off-patent markets [7]. Data on 35 oral active substances were derived from IMS Health. Medicine values were calculated on the basis of ex-manufacturer prices. The analysis included countries with a high market share of generic medicines (volume of 40% or more) as well as countries with a low market share of generic medicines (volume of 20% or less), see Figure 1. Data used were from June 2002 until March 2006.
The authors calculated the evolution of off-patent market values, i.e. price x volume, of 35 selected active substances. The decrease of market value in markets with a high share of generic medicines was more substantial (−26.6%) than the decrease in markets with a low share of generic medicines (−0.06%), although this was not statistically evaluated. In high generics market share markets, medicine prices dropped by −43.18% and in low market share markets by −21.56%. Thus, the extent to which price competition from generic medicines leads to price reductions appears to vary according to the market share of generic medicines.
Markets with a high market share of generic medicines tended to see a larger decrease in medicine prices than markets with a low market share of generic medicines. This observation suggests that the generic medicines industry is able to deliver competitive prices if it is ensured a high market share of the off-patent market. This high market share is dependent on demand-side policies. On the one hand, countries with a high market share of generic medicines have incentives in place for physicians, pharmacists, and/or patients to demand generic medicines. On the other hand, there are few incentives to stimulate demand for generic medicines in countries with a low market share of generic medicines [8]. This is corroborated by recent work from Godman et al., which suggests that European countries can enhance prescribing efficiency by implementing measures to lower generic medicine prices in combination with demand-side policies such as prescribing restrictions on originator medicines [9, 10].
Generic medicine pricing policies
Free market versus price regulation
European generic medicine pricing systems tend to be: a) a free-market approach, where manufacturers are (relatively) free to set generic medicine prices; b) a price-regulated system, where generic medicine prices are set on a regulatory basis, e.g. by law; or c) a combination of these two approaches, e.g. price regulation for the first generic medicine(s) and a free-market approach for following medicines [4, 11, 12].
In Europe, the reimbursement status of a generic medicine tends to determine which pricing system applies to a product. The literature indicates that penetration of generic medicines is more successful in countries that permit free pricing of medicines than in countries that have price regulation [13-15]. In countries where a free-market approach prevails, manufacturers of originator medicines can charge premium prices both before and after patent expiry, thereby attracting market entry of generic medicines. Manufacturers of generic medicines are engaged in price competition and are able to raise their market share by offering price reductions on their products. As a result, in countries with free pricing, the price difference between originator and generic medicines tends to be higher than in countries with price regulation [14, 16].
In contrast, in countries that rely on regulated prices, regulation drives down the price of the originator medicine over the life cycle of the medicine, thus discouraging market entry of generic medicines [15]. The limited diffusion of generic medicines in such markets restricts price competition following patent expiration, although competition in the form of discounts to pharmacists may occur [16]. In response to this, countries such as France have introduced generics substitution targets to sustain generic medicine market entry and price competition [17].
A market survey conducted by the European Generic medicines Association in 2011 revealed that 80% of European countries imposed price regulation and 20% adhered to free-market pricing [18]. Of those countries with pricing regulation, 37% set the price of generic medicines at a predetermined percentage below the price of the originator medicine, 31% set a maximum price for generic medicines, and 16% based the generic medicine price on the average price of medicines in a selection of European countries. The mechanism used to link the prices of generic and originator medicines may produce the following incentive: if a generic medicine needs to be priced at, for example, 55% or more below the price of an originator medicine throughout its life cycle, as is the case in France, then manufacturers of originator medicines can lower prices in an attempt to drive generic medicines out of the market.
The mechanism of price regulation may also have an impact on the extent to which price competition takes place and, hence, can influence generic medicine pricing. A Norwegian study has reported that a reference pricing system, i.e. a system where the regulator establishes a common reimbursement level for a group of interchangeable medicines, stimulated generics competition to a greater extent and led to lower prices than did price-cap regulation, i.e. a system where the regulator sets a maximum price that can be charged for a medicine [19]. The recent literature suggests that mechanisms to regulate prices such as price caps or a reference pricing system reduce prices of originator and generic medicines and may encourage price competition beyond the price reductions imposed by regulation in some countries, e.g. Austria, Lithuania [11, 12], but not in others, such as Slovenia or Spain [20].
Reference pricing
The generic medicine price level is likely to be influenced by the operation of a reference pricing system. Given that medicines priced above the reference price are likely to lose market share as a result of the additional patient co-payment, manufacturers have an incentive to price generic and originator medicines at or below the reference price. Also, in some countries such as Belgium, generic medicines need to be priced at or below the reference price if they wish to gain reimbursement [21].
The market survey conducted by the European Generic medicines Association found that 80% of European countries had a reference pricing system in 2011 [18]. When setting reference prices, the majority of countries took into account the prices of existing medicines: the reference price was based on either the lowest priced medicine (47% of countries), the lowest priced generic medicine (21% of countries), the average price of medicines (11% of countries), the average price of generic medicines (5% of countries), or other measures (16% of countries). Reference prices were established by active substance (42% of countries), therapeutic class (31% of countries), pharmacological class (18% of countries), or by another mechanism (9% of countries).
By calculating the reference price as a function of the prices of existing medicines, generic medicine manufacturers may have an incentive to compete, thereby driving down (reference) prices of medicines [22]. Nevertheless, it should be noted that competition does not exist in all countries that have adopted this mechanism of setting reference prices. Therefore, in addition to setting reference prices as a function of the prices of existing medicines, some countries have established a fixed minimum price difference between generic and originator medicines, e.g. France, Italy and Portugal [8]. This approach has the benefit of guaranteeing savings to the third-party payer, although it is not clear at what level maximum prices of generic medicines need to be set. The third-party payer will lose out if reimbursement prices of generic medicines are established at a higher level than would have been observed in a competitive market [22].
Discounting
In some European countries, competition between generic medicine manufacturers takes the form of discounting to the distribution chain rather than price competition. Indeed, two studies found that generic medicine discounts ranged from 20–70% off the wholesaler selling price in France and maximum discounts exceeded 50% of the Drug Tariff price in UK [23, 24]. Retail prices overestimate the value of generic medicines if generic medicines manufacturers compete by offering discounts to the distribution chain. This is because retail prices do not take these discounts into account. In the author’s opinion, the practice of discounting is not clear to market actors and is not fair as wholesalers and pharmacists are not rewarded for services rendered, but rather for their ability to negotiate discounts on artificial prices [22].
Such a system may financially benefit wholesalers and pharmacists, but it is not sustainable in the long run given that countries increasingly compare prices across Europe and given that healthcare payers and patients do not capture the potential savings from a generic medicines market where companies compete on price [25]. In response to this, France has regulated the size of discounts awarded in the distribution chain [24] and The Netherlands has introduced claw-back mechanisms that aim to recover the discounts that pharmacists receive. The introduction of the ‘Manufacturer’ and ‘Wholesaler’ schemes in UK made the manufacturing costs and discounts more transparent, leading to lower prices for generic medicines [5].
However, this type of government intervention is unlikely to be as efficient as a market mechanism where generic medicine manufacturers compete on the basis of prices rather than discounts to the distribution chain. Price competition between generic medicines manufacturers is transparent and easy for all market actors to understand, and ensures that prices paid by healthcare payers and patients reflect value [15]. For instance, Poland will introduce legislation in 2012 that outlaws discounting to the distribution chain and promotes price competition. However, such legislation also needs to address the issue of pharmacist remuneration as pharmacists are likely to lose an important source of income if discounts are outlawed.
Tendering
Tendering is a mechanism whereby a purchaser buys medicines based on a competitive bidding process where the contract is granted to the pharmaceutical supplier who offers the best bid following strict criteria [26]. Whereas tendering procedures are widely used in the hospital sector, in the last couple of years they are also being rolled out in ambulatory care in an increasing number of European countries with a view to constraining pharmaceutical expenditure [27]. Few data are available to date on the current status and impact of tendering procedures in Europe.
The literature suggests that significant pharmaceutical budget savings can be achieved from tenders, but the overall results are unclear [28]. One study pointed to pharmaceutical budget savings as a result of significant reductions in medicine prices in Denmark, Germany and The Netherlands [29]. However, although physicians and patients have incentives (in the form of lower patient co-payment) to demand medicines that are included in the tender in Germany, physicians still have the right to write ‘do not substitute’ on the prescription form. Furthermore, the Belgian experience indicates that tendering produced savings for one specific medicine, but these savings were offset by the fact that physicians switched their prescribing patterns to medicines with a similar therapeutic indication that did not fall under the tendering procedure, so-called ‘re-allocation of demand’, and total expenditure actually increased [30].
Countries that have implemented tendering procedures have sometimes witnessed a decrease in pharmaceutical investments, a slowdown in the development of the generic medicine market, short-term absences of some medicines due to logistic shortages, a reduction in pharmacist remuneration, and problems with patient compliance [29]. Finally, tendering systems in countries such as Germany are regionally based and are initiated between individual sickness funds and pharmaceutical companies and, therefore, are less transparent than in countries such as Belgium, which has a national tender.
Using evidence from the literature on generic medicine pricing in Europe, this article suggests that competition from Indian generic medicine manufacturers, international comparisons of generic medicine prices, some mechanisms to set (reference) prices, and efforts to move away from competition by discount to competition by price are likely to put pressure on generic medicine prices in European countries and to generate additional savings to healthcare payers and patients. These savings may depend on the implementation of a free-market approach because more generics competitors tend to enter the market and price competition is stronger in less-regulated markets. Although tendering systems may reduce (generic) medicine prices in the short term, little is known about the overall long-term impact of such systems.
Once the patent of an originator medicine expires, generic medicines can enter the market. Generic medicines have the same quality, safety and therapeutic efficacy as the originator medicine, but are less expensive than originator medicines. In an era of ageing populations and rising healthcare costs, generic medicines allow patients to access safe, effective, high quality medicines at 20–80% of the price of branded originator medicines.
No sources of funding were used to assist in the preparation of this manuscript. The author has no conflicts of interest that are directly relevant to the content of this manuscript.
References
1. European Commission [homepage on the Internet]. Pharmaceutical Forum: First Progress Report, 29 September 2006. 2007 [cited 2011 Dec 11]. Available from: http://ec.europa.eu/health/ph_overview/other_policies/pharmaceutical/pharma_forum_progres_report092006_en.pdf
2. Cameron A, Laing R. Cost savings of switching private sector consumption from originator brand medicines to generic equivalents. World Health Organization [homepage on the Internet]. 2010 [cited 2011 Dec 11]. Available from: http://www.who.int/healthsystems/topics/financing/healthreport/35MedicineCostSavings.pdf
3. Simoens S. International comparison of generic medicine prices. Curr Med Res Opin. 2007 Nov;23(11):2647-54.
4. Godman B, Shrank W, Wettermark B, Andersen M, Bishop I, Burkhardt T, et al. Use of generics – a critical cost containment measure for all healthcare professionals in Europe? Pharmaceuticals. 2010;3:2470-94.
5. McGinn D, Godman B, Lonsdale J, Way R, Wettermark B, Haycox A. Initiatives to enhance the quality and efficiency of statin and PPI prescribing in the UK: impact and implications. Expert Rev Pharmacoecon Outcomes Res. 2010 Feb;10(1):73-85.
6. Adriaen M, De Witte K, Simoens S. Pricing strategies of originator and generic medicines following patent expiry in Belgium. Journal of Generic Medicines. 2008;5:175-87.
7. Dylst P, Simoens S. Does the market share of generic medicines influence the price level? A European analysis. Pharmacoeconomics. 2011;29(10):875-82. doi:10.2165/11585970-000000000-00000
8. Simoens S, De Coster S. Sustaining generic medicines markets in Europe. Journal of Generic Medicines. 2006;3(4):257-68.
9. Godman B, Shrank W, Andersen M, Berg C, Bishop I, Burkhardt T, et al. Comparing policies to enhance prescribing efficiency in Europe through increasing generic utilization: changes seen and global implications. Expert Rev Pharmacoecon Outcomes Res. 2010 Dec;10(6):707-22.
10. Godman B, Sakshaug S, Berg C, Wettermark B, Haycox A. Combination of prescribing restrictions and policies to engineer low prices to reduce reimbursement costs. Expert Rev Pharmacoecon Outcomes Res. 2011 Feb;11(1):121-9.
11. Garuoliene K, Godman B, Gulbinovic J, Wettermark B, Haycox A. European countries with small populations can obtain low prices for drugs: Lithuania as a case history. Expert Rev Pharmacoecon Outcomes Res. 2011 Jun;11(3):343-9.
12. Godman B, Burkhardt T, Bucsics A, Wettermark B, Wieninger P. Impact of recent reforms in Austria on utilization and expenditure of PPIs and lipid-lowering drugs: implications for the future. Expert Rev Pharmacoecon Outcomes Res. 2009 Oct;9(5):475-84.
13. Martikainen J, Kivi I, Linnosmaa I. European prices of newly launched reimbursable pharmaceuticals–a pilot study. Health Policy. 2005 Nov;74(3):235-46.
14. Schulz A. Evolving dynamics of US and European generics markets. Journal of Generic Medicines. 2004;2(1):23-32.
15. Dylst P, Simoens S. Generic medicine pricing policies in Europe: current status and impact. Pharmaceuticals. 2010;3:471-81.
16. Magazzini L, Pammolli F, Riccaboni M. Dynamic competition in pharmaceuticals. Patent expiry, generic penetration, and industry structure. Eur J Health Econ. 2004 Jun;5(2):175-82.
17. Sermet C, Andrieu V, Godman B, Van GE, Haycox A, Reynier JP. Ongoing pharmaceutical reforms in France: implications for key stakeholder groups. Appl Health Econ Health Policy. 2010;8(1):7-24.
18. European Generic medicines Association. 2011 market review: the European generic medicines markets. Brussels: European Generic medicines Association; 2011.
19. Brekke K, Grasdal A, Holmas T. Regulation and pricing of pharmaceuticals: reference pricing or price cap regulation? European Economic Review. 2009; 53:170-85.
20. Puig-Junoy J. Impact of European pharmaceutical price regulation on generic price competition: a review. Pharmacoeconomics. 2010 Aug 1;28(8):649-63.
21. Fraeyman J, Van Hal G, De Loof H, Remmen R, De Meyer G, Beutels P. Potential impact of policy regulation and generic competition on sales of cholesterol lowering medication, antidepressants and acid blocking agents in Belgium. Acta Clinica Belgica. Forthcoming 2012.
22. Simoens S. Generic medicine pricing in Europe: current issues and future perspective. Journal of Medical Economics. 2008;11:171-5.
23. Kanavos P. Do generics offer significant savings to the UK National Health Service? Curr Med Res Opin. 2007 Jan;23(1):105-16.
24. Kanavos P, Taylor D. Pharmacy discounts on generic medicines in France: is there room for further efficiency savings? Curr Med Res Opin. 2007 Oct;23(10):2467-76.
25. Simoens S. Trends in generic prescribing and dispensing in Europe. Expert Rev Clin Pharmacol. 2008;1(4):497-503.
26. Leopold C, Habl C, Vogler S. Tendering of pharmaceuticals in EU Member States and EEA countries. Results from the country survey. Vienna: ÖBIG Forschungs-und Planungsgesellschaft mbH; 2008.
27. Kanavos P, Seeley L, Vandoros S. Tender systems for outpatient pharmaceuticals in the European Union: evidence from the Netherlands, Germany and Belgium. London School of Economics. European Commission [homepage on the Internet]. 2009 [cited 2011 Dec 11]. Available from: http://ec.europa.eu/enterprise/sectors/healthcare/files/docs/study_pricing_2007/tendering_systems_en.pdf
28. Dylst P, Vulto A, Simoens S. Tendering for outpatient prescription pharmaceuticals: what can be learned from current practices in Europe? Health Policy. 2011 Jul;101(2):146-52.
29. PricewaterhouseCoopers. Ontario public drug programs competitive agreement initiative. Ottawa: PricewaterhouseCoopers; 2007.
30. Arickx F, Bormans V, Leveque F, Mortier M, Soete E, Vanhaeren E. Public tendering for off patent medicines in Belgium – the simvastatin case. Brussels: RIZIV/INAMI; 2009.
|
Author: Professor Steven Simoens, MSc, PhD, Research Centre for Pharmaceutical Care and Pharmacoeconomics, Katholieke Universiteit Leuven, Onderwijs en Navorsing 2, PO Box 521, 49 Herestraat, BE-3000 Leuven, Belgium |
Disclosure of Conflict of Interest Statement is available upon request.
Permission granted to reproduce for personal and non-commercial use only. All other reproduction, copy or reprinting of all or part of any ‘Content’ found on this website is strictly prohibited without the prior consent of the publisher. Contact the publisher to obtain permission before redistributing.
Related articles
Payers endorse generics to enhance prescribing efficiency: impact and future implications, a case history approach
The impact of pharmaceutical pricing and reimbursement policies on generics uptake: implementation of policy options on generics in 29 European countries─an overview
European payer initiatives to reduce prescribing costs through use of generics
Source URL: https://gabi-journal.net/a-review-of-generic-medicine-pricing-in-europe.html
Copyright ©2024 GaBI Journal unless otherwise noted.