Biosimilar antibody drug conjugates: considerations of higher order structure and aggregation
Published on 2023/09/14
Generics and Biosimilars Initiative Journal (GaBI Journal). 2023;12(3):108-12
|
Abstract: |
Submitted: 27 June 2023; Revised: 1 August 2023; Accepted: 18 September 2023; Published online first: 2 October 2023
ADCs-The promise of precisely targeted drug delivery
Antibody drug conjugates (ADCs) are antibody-based anticancer therapeutics consisting of monoclonal antibodies attached to a cytotoxic drug via a linker of some kind. These highly targeted delivery systems offer the promise of lower cytotoxic drug levels in the patient and, as a result, potentially reduced drug side-effects [1].
The first globally approved ADC was Pfizer and Wyeth’s Mylotarg (gemtuzumab ozogamicin) in 2000, however, it was not until 2011 that the second ADC approval came in the form of Seagen and Takeda’s Adcetris (brentuximab vedotin). These early ADCs came with ‘black box’ warnings making clear that although the benefits outweighed the risks, there remained issues with toxicity. Over the next decade companies explored improved conjugation chemistries to link their non-proteinaceous drug to the antibody vector of choice with 14 ADCs in total receiving US Food and Drug Administration (FDA) approval to date, with many reaching blockbuster statuses.
The question is though, are ADCs a class of drug that may be almost immune to biosimilar competition given their complexity? Undoubtedly the development challenges are far greater which may limit the number of biosimilar sponsors willing to take them on. In 2021, the Drugs Controller General of India approved Zydus Cadila’s ‘similar biological’ of Genentech/Roche’s Kadcyla (trastuzumab emtansine) marketed as Ujvira for treating both early and advanced human epidermal growth factor receptor 2 (HER2) positive breast cancer. To date, neither FDA nor the European Medicines Agency (EMA) have yet approved a biosimilar ADC under their respective regulatory pathways.
Structural characterization and demonstrating biosimilarity of ADCs
The requirements for structural characterization of novel ADCs and potential biosimilar versions are the same as for other biomolecules in that the precepts of ICH Q6B [2] should be followed to achieve a full and detailed overall picture of primary and higher order structure (HOS). However, it must be noted that the requirement for conjugation and the nature of the conjugation chemistry itself mean that investigations need to be performed to:
- determine the drug/antibody ratio (DAR)
- locate the sites of attachment of the drug
- look for reaction by-products attached in place of the drug, and
- determine if any free linker is present on the ADC.
Careful analysis of conjugation products must be carried out to ensure that the conjugation itself has proceeded as expected. This includes determining that the antibody has not been adversely affected through, for example, oxidation, deamidation, non-specific drug binding or any other side reactions that are chemically feasible under the conjugation conditions used.
Many of the structural analyses performed will involve an analytical assessment of the primary structure, i.e. amino acid sequence, but it is important to remember that the attachment of drug groups with their own characteristics of size, charge distribution and hydrophobicity/hydrophilicity at various points could also influence secondary and tertiary structure, as well as creating the potential for aggregation.
For these reasons, it is important that the HOS is also investigated to fully understand the impact of the conjugation process on the monoclonal antibody (mAb) and the resultant ADC. It must be pointed out that alteration of the HOS between the parent mAb and the ADC is not necessarily in itself problematic, after all the ADC is not being marketed as a biosimilar to the parent mAb. Rather, the data are generated to give a better understanding of the product following conjugation. Furthermore, assessment of ADC HOS across different batches of product provides part of the structural characterization evidence for batch-to-batch consistency (or otherwise).
Alteration of the HOS of an ADC when compared to its native mAb may lead to questions regarding potential immunogenicity or could be a cause of increased aggregation, if observed. Furthermore, from a functional point of view, HOS changes may impact mAb target binding and therefore affect the ability of the mAb to deliver the drug to the site of action.
Analysis of secondary and tertiary structure
Secondary and tertiary structural techniques are used routinely in protein analysis, the most frequently used being circular dichroism (CD), fourier transform-infra-red (FT-IR), fluorescence analysis and nuclear magnetic resonance (NMR; both 1D (1 H) and 2D (1 H- 13 C) [3, 4]. These techniques are all equally applicable to ADCs since the protein structural features are preserved in ADCs, as would be expected. Each of these techniques works on a different principle and therefore will probe HOS in slightly different ways, resulting in unique outputs from each technique.
Circular dichroism
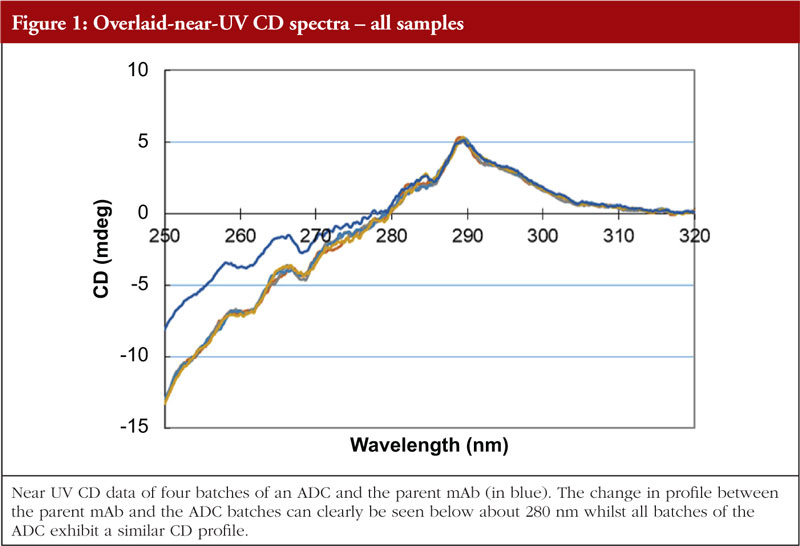
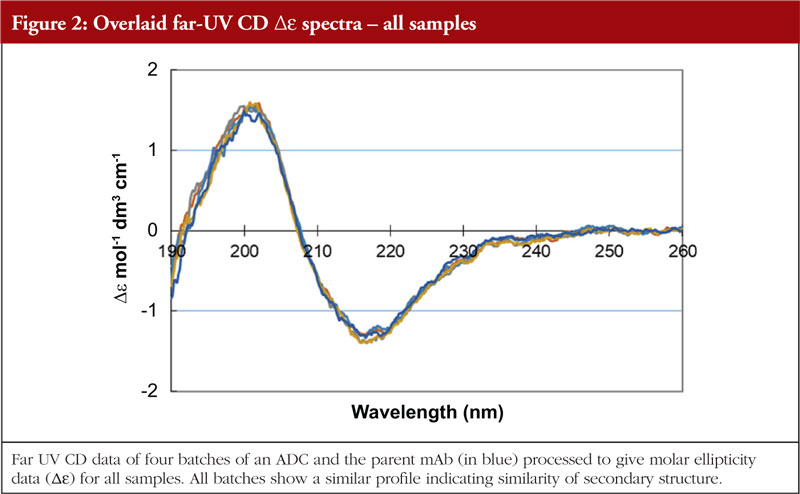
Circular dichroism measures the differences in absorbance by the sample of right and left hand circularly polarized light. This technique works well for chiral molecules and, since proteins exhibit chirality, has proven effective for their analysis. Wavelengths used are in the far and near UV region, giving information on secondary and tertiary structure, respectively. The far UV data are computer processed and, with database searching (data obtained from analysis of proteins with known secondary structures), provide a breakdown of secondary structural features. The protein component of ADCs is just as amenable to CD analysis as the parent monoclonal antibody; thus, CD data can be generated allowing an investigation into the similarity, or otherwise, of the HOS structural profiles. The data shown in Figures 1 and 2 show the stacked near and far UV data, respectively, for several ADC batches and the parent mAb [unpublished results, 5]. All data images presented in this article are from these same ADC batches and parent mAb, analysed by different HOS techniques. The difference in the spectra in the region below 280 nm in Figure 1 is consistent with the difference in absorbance between the disulphides and the pair of free/conjugated thiols as a result of the conjugation chemistry (pers. comm).
FT-IR
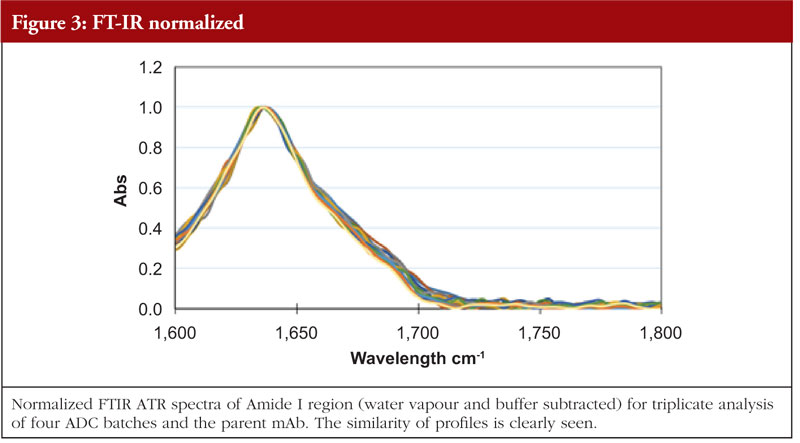
FT-IR uses infra-red light to investigate various bond flexures in the protein such as C=O, C-N, C-H, C-C and N-H. The chemical environments produced by the HOS features will have an impact on the precise wavelengths at which these flexures are seen. The absorption profile generated is subject to Fourier Transform mathematical interpretation and database interrogation to provide an output of relative abundances of secondary structural features. Figure 3 shows the FT-IR profiles of the same four ADCs and the parent mAb as shown in the previous figures [5].
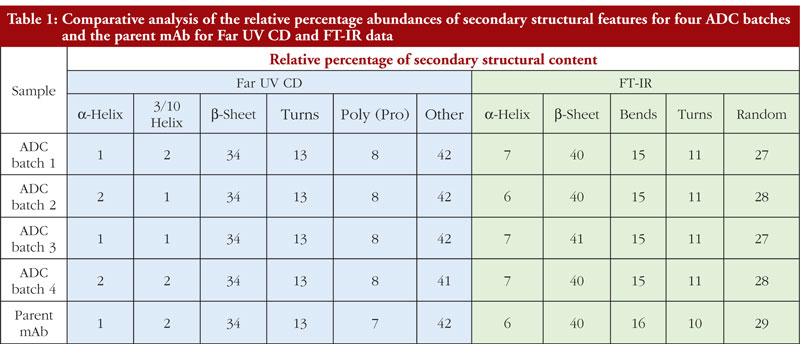
It is very important to bear in mind that secondary structure analysis by different techniques will provide data that will reflect the sensitivity of that technique for particular secondary structural features. In other words, Far UV CD and FT-IR data, when processed, are unlikely to provide the same relative ratios of secondary structural features. This can be seen in Table 1 where the Far UV CD and FT-IR data for the four ADCs and parent mAb analysed in this study are compared. The fact that different values are obtained between these techniques is not in itself a problem since what matters is that batches compare to one another within the same technique and data set. The data are not being used to give a precise structural definition for the ADCs but are aiming to demonstrate similarity of result and therefore similarity of structure between batches.
Fluorescence
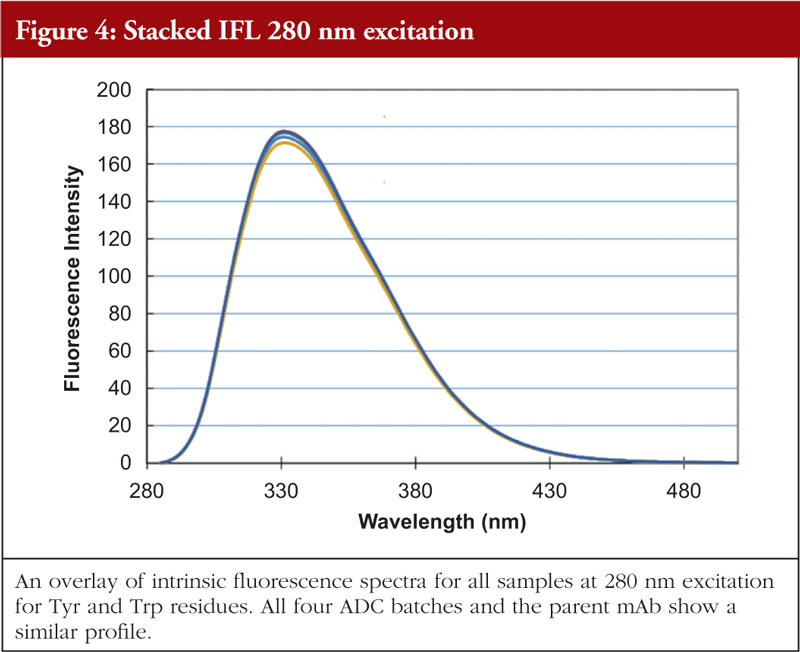
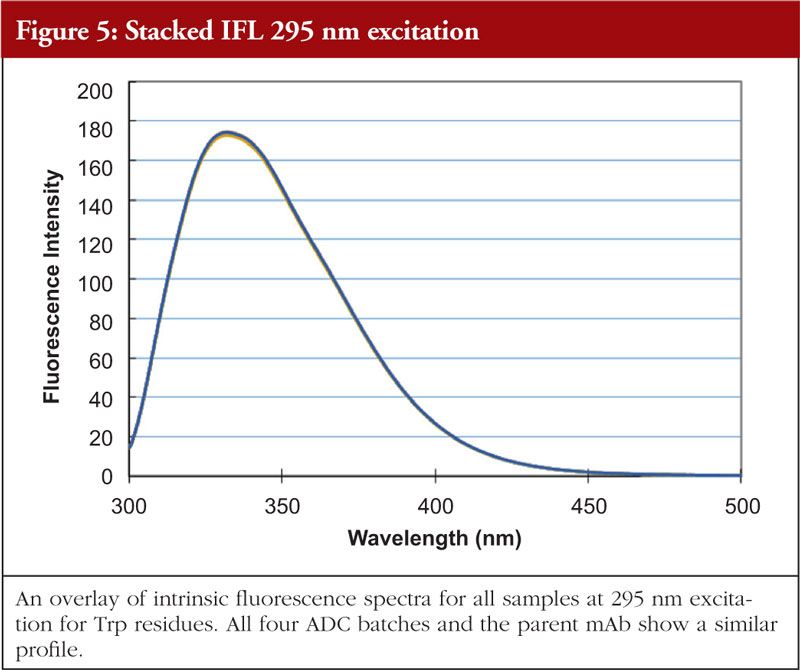
Fluorescence analysis can be both intrinsic to the molecule and extrinsic by means of fluorophore addition to the sample. In the intrinsic sense, fluorescent analysis involves measuring the fluorescence profile of tyrosine and tryptophan residues in the molecule using a spectrophotometer. The profile generated is related to the chemical environments that these amino acids find themselves in and thus the fluorescence profile gives information about the overall nature of the Tyr and Trp chemical environments. Relative amounts of secondary structural features are not determined with this technique. An example of intrinsic fluorescence is given in Figures 4 and 5 below for the same ADC batches and parent mAb as shown in the previous figures [5].
1D and 2D NMR
NMR relies on measuring the response of magnetic nuclei in the sample (most commonly 1 H and 13 C) to a radio frequency pulse applied perpendicular to a constant magnetic field. As the magnetic nuclei relax back to their ground state following the RF pulse they emit electromagnetic radiation, giving rise to the NMR signal. Again, the nature of the chemical environment that these atomic nuclei find themselves in will influence their relaxation profiles and this in turn influences the NMR profile.
A larger amount of structural information can be obtained by monitoring 2 magnetic nuclei in one experiment &ndash a so-called 2D NMR analysis. Most commonly 1 H and 13 C are measured but 1 H and 15 N measurements can also be used (although the relative abundance of 15 N is lower than 13 C). This gives rise to an intricate pattern of signals that are a function of the local environments the 1 H and 13 C nuclei find themselves in.
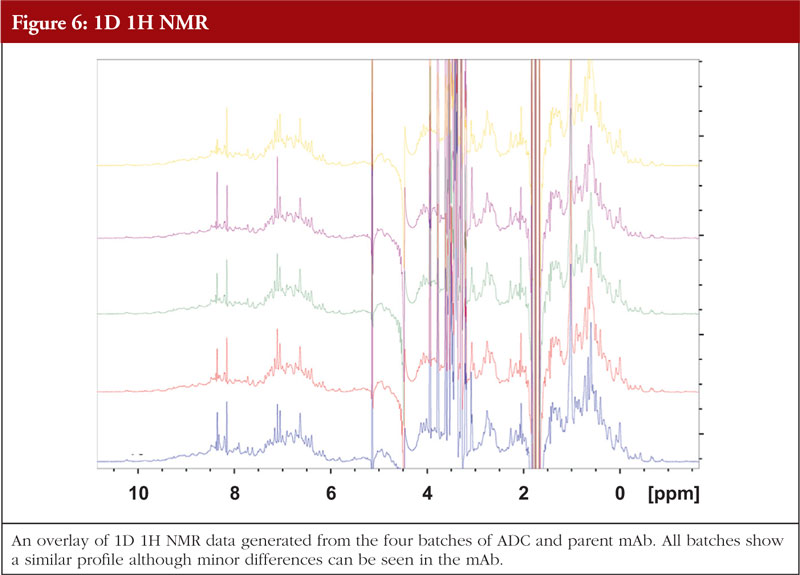
For ADCs and monoclonal antibodies, NMR particularly in the 2D sense, can be challenging due to the large size of the molecule, structural flexibility and long correlation time. Nonetheless, there are techniques available that allow data to be generated on such large proteins. Figure 6 gives an example of 1D NMR data generated for the same four batches of ADCs and the parent mAb as shown in previous figures (unpublished data).
Since these techniques are used to assess the HOS of a molecule, it is critical to analyse the unconjugated antibody alongside the ADC to demonstrate what to expect from the data from the mAb’s native state and to investigate if any changes have occurred in the HOS as a result of the conjugation process. One other point to consider is that each of these techniques described above will interact with the sample as the physics of the technique demands. Thus, since each technique works from different physical principles it follows that the outputs will be different between the techniques. This is perfectly fine from a structural characterization point of view and is a very good example of orthogonality, where data from different analytical approaches are used to support conclusions from the various techniques, forming a more self-sustaining whole.
The idea of orthogonality in HOS analysis is just as applicable for ADCs as it is for other proteins, since each individual technique will have its own sensitivities for different structural features such as CD having greater sensitivity for alpha helices compared to FT-IR. Any perturbation of the HOS in the ADC may disrupt these motifs to a greater or lesser degree both locally to the site(s) of conjugation or more globally across the molecule, therefore techniques need to be as all-encompassing as possible.
It is important to bear in mind that the ADC drug and linker will also have their own spectroscopic profile. Depending on the drug, it may have absorption characteristics in the spectral range of some of the instrumentation (CD in particular). A spectroscopic profile of the native drug is therefore absolutely necessary when assessing HOS data from an ADC to help assess any impact the drug is having on the overall ADC profile. Thus, comparison of the drug, ADC and native mAb can explain features seen in the ADC absorbance profile.
Aggregation
The chemical processes used to produce ADCs could result in enhanced aggregation as a result of structural disruption of the mAb and this needs to be investigated. Again, a sample of the native mAb should be analysed alongside the ADCs to provide baseline aggregate levels.
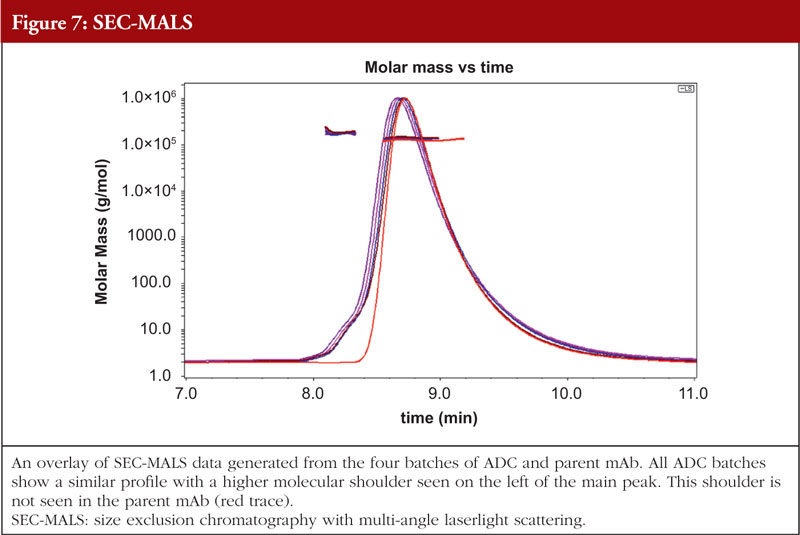
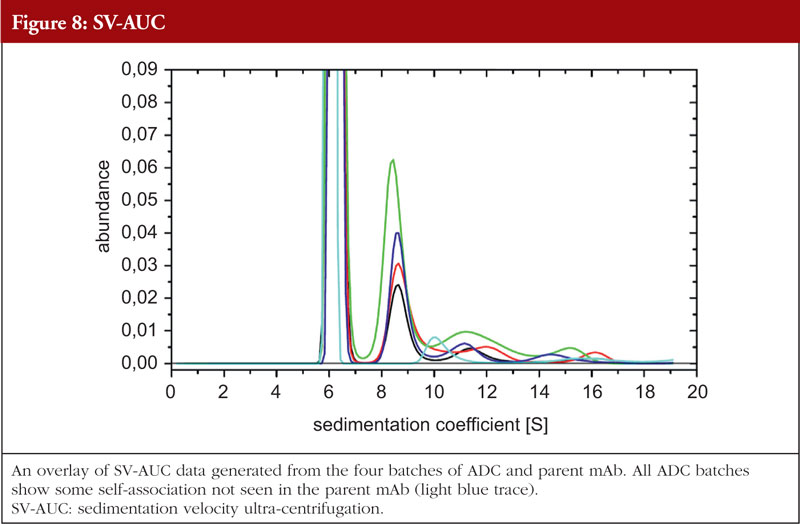
Just as with HOS analyses, orthogonality is very important in aggregation studies. This comes out of the general principle of the value of orthogonal investigations, as recognized by the regulatory agencies, stemming from the fact that different aggregation techniques will give different values for the level of aggregation due to the nature of the techniques themselves. A combination of sedimentation velocity ultra-centrifugation (SV-AUC) and size exclusion chromatography with multi-angle laserlight scattering (SEC-MALS) for aggregation studies have been shown to be applicable to ADCs. Data from the SEC-MALS and SV-AUC analyses of the same four ADC batches and the parent mAb as shown in previous Figures are shown in Figures 7 and 8, respectively (unpublished results). The parent mAb is likely to have slightly different hydrodynamic properties to the ADCs and this will result in slightly different sedimentation coefficients.
Discussion: HOS analysis – a necessity for ADC development
The range of HOS techniques available and applicable to structural analysis of biopharmaceutical products are equally applicable, and indeed vital, for the structural characterization of ADCs in order to gain a full structural understanding of the product. The fact that these techniques all have different biophysical attributes and thus analyse HOS from different perspectives is of great significance for building up a strong picture of biopharmaceutical HOS. When these techniques are used in an orthogonal manner, the various different features of individual techniques such as CD, FT-IR and NMR, with their unique biophysical properties, serve as meaningful molecular probes across different aspects of structure. Together with comparative data for the parent mAb, evaluations can be made not only with regard to batch-to-batch comparability for the ADC or in an ADC biosimilarity study but also for assessment of change in HOS compared to the parent mAb. This knowledge can be relevant for understanding any impact in terms of function or possibly aggregation that may be seen with the ADC. Furthermore, investigations of the extent of structural change between parent mAb and ADC can lead to the investigation of new conjugation chemistries that have less impact on HOS, thus producing mAb drug scaffolds more closely aligned structurally to the parent mAb.
Orthogonality of protein analytical aggregation techniques can also be successfully applied to ADCs with the data being used to either confirm satisfactory levels of aggregation or suggest possible modifications to process/purification procedures if levels are outside an expected or acceptable range. Thus, aggregation studies of ADCs are inextricably linked to HOS investigations and product development.
Conclusion
In summary, if ADCs are to become targets for biosimilar development, then, as with all biosimilar structural studies, their HOS and propensity to aggregate needs to be investigated using appropriate techniques such as those described here.
Funding sources
This paper is funded by BioPharmaSpec Ltd (www.biopharmaspec.com).
Competing interests: Dr Richard L Easton joined BioPharmaSpec in 2016 as Technical Director for Structural Analysis and is responsible for management of all aspects of carbohydrate and glycoprotein characterization at the primary structure level.
Provenance and peer review: Not commissioned; externally peer reviewed.
References
1. Drago JZ, Modi S, Chandarlapaty S. Unlocking the potential of antibody-drug conjugates for cancer therapy. Nat Rev Clin Oncol. 2021;18(6):327-44.
2. European Medicines Agency. ICH Q6B Specifications: test procedures and acceptance criteria for biotechnological/biological products – Scientific guideline [homepage on the Internet]. [cited 2023 Aug 1]. Available from: https://www.ema.europa.eu/en/ich-q6b-specifications-test-procedures-acceptance-criteria-biotechnological-biological-products
3. Astier A. Importance of the determination of the higher order structure in the in-use stability studies of biopharmaceuticals. Generics and Biosimilars Initiative Journal (GaBI Journal). 2020;9(2):49-51. doi: 10.5639/gabij.2020.0902.009
4. Vieillard V, Astier A, Paul M. Extended stability of a biosimilar of trastuzumab (CT-P6) after reconstitution in vials, dilution in polyolefin bags and storage at various temperatures. Generics and Biosimilars Initiative Journal (GaBI Journal). 2018;7(3):101-10. doi: 10.5639/gabij.2018.0703.022
5. McKee C, Chapman C, Bayley C. SDE-100 a stochastic cysteine linked vedotin ADC: assessing comparability of higher order structure using multiple orthogonal analytical approaches. Results presented as poster at 13th World ADC; 13&ndash16 March 2023; London.
|
Author: Richard L Easton, BSC (Hons), DIC, PhD, Technical Director – Structural Analysis, BioPharmaSpec Ltd, Suite 3.1 Lido Medical Centre, St Saviour, Jersey, JE2 7LA |
Disclosure of Conflict of Interest Statement is available upon request.
Copyright © 2023 Pro Pharma Communications International
Permission granted to reproduce for personal and non-commercial use only. All other reproduction, copy or reprinting of all or part of any ‘Content’ found on this website is strictly prohibited without the prior consent of the publisher. Contact the publisher to obtain permission before redistributing.