Continuous manufacturing versus batch manufacturing: benefits, opportunities and challenges for manufacturers and regulators
Published on 2021/01/05
Generics and Biosimilars Initiative Journal (GaBI Journal). 2021;10(1):44-56.
Author byline as per print journal:
Adjunct Associate Professor Sia Chong Hock, BSc (Pharm), MSc; Teh Kee Siang, BSc (Pharm)(Hon); Associate Professor Chan Lai Wah, BSc (Pharm)(Hon), PhD
|
Continuous manufacturing (CM) is the integration of a series of unit operations, processing materials continually to produce the final pharmaceutical product. In recent years, CM of pharmaceuticals has transformed from buzzword to reality, with at least eight currently approved drugs produced by CM. Propelled by various driving forces, manufacturers and regulators have recognized the benefits of CM and are awaiting the completion of the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) Q13, a harmonized guideline on CM that would be implemented by ICH members. |
Submitted: 30 November 2020; Revised: 20 December 2020; Accepted: 21 December 2020; Published online first: 6 January 2021
Introduction
Continuous manufacturing (CM) is the integration of a series of unit operations, processing materials continually to produce the final pharmaceutical product. This CM technology started in the eighteenth century during the first Industrial Revolution and has since been adopted by many industries [1]. However, it is only in recent years that CM of pharmaceuticals has transformed from buzzword to reality.
To date, there has been no standardized definition of CM, and the terms ‘continuous manufacturing’, ‘continuous production’ and ‘continuous processing’ are often intermingled [2]. Nonetheless, these terms are not interchangeable as they have different nuances. As the name suggests, ‘continuous production’ refers to a production schedule operating continually for 24 hours, seven days a week [2]. On the other hand, ‘continuous processing’ refers to a single unit operation where raw materials are continuously being loaded, processed, and unloaded without interruption [2].
There are many interpretations of CM and its related terminologies. However, end-to-end CM according to the US Food and Drug Administration (FDA), refers to an approach where the drug substance and drug product process steps are fully integrated into a single continuous system [3]. On the other hand, the hybrid approach is a combination of batch and continuous processing steps [3]. The pharmaceutical industry is increasingly adopting hybrid systems as it combines the advantages of batch and continuous processes [4–6].
Although significant progress is evident, the uptake of CM in the pharmaceutical industry remains sluggish due to various challenges [5–8]. Moreover, a lack of harmonized regulatory guidelines on CM has resulted in uncertain regulatory expectations by different regulatory authorities (RAs) [8]. To overcome the regulatory challenges and to reconcile CM-related concepts, the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) is developing a new quality guideline, ICH Q13: Continuous Manufacturing of Drug Substances and Drug Products [9].
At present, multiple studies have elaborated on significant technical and regulatory challenges for the CM of specific dosage forms, while others have conducted economic analyses on CM [10–14]. For example, Lee et al. [10] reviewed the benefits of CM, emphasized prime quality considerations and proposed solutions to address them. Additionally, a recent review by Vanhoorne and Vervaet [11] presented an overview of the technical aspects of CM and discussed regulatory guidelines on CM, specifically for oral solid dosages (OSDs). Despite many studies, to date, there have been few publications that review existing regulatory guidelines, highlight the latest challenges or make recommendations that are applicable for all pharmaceutical and biopharmaceutical products.
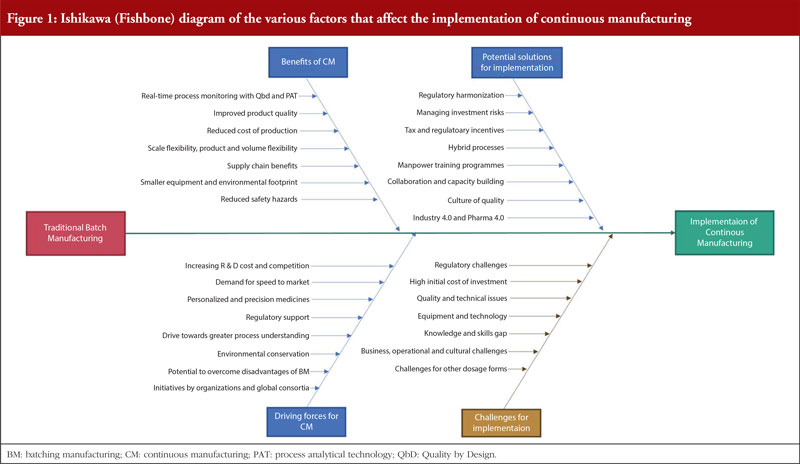
As portrayed in Figure 1, the implementation of CM is attributed to many factors. Therefore, this review aims to identify the benefits and opportunities of CM, assess the current extent of implementation, review existing regulatory guidelines and comprehensively highlight the existing challenges. The review also includes recommendations to facilitate the implementation of CM. The general concepts discussed in this review apply to all pharmaceutical dosage forms and biopharmaceutical products.
Overview of continuous manufacturing
CM is a combined process consisting of a sequence of more than one unit operation, developed to process materials continually to produce the final product [3]. As shown in Figure 2, CM is the integration of individual continuous unit operations with process analytical technology (PAT) which monitors and controls the critical process parameters (CPPs), critical material attributes (CMAs) and critical quality attributes (CQAs) [10]. Furthermore, CM streamlines manufacturing processes by eliminating work-up unit operations [10]. As such, CM equipment is typically smaller and is located within a single facility [10]. Batch manufacturing (BM), on the other hand, involves discrete unit processes with off-line quality testing and storage before each step [10]. Moreover, BM involves shipping of intermediates from one facility to another.
QbD and PAT
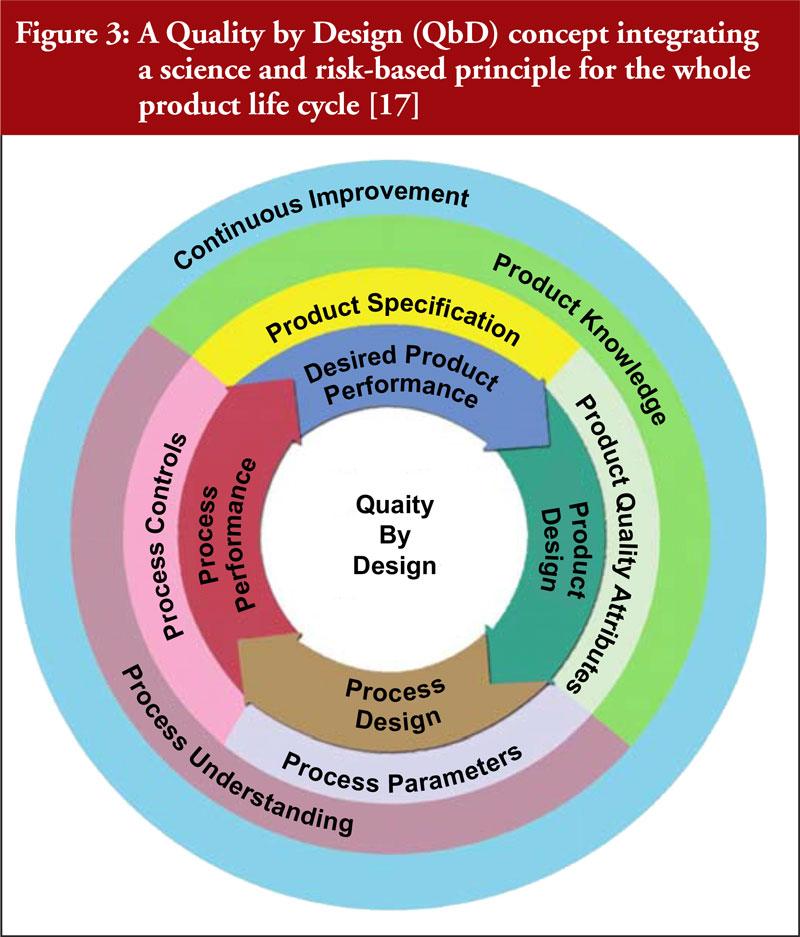
CM can improve pharmaceutical manufacturing with an enhanced development approach of Quality by Design (QbD) and the use of PAT [3, 10, 11]. As depicted in Figure 3, a comprehensive QbD approach allows for continuous improvement through product and process understanding to ensure better product quality [11, 15–17].
PAT is necessary for highly automated processes and continuous processing, as it fulfils quality requirements, such as residence time distribution (RTD) [18–21]. RTD refers to the distribution of time that materials remain in a unit operation; thus, it is critical for material characterization [21]. PAT uses multiple data sources for real-time product quality monitoring and control to achieve an integrated QbD quality system [10, 22]. CQAs such as the percentage of active pharmaceutical ingredient (API), particle-size distribution, granule size, and many others can be monitored with PAT [23]. As PAT and QbD tools can also be used in BM, manufacturers would be able to gain a better understanding of these processes, thereby facilitating a smoother transition to CM [22].
Benefits of CM
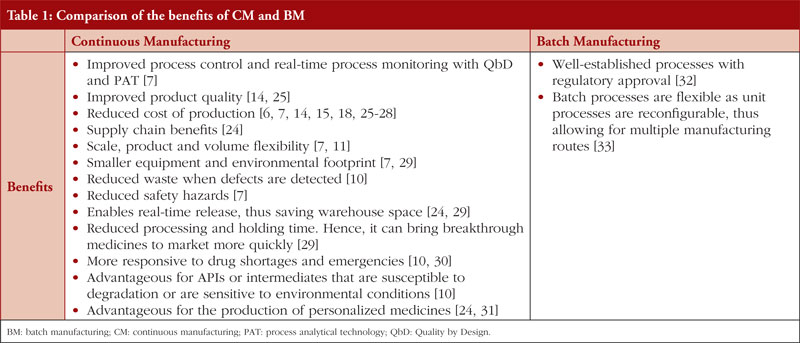
As listed in Table 1, there are various benefits of CM, which have been recognized for more than a decade [24].
In contrast to BM, CM provides greater scale flexibility in terms of its ability to scale-up production without any hindrance [10, 34, 35]. As depicted in Figure 2, CM eliminates the need for off-line testing and storage, thereby reducing the number of manufacturing steps [24, 29, 36]. Therefore, CM is more efficient than BM, as it reduces the processing time from raw materials to finished drug products, potentially by several months.
Also, CM has a greater response capacity to supply chain disruptions and drug shortages [10, 30]. This benefit of CM is accentuated in pandemics such as COVID-19, where ramped-up production of vaccines is required [29, 37]. As a result, there could be vast implications on global health and recovery of economies. On the other hand, it is difficult to adjust the production schedule of BM to meet changing demands [31], as building a new BM line in response to crises takes several months [10].
Benefits and opportunities for generics manufacturers
Realistically, generic manufacturers operate on low-profit margins and need to constantly take measures to keep drug prices low [38]. With increased processing speed and control from CM, it will lower the cost of production and grant tremendous cost advantages, especially with high-volume production [39]. Furthermore, as CMAs and CPPs are kept constant, there would be lower batch-to-batch variability [10]. Hence, CM allows generic manufacturers to match the narrow process variability of branded drugs [38]. Therefore, adopting CM is an opportunity for generics manufacturers to be prepared for new drug products from originator manufacturers [38]. At present, generics companies such as Dr Reddy’s Laboratories, Mylan Pharmaceuticals and Aurobindo are developing CM lines in India [4], while Lupin Pharmaceuticals is developing a continuous purification process for a biosimilar monoclonal antibody drug [40]. Therefore, CM is beneficial for both brand-name and generics manufacturers.
Driving forces of CM
The principal driving force for the implementation of CM is its potential to cut production costs, as manufacturers seek to maximize profits [14, 41]. Pharmaceutical companies are facing a threat to their earnings due to competition from generics and biosimilars manufacturers, increasing research and development costs, a forecasted low growth rate in developed economies, and greater demand for the affordability of drugs for patients [42]. As such, manufacturers are exploring the use of CM as it has proven to reduce operating expenses and capital expenditure [18].
Secondly, there is an increasing demand for speed to market for breakthrough therapies [14, 24, 31, 43]. CM can accelerate product development, delivering life-saving medicines to patients more quickly [31] without compromising product quality [11, 14, 24].
Thirdly, the pharmaceutical industry is transiting from large volume blockbuster drugs towards the production of personalized medicines [24]. This requires a shift from the current BM system towards CM, which is flexible in volume output and product variety [24, 31]. This feature is beneficial for the production of personalized medicines.
Lastly, environmental conservation will be a point of contention in the coming years, mounting greater external pressure on manufacturers. As CM supports greener processes and is proven to have reduced environmental footprint [11, 25, 28, 44, 45], the Pharmaceutical Roundtable recommends CM as the top priority in green engineering research [46]. These external trends, coupled with its compelling benefits, serve as fundamental driving forces for the adoption of CM.
Initiatives by international organizations to advance CM
Furthermore, there are various initiatives taken by international organizations to advance CM. In particular, the International Society for Pharmaceutical Engineering (ISPE) organized the 2020 ISPE Continuous Manufacturing Virtual Workshop, whilst the Massachusetts Institute of Technology (MIT) and the Continuous Manufacturing and Crystallisation Consortium (CMAC) organized the International Symposium on Continuous Manufacturing of Pharmaceuticals (ISCMP), which brought together various stakeholders and culminated in white papers on CM [47]. As listed in Table 2, there is an increasing number of global consortia collaborating to develop and share knowledge on advanced manufacturing technology [48]. Undeniably, these initiatives would accelerate the transition from BM to CM.
Extent of implementation of CM
From the first approval of drug products manufactured by CM in 2015, there have been a total of at least eight drug products approved. However, all of the currently approved drugs are OSDs. Hence, further development of CM for API, biopharmaceuticals and other pharmaceutical dosage forms is imperative.
The approved drugs in Table 3 utilize separately produced APIs [52]. At present, while there is a development of end-to-end CM that integrates continuous API production with drug product processes, there are none that comply with the current good manufacturing practice (cGMP) standards as of yet [25, 56]. This owes to the fact that there are more technical challenges in continuous processes for drug substances compared to drug products [25, 52, 57, 58]. Continuous API manufacturing is more complicated due to longer residence time [25, 52], higher quantity and diversity of unit operations [25, 57, 58], and intractably greater complexity of distinguished key molecules [57]. !However, many organizations are developing continuous flow processes for APIs [44, 52, 58-64]. For example, the Novartis-MIT collaboration has demonstrated that there are opportunities for integration of API and drug product processes with their end-to-end CM of aliskiren hemifumarate tablets [25]. Hence, cGMP-compliant end-to-end CM would most likely be realized in the future.
Similar to the CM of APIs, there is currently no fully continuous bioprocessing facility [13]. As today’s continuous downstream unit operations are still in their nascent stage of development, hybrid systems are expected to be implemented before end-to-end systems [30]. Nonetheless, the development of continuous bioprocessing is also gaining momentum [58]. Therefore, drugs in various pharmaceutical dosage forms would likely be approved in the coming years as development of CM progresses.
Existing regulatory guidelines on CM
Currently, there are three main regulatory guidelines on CM from ICH, FDA and ASTM International. As stated in Table 4, ICH Q13 and FDA guidance are still a work-in-progress. Other RAs and international organizations such as the World Health Organization (WHO), the Pharmaceutical Inspection Co-operation Scheme (PIC/S), and the Association of Southeast Asian Nations (ASEAN) do not have established guidelines on CM. As a result of this regulatory uncertainty, manufacturers would unlikely take the risk to implement CM as adopting new technology may lead to delays in regulatory approval, and consequently delays in delivering drug products to patients.
Nonetheless, the regulatory expectations for both BM and CM are essentially the same [3, 7]. Regardless of the mode of production, manufacturers are expected to have technically sound and risk-based processes to produce quality products [7]. For existing products manufactured by BM, filing for a post-approval change for the implementation of CM is a requirement by most RAs [3].
Notably, a key difference among the guidelines is that FDA draft guidance does not apply to biopharmaceuticals and APIs. Many stakeholders have expressed concern about these differences and expect FDA guidance to be aligned with ICH Q13 to ensure global harmonization [67]. Harmonization can be achieved through ICH Guidelines, since ICH is well-represented by regulatory and industry members [68].
While ICH guidelines are not mandated by law, ICH members are expected to implement ICH guidelines in Step 5 of ICH Procedure, as illustrated in Figure 4 [69]. Therefore, with the implementation of the harmonized ICH guidelines by RAs around the world, manufacturers will have greater clarity on the regulatory expectations of the various countries.
Challenges and opportunities in implementing CM
Although there is some progress, there is still considerable traction in the uptake of CM in the pharmaceutical industry. As listed in Table 5, there are various shortcomings of CM which may hinder manufacturers from adopting this technology.
Regulatory challenges
In multiple studies, the authors concur that the top barrier for implementation of CM is the regulatory challenges stemming from a lack of a globally harmonized regulatory guideline on CM [11, 23, 29, 70, 71]. Harmonization is crucial; otherwise, manufacturers must obtain approval from different RAs to market their products in various countries [72]. Historically, RAs tended to suppress post-approval changes to drug products, inadvertently ingraining a fixed mindset of batch processes in manufacturers’ production philosophy [76]. Furthermore, fear of regulatory delays has hindered the adoption of CM by manufacturers [72].
Nonetheless, regulatory support for CM has grown in recent years. FDA, EMA and PMDA have established specialized teams to promote the adoption of CM. Established in 2014, FDA Emerging Technology Programme (ETP) aims to help manufacturers overcome implementation challenges [77, 78]. Furthermore, regulatory guidelines on key prerequisites of CM has been published to aid in the implementation. Guidelines include the definition of a ‘batch’ [3], process validation [3, 79], continuous process verification [80, 81], and PAT [19]. Also, with the implementation of ICH Q12 guideline in 2019 [82], unnecessary post-approval applications are reduced to promote manufacturing innovations [83]. Therefore, while regulatory uncertainty is currently the top barrier, it is plausible that ICH Q13 will address this challenge once the guideline is implemented.
High initial cost of investment
The development of continuous processes is costly, owing to the use of PAT and automation software [38]. The high initial cost is another crucial barrier for manufacturers in adopting CM [41], due to the difficulty in justifying the case for new equipment as existing batch equipment is still functional with established regulatory approval [49, 73]. Hence, investing in CM is not a priority for most manufacturers [49]. In particular, this is a significant barrier for generics manufacturers operating on low-profit margins [4, 84]. Unpredictable demand for generic drugs would further deter a generics manufacturer from investing in CM [85].
Despite the high initial capital investment required for CM equipment, manufacturers can expect to reap economic benefits, especially with high-volume production. In multiple studies, CM has proven to reduce the cost of production [6, 18, 25–28]. An analysis conducted by the Novartis-MIT Center for Continuous Manufacturing showed a significant reduction in labour cost, in-process inventory and energy consumption, resulting in more substantial cost savings [18].
Nonetheless, economic analyses for the CM of biopharmaceuticals show conflicting results. Studies conducted by Pollock et al. [86] and Klutz et al. [43] on continuous antibody production affirm that the hybrid approach is more economically favourable than end-to-end CM [43, 86]. However, Hammerschmidt et al. [87] contend that fully continuous processes allow for the most significant cost savings. The differences in findings are possibly due to the complexity of a myriad of factors in biopharmaceutical production [88]. Nevertheless, most economic analyses maintain that BM is the least economically favourable approach compared to CM [13, 43, 86, 87, 89]. Research has shown that the cost savings could outweigh the high initial cost of implementation [90]. Therefore, CM is an opportunity for manufacturers to generate long-term profits [91].
Quality, safety and technical considerations
Material traceability is a key quality concern as characterization of raw materials and intermediate properties are more complex in CM. It is difficult to define the start and end of each batch of product in CM processes [10, 70, 72, 92, 93]. This is exceptionally challenging in low-volume and low-dose drug products due to the high amount of excipients used [70]. To overcome this obstacle, RTD monitored by PAT is a potential solution to ensure material traceability by determining the time taken for the material to pass through each unit operation [70, 93–95].
Additionally, advanced process control strategies are critical for CM to assure process performance and product quality [80, 96]. According to ICH Q8(R2) and Q10 [80, 96], control strategies include material and product attributes, operating conditions, product specifications, and process control. ICH Q8(R2), Q9 and Q10 quality trio provide guidance on developing control strategies that incorporate QbD [80] and risk management [97]. As part of the control strategy, real-time process management segregates any non-conforming material, thereby ensuring the high quality of the drug product [73]. Also, start-up and shutdown can be minimized to reduce material loss and cost incurred [92].
Although CM is generally safer than BM in that there are fewer transition steps [15], still it presents critical safety considerations. Manufacturers need to prevent overfilling of material, over-pressurization of the system, and other potential hazards not found in BM [72]. In recent years, some publications have addressed these technical issues on CM [10, 73]. Moreover, many equipment manufacturers are collaborating with the pharmaceutical industry to overcome the technical challenges of CM. Therefore, technical challenges are not necessarily substantial barriers for manufacturers. As opined by Janet Woodcock, Director of FDA CDER, making the business case is a greater barrier than technical issues for biopharmaceuticals [29].
Equipment and technological challenges
There is currently a shortage of available CM equipment [14, 72]. Smaller manufacturers lacking such capabilities have to outsource their production to a limited number of contract manufacturing organizations (CMOs) with CM equipment [11, 49]. In this regard, manufacturers should also consider the risk of material cross-contamination and data security when engaging CMOs [98].
Apart from the lack of CM equipment, research on CM technology was initiated mainly by academic institutions and equipment manufacturers, without active involvement from the pharmaceutical industry [11]. Consequently, this led to late-stage adjustments to the equipment by pharmaceutical manufacturers, further delaying the adoption of CM [11]. The slow implementation by pharmaceutical manufacturers has decelerated the rate of innovation by equipment vendors [72, 99].
In addition, as asserted by several studies [33, 73], batch unit operations involve discrete equipment that can be easily rearranged to enable multiple manufacturing routes. However, discrete batch unit operations come at the expense of lower plant and equipment productivity [33]. While CM is currently less flexible in this aspect, ‘plug-and-play’ continuous equipment comprising distinct reconfigurable unit processes are being developed [23, 100].
Therefore, this challenge will presumably become a stumbling block of the past, as interest in CM is escalating amongst pharmaceutical manufacturers, spurring greater involvement in the innovation process. Biopharmaceutical manufacturers are also collaborating with equipment vendors to innovate new CM technologies for biopharmaceuticals [99].
Knowledge and skills gap
Highly skilled personnel are required to develop and implement CM technology in cGMP facilities [41]. As CM is still in its infancy in the pharmaceutical industry, there is a lack of personnel with the relevant skills and knowledge [41, 49, 57, 70, 72]. This is a hurdle for both manufacturers and regulators as continuous systems require statistically trained personnel to understand the data generated [72]. Although training has been conducted, addressing the knowledge gap would require greater multidisciplinary collaboration [41] and commitment from all relevant stakeholders [72].
Inevitably, the adoption of CM will also cause some manual jobs to become obsolete due primarily to automation [74]. However, since there is a need for highly skilled personnel, CM presents an opportunity to create new jobs in R & D and testing [74]. Additionally, reduction in labour intensity will also produce less human errors [4]. Therefore, there would be an increase in new job openings requiring highly competent workers.
Business, operational and cultural challenges
Optimally, CM should be implemented in the early phases of drug development [92] to eliminate regulatory requirements needed to prove equivalence to current batch processes [77]. Correspondingly for biopharmaceuticals, it is more effective to implement CM at the clinical stages rather than modifying existing batch processes [77]. This is because biopharmaceuticals are inherently more complex and highly process-dependent [77]. However, the steep learning curve for the CM of a new drug product may encumber a tight launch timeline [77]. Therefore, aligning the manufacturing innovation with a clinical trial timeline is an uphill task and may require structural changes within the organization [72].
Furthermore, regulatory uncertainty has led to a conservative culture in the industry, delaying the adoption of new technologies [57]. Hence, mindset and cultural changes are needed within the pharmaceutical industry to shift its production philosophy of BM [72, 73]. Additionally, new technologies need to have significant proven benefits before they are implemented widely in the pharmaceutical industry [72, 92]. Hence, it is paramount for international organizations to publish success stories of CM to build confidence amongst manufacturers [57].
Challenges for pharmaceutical and biopharmaceutical products
As presented in Table 3, all of the approved drugs manufactured by CM are OSDs. Tablets represent the majority of pharmaceutical dosage forms [73, 92], and there are existing technologies for the CM of OSD. Also, FDA draft guidance is tailored towards small-molecule OSD [3]. These factors enable manufacturers to implement CM for OSDs with greater ease. Nonetheless, a one-size-fits-all approach would not be feasible for all dosage forms. Off-the-shelf equipment made for small-molecule drug production is not applicable for biopharmaceuticals [13, 101, 102]. In light of the complexity of biopharmaceutical manufacturing processes and products, CM of biopharmaceuticals is more technically challenging than OSDs. Furthermore, in the manufacture of biological vaccines, there is a need to develop continuous processes for viral inactivation, ultrafiltration and diafiltration [103]. Therefore, more research and investments into biopharmaceutical CM are required to actualize it [90].
Nevertheless, there is promising progress in the development of CM for newer dosage forms. At present, PAT tools are available for formulations such as suspensions [104, 105], liquids [104, 106] and emulsions [104, 107]. Also, as reported by Worsham et al. [12], there are economical and quality benefits for the CM of liposomal drug products. Therefore, CM for various pharmaceutical dosage forms and biopharmaceuticals is gaining momentum and is expected to become more prevalent in the future.
Recommendations and future perspectives
Despite the challenges faced by manufacturers and regulators, progress towards the transition to CM is evident. In this section, key recommendations are proposed to facilitate the implementation of CM.
Regulatory harmonization
As emphasized throughout this review, regulatory harmonization is imperative to address the current regulatory uncertainty in the industry. Without harmonization, implementation will remain sluggish as regulators endeavour to understand new CM technologies [58]. This issue would be addressed in the upcoming ICH Q13, expected to be completed in 2022. With the ICH Q13, there would be harmonised expectations for dossier approval and lifecycle management [108]. Consistent regulatory assessment and oversight would likely speed up the adoption of CM [49, 65]. The benefits of harmonized regulations include process improvements, development of new manufacturing methods to produce new molecules, and ultimately improvement in the access of medicines to patients [65]. Currently, many of the major regulators are working on ICH Q13 as members of ICH. While ICH guidelines are not mandatory, it is still worthwhile for regulators that are not part of ICH to use the published guidelines as a reference to create their specific guidelines on CM. Assuredly, with global regulatory harmonization, implementation of CM would substantially increase, as manufacturers will have greater clarity on the regulatory requirements.
Management of financial risks
Industry-wide pre-competitive initiatives such as efforts by global consortia could be organized to de-risk investments [24]. Also, CM technology that is applicable for multiple products will reduce the investment risk [33]. It is also essential to conduct a comprehensive analysis on the functions, costs and benefits of new technology [33]. Currently, most economic analyses are performed on finished drug products, but not on APIs [57]. Hence, there is still a need to develop a business case for the CM of APIs [109, 110].
Sustained financial investments from the government will also alleviate the high initial cost [72]. Historically, tax and regulatory incentives have led to industry-wide advancements [49]. Tax incentives, namely in Ireland, Singapore, and Puerto Rico allowed pharmaceutical manufacturing hubs to thrive [49]. As CM would ultimately benefit patients with improved access to medicines [65], governments should provide tax incentives for CM [49]. Today, government support for CM technology is steadily increasing [58, 111].
In addition, regulators should consider granting regulatory incentives to manufacturers for implementing CM, to expedite the approval process and grant a patent exclusivity period for drugs manufactured via CM [49]. With these incentives, manufacturers would be motivated to adopt CM as they would have the opportunity to break even faster and make greater profits.
Hybrid processes
Implementing the full end-to-end continuum of CM might be a quantum leap for manufacturers given the myriad of challenges. To overcome this hurdle, manufacturers can employ hybrid approaches to implement CM in a stepwise and progressive manner [33]. By combining the advantages of batch and continuous processes [112], manufacturers can leverage on hybrid processes to generate revenue to offset the high initial cost [39], increase output [36], and gain experience with continuous unit processes. Hybrid approaches are also economically favourable in the production of antibodies [43, 86]. Through this approach, manufacturers can assess the benefits of continuous processes and progressively transition towards end-to-end CM, thereby reaping all the benefits of CM for both small-molecule drugs and biopharmaceuticals [13, 41].
Capacity building, collaboration and publication
Training programmes must be conducted to address the lack of skilled workers in CM technology [67, 75, 113]. Currently, there are training programmes organized by various organizations, including ISPE, C-SOPS and the United States Pharmacopoeia (USP). The pharmaceutical industry can also learn from other industries that have implemented CM and adapt the processes for their products [57, 73, 75]. Implementation of CM also requires cross-departmental collaboration and institutional partnerships [24]. Manufacturers should engage in constant communication with regulators and industry experts [7, 84]. Global conferences, such as ISCMP [47] and national initiatives such as the ‘Pharma Innovation Programme Singapore’ [63] are cornerstones for fostering collaboration to drive innovation forward.
In addition, the publication of success stories and challenges of CM would build confidence in the industry and establish best practice standards [57]. Training programmes and publications are equally indispensable to ensure that the industry is well-equipped with the knowledge and skills needed to implement CM [49, 70].
Culture of quality
For decades, the pharmaceutical industry has been utilizing BM, steadily instilling a narrow mindset and conservative culture within manufacturing organizations [37, 57]. In a published interview, Jayjock [114] asserts that shifting organization mindsets is the most challenging issue of adopting CM. In recent years, however, manufacturers are growing in receptivity towards CM, and are adopting pharmaceutical quality systems (PQS) which are crucial for CM [96]. Nonetheless, PQS has its limitations as quality outcomes are contingent on people making quality choices [115]. Hence, manufacturers need to embrace a culture of quality to produce quality products via CM [116]. Organizations can also adopt Kaizen or Lean Six Sigma principles to develop cross-functional continuous improvement of quality [115, 117].
Industry 4.0 and Pharma 4.0
At present, there is an astounding amount of data collected in the pharmaceutical industry that is underutilized [117], with data integrity still being a pressing issue [20]. These issues would likely be exacerbated in CM due to an increase in data collection from systems such as PAT [117].
Nonetheless, the pharmaceutical industry is progressing towards Industry 4.0 to overcome the challenges highlighted above. Pharma 4.0, modelled after Industry 4.0, is driven by technological advancements, such as big data, Artificial Intelligence (AI) and cloud-computing [20]. These technologies process, store and convert data into useful knowledge [22]. For instance, cloud computing [21] and deep neural networks (DNN) [118] can support the implementation of PAT in CM systems. With the support of guidelines [119] and pharmacopoeias [120] on computerized systems, uptake of these technologies has been rapid. Organizations are also working with regulators to integrate AI into CM to enable real-time release [121].
With time, Industry 4.0 would cause a paradigm shift in pharmaceutical manufacturing. As technology advances, machine learning and predictive analytics may even enable preventative maintenance without shutting down continuous processes [122]. Though CM aligns with Pharma 4.0 objectives [90], manufacturers should conduct more research in integrating new technologies into CM, as data security processes must be robust to prevent cyber threats [122]. Also, manufacturers are advised to start with minimal cGMP impact processes [117].
Conclusion
The pharmaceutical industry and regulators have recognized the benefits and opportunities of CM. Propelled by various driving forces, several manufacturers have successfully adopted this technology. Since the CM of pharmaceuticals is an emerging field, there are still significant challenges that need to be overcome. However, with the implementation of ICH Q13, regulators would have specific guidelines on CM, thereby eliminating the top barrier to implementation. Thereafter, the implementation of CM would substantially increase with regulatory harmonization. Nonetheless, due to the complexity of pharmaceutical processes and products, there is no one-size-fits-all solution. Therefore, it must be emphasized that while regulators work towards creating a harmonized guideline on CM, manufacturers should work on overcoming existing cost, technical, and cultural challenges.
Perhaps these challenges could be seen as opportunities for growth. Other industries have paved the way forward towards an optimistic future. Now it is time for the pharmaceutical industry to rise to the challenge, seize the opportunities and revolutionise pharmaceutical manufacturing with CM. Ultimately, the world would benefit from greater response capacities to drug shortages and pandemics, while contributing to environmental conservation. Closer to heart, individual patients’ lives would be improved with greater access to life-saving medicines.
Competing interests:
None.
Provenance and peer review: Not commissioned; externally peer reviewed.
Authors
Adjunct Associate Professor Sia Chong Hock, BSc (Pharm), MSc
Teh Kee Siang, BSc (Pharm)(Hon)
Associate Professor Chan Lai Wah, BSc (Pharm)(Hon), PhD
Department of Pharmacy, National University of Singapore, 18 Science Drive 4, Singapore 117543
References
1. Wollenhaupt G. What pharma manufacturing can learn from M&Ms. Supply Chain Dive. 2019 Nov 12.
2. Jayjock E, LaMarche K. What is continuous manufacturing, anyway? Agreeing on a (proper) definition. Pharmaceutical Online. 2016 Aug 30.
3. U.S. Food and Drug Administration. Quality considerations for continuous manufacturing. Guidance for industry. 2019 [homepage on the Internet]. [cited 2020 Dec 20]. Available from: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/quality-considerations-continuous-manufacturing
4. Jhamb K. Continuous manufacturing – continuous manufacturing in pharmaceuticals: implications for the generics market. Drug Development & Delivery. 2019 Nov/Dec.
5. Hayek M. Continuous manufacturing: the clock is ticking. Is pharma ready? Accenture. 2017 Dec 4.
6. Inada Y. Continuous manufacturing development in pharmaceutical and fine chemicals industries. Mitsui & Co. Global strategic studies institute monthly report. 2019 Dec.
7. Allison G, Cain YT, Cooney C, Garcia T, Bizjak TG, Holte O, et al. Regulatory and quality considerations for continuous manufacturing. May 20-21, 2014 Continuous Manufacturing Symposium. J Pharm Sci. 2015;104(3):803-12.
8. International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use. ICH Q13: Continuous manufacturing of drug substances and drug products. Final Business Plan. 14 November 2018 [homepage on the Internet]. [cited 2020 Dec 20]. Available from: https://database.ich.org/sites/default/files/Q13%20Business%20Plan.pdf
9. International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use. ICH Q13: Continuous manufacturing of drug substances and drug products. EWG Work Plan. 2020 [homepage on the Internet]. [cited 2020 Dec 20]. Available from: https://database.ich.org/sites/default/files/Revised_Q13_EWG_WorkPlan_2020_0325.pdf
10. Lee SL, O’Connor TF, Yang X, Cruz CN. Modernizing pharmaceutical manufacturing: from batch to continuous production. J Pharm Innov. 2015;10(3):191-9.
11. Vanhoorne V, Vervaet C. Recent progress in continuous manufacturing of oral solid dosage forms. Int J Pharm. 2020;579:119194.
12. Worsham RD, Thomas V, Farid SS. Potential of continuous manufacturing for liposomal drug products. Biotechnol J. 2019;14(2):e1700740.
13. Fisher AC, Kamga M-H, Agarabi C, Brorson K, Lee SL, Yoon S. The current scientific and regulatory landscape in advancing integrated continuous biopharmaceutical manufacturing. Trends Biotechnol. 2019;37(3):253-67.
14. Karst DJ, Steinebach F, Morbidelli M. Continuous integrated manufacturing of therapeutic proteins. Curr Opin Biotechnol. 2018;53:76-84.
15. Toro JL. Continuous manufacturing and its regulatory challenge. Contract Pharma. 2019.
16. Zacché M, Andersson M. The advantages of a ‘Quality by Design’ approach in pharma drug development. Pharma Manufacturing. 2020.
17. Torres M. Challenges in implementing Quality by Design: an industry perspective. Bioprocess Int. 2015 Jun 16.
18. Schaber SD, Gerogiorgis DI, Ramachandran R. Economic analysis of integrated continuous and batch pharmaceutical manufacturing: a case study. Ind Eng Chem Res. 2011;50(17):10083-92.
19. U.S. Food and Drug Administration. PAT – A framework for innovative pharmaceutical development, manufacturing, and quality assurance. Guidance for industry. 2004 [homepage on the Internet]. [cited 2020 Dec 20]. Available from: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/pat-framework-innovative-pharmaceutical-development-manufacturing-and-quality-assurance
20. Binggeli L, Heesakkers H, Wölbeling C, Zimmer T. Pharma 4.0™: Hype or reality? Pharm Eng. 2018 Jul/Aug.
21. Dahlgren G, Macias KA, Moreira AR, Thompson DR, Herwig C, Dream R. Quality & regulatory solutions for PAT in continuous manufacturing. Pharm Eng. 2020 Sep/Oct.
22. Why you need PAT to move from batch to continuous processing. Manufacturing Chemist. 2019 Apr 9.
23. Fonteyne M, Vercruysse J, De Leersnyder F, Van Snick B, Vervaet C, Remon JP, et al. Process analytical technology for continuous manufacturing of solid-dosage forms. TrAC Trends in Analytical Chemistry. 2015;67:159-66.
24. Srai JS, Badman C, Krumme M, Futran M, Johnston C, et al. Future supply chains enabled by continuous processing-opportunities and challenges. May 20-21, 2014 Continuous Manufacturing Symposium. J Pharm Sci. 2015;104(3):840-9.
25. Burcham CL, Florence AJ, Johnson MD. Continuous manufacturing in pharmaceutical process development and manufacturing. Annu Rev Chem Biomol Eng. 2018;9(1):253-81.
26. Lawton S, Steele G, Shering P, Zhao L, Laird I, Ni X-W, et al. Continuous crystallization of pharmaceuticals using a continuous oscillatory baffled crystallizer. Org Process Res Dev. 2009;13(6):1357-63.
27. Ho C-H, Yi J, Wang X. Biocatalytic continuous manufacturing of diabetes drug: plantwide process modeling, optimization, and environmental and economic analysis. ACS Sustainable Chem Eng. 2019;7(1):1038-51.
28. Jolliffe HG, Gerogiorgis DI. Plantwide design and economic evaluation of two continuous pharmaceutical manufacturing (CPM) cases: ibuprofen and artemisinin. Comput Aided Chem Eng. 2015;37:2213-18.
29. Continuous manufacturing for the modernization of pharmaceutical production. Proceedings of a Workshop; 2018. The National Academies of Sciences Engineering and Medicine. Washington, DC: The National Academies Press.
30. Konstantinov KB, Cooney CL. White paper on continuous bioprocessing. May 20-21, 2014 Continuous Manufacturing Symposium. J Pharm Sci. 2015;104(3):813-20.
31. Littler B. Why aren’t more CDMOs using continuous manufacturing for API production? Outsource Pharma. 2019 Oct 8.
32. McLaughlin S. Continuous manufacturing vs batch manufacturing in the pharmaceutical industry. SL Controls. 2019.
33. Singh R, Boukouvala F, Jayjock E, Ierapetritou M, Muzzio F. Flexible multipurpose continuous processing of pharmaceutical tablet manufacturing process. GMP news, European Compliance Academic (ECE), 2012.
34. Mascia S, Heider PL, Zhange H, Lakerveld R, Benyahia B, Barton PI, et al. End-to-end continuous manufacturing of pharmaceuticals: integrated synthesis, purification, and final dosage formation. Angew Chem Int Ed Engl. 2013;52(47):12359-63.
35. Vervaet C, Remon JP. Continuous granulation in the pharmaceutical industry. Chem Eng Sci. 2005;60(14):3949-57.
36. Hernandez R. Continuous manufacturing: a changing processing paradigm. BioPharm. 2015 Mar 31.
37. Continuous pharmaceutical manufacturing – why the Industry should accelerate the adoption of this technology. CHEManager. 2020
38. Pazhayattil A, Ingram M, Kumar R, Rao S, Meier R. Is continuous manufacturing a good fit for generic drug products? Pharmaceutical Online. 2019 Jul 25.
39. Pazhayattil A, Spes J, Chaudhary RS. Continuous manufacturing: a generic industry perspective. PharmTech. 2017 May 29.
40. Lupin signs an agreement with CSIR-NCL and DST for the development of a continuous manufacturing process for biosimilar monoclonal antibody therapeutic (mAb). Biosimilar Development. 2018.
41. Poechlauer P, Manley J, Broxterman R, Gregertsen B, Ridemark M. Continuous processing in the manufacture of active pharmaceutical ingredients and finished dosage forms: an industry perspective. Org Process Res Dev. 2012;16(10):1586-90.
42. Otto R, Santagostino A, Schrader U. Rapid growth in biopharma: challenges and opportunities. McKinsey & Company. 2014 Dec 1.
43. Klutz S, Holtmann L, Lobedann M, Schembecker G. Cost evaluation of antibody production processes in different operation modes. Chem Eng Sci. 2016;141:63-74.
44. Rogers L, Jensen KF. Continuous manufacturing – the Green Chemistry promise? Green Chem. 2019;21(13):3481-98.
45. Poechlauer P, Colberg J, Fisher E, Jansen M, Johnson MD, Koenig SG, et al. Pharmaceutical roundtable study demonstrates the value of continuous manufacturing in the design of greener processes. Org Process Res Dev. 2013;17(12):1472-8.
46. Hu C, Testa CJ, Born SC, WU E, Shvedova K, Sayin R, et al. E-factor analysis of a pilot plant for end-to-end integrated continuous manufacturing (ICM) of pharmaceuticals. Green Chem. 2020;22(13):4350-6.
47. 4th International Symposium on Continuous Manufacturing of Pharmaceuticals. [cited 2020 Dec 20]. Available from: http://iscmp.mit.edu/
48. Sandler S. How global consortia are advancing continuous manufacturing. Pharm. Eng. 2018 Nov/Dec.
49. Badman C, Cooney CL, Florence A, Konstantinov K, Krumme M, Mascia S, et al. Why we need continuous pharmaceutical manufacturing and how to make it happen. J Pharm Sci. 2019;108(11):3521-3.
50. Krishnan H. ConsiGmaâ„¢ continuous manufacturing OSD road to operational excellence & accelerated drug development. Indian Pharmaceutical Alliance.
51. Pagliarulo N. Pharma’s slow embrace of continuous manufacturing. BioPharma Dive. 2018 Sep 24.
52. Balogh A, Domokos A, Farkas B, Farkas A, Rapi Z, et al. Continuous end-to-end production of solid drug dosage forms: coupling flow synthesis and formulation by electrospinning. Chem Eng J. 2018;350:290-9.
53. Sakurai K. The landscape of continuous manufacturing in Japan. 2018.
54. Yu LX, Kopcha M. The future of pharmaceutical quality and the path to get there. Int J Pharm. 2017;528(1-2):354-9.
55. Nasr MM, Krumme M, Matsuda Y, Trout BL, Badman C, et al. Regulatory perspectives on continuous pharmaceutical manufacturing: moving from theory to practice: September 26-27, 2016, International Symposium on the Continuous Manufacturing of Pharmaceuticals. J Pharm Sci. 2017;106(11):3199-3206.
56. Mullin R. Off the drawing board. Chem & Eng News. 2019;97(17).
57. Baxendale IR, Braatz RD, Hodnett BK, Jensen KF, Johnson MD, Sharratt P, et al. Achieving continuous manufacturing: technologies and approaches for synthesis, workup, and isolation of drug substance. May 20-21, 2014 Continuous Manufacturing Symposium. J Pharm Sci. 2015;104(3):781-91.
58. Hausner DB, Moore CMV. Continuous manufacturing current status. Pharm Eng. 2018 May/Jun.
59. Thomson N. Flexible API supply technologies: Pfizer’s strategy for API continuous development and manufacturing. Am Pharm Rev. 2020 May.
60. Wright R. Pfizer’s hybrid approach to implementing continuous manufacturing processes. Pharmaceutical Online. 2013 Aug 30.
61. GSK opens two mfg. facilities in Singapore. Contract Pharma. 2019.
62. O’Dwyer P. Eli Lilly invests €35m in Kinsale. Irish Examiner. 2016 Apr 6.
63. ASEAN pharma report: opportunities & threats 2020 and beyond. CPhI South East Asia Report 2020.
64. Shanley A. Continuous API manufacturing. PharmTech. 2017;41:52.
65. International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use. ICH Q13: Continuous manufacturing of drug substances and drug products. 2018 [homepage on the Internet]. [cited 2020 Dec 20]. Available from: https://www.ich.org/page/quality-guidelines
66. ASTM International. ASTM E2968-14. Standard guide for application of continuous processing in the pharmaceutical industry. 2014 [homepage on the Internet]. [cited 2020 Dec 20]. Available from: https://www.astm.org/Standards/E2968.htm
67. Arnum PV. Pharma industry weighs in on continuous manufacturing. Value Chain Insights. 2019 Jun 19.
68. International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use [homepage on the Internet]. [cited 2020 Dec 20]. Available from: https://www.ich.org/
69. International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use. Formal ICH procedure [homepage on the Internet]. [cited 2020 Dec 20]; Available from: https://www.ich.org/page/formal-ich-procedure
70. Moghtadernejad S, Escotet-Espinoza MS, Oka S, Singh R, Liu Z, Román-Ospino D, et al. A training on: continuous manufacturing (direct compaction) of solid dose pharmaceutical products. J Pharm Innov. 2018;13(2):155-87.
71. Lee K, Mantle J. Summary from advanced manufacturing technology workshop held at 6th Accelerating Biopharmaceutical Development Meeting. PDA J Pharm Sci Technol. 2020:pdajpst.2020.011429.
72. Promoting continuous manufacturing in the pharmaceutical sector. Center for Health Policy at Brookings. 2015 Oct 19.
73. Ierapetritou M, Muzzio F, Reklaitis G. Perspectives on the continuous manufacturing of powder-based pharmaceutical processes. AIChE J. 2016;62(6):1846-62.
74. Muzzio FJ. Want to cut healthcare costs? Try automation. The Hill. 2017.
75. McCarthy S. Converting to a “batch-less†world: quality implications of continuous manufacturing. Pharmaceutical Processing World. 2019.
76. Plumb K. Continuous processing in the pharmaceutical industry: changing the mind set. Chem Eng Res Des. 2005;83(6):730-8.
77. Martin J. Is continuous manufacturing right for your drug product? Pharmaceutical Online. 2019 May 30.
78. U.S. Food and Drug Administration. Emerging Technology Program. 2019 [homepage on the Internet]. [cited 2020 Dec 20]. Available from: https://www.fda.gov/about-fda/center-drug-evaluation-and-research-cder/emerging-technology-program.
79. U.S. Food and Drug Administration. Guidance for industry. Process validation: general principles and practices. 2011 [homepage on the Internet]. [cited 2020 Dec 20]. Available from: https://www.fda.gov/files/drugs/published/Process-Validation–General-Principles-and-Practices.pdf
80. International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use. ICH Q8(R2): pharmaceutical development. 2009 [homepage on the Internet]. [cited 2020 Dec 20]. Available from: https://database.ich.org/sites/default/files/Q8_R2_Guideline.pdf
81. ASTM International. ASTM E2537-16, Standard guide for application of continuous process verification to pharmaceutical and biopharmaceutical manufacturing. 2016 [homepage on the Internet]. [cited 2020 Dec 20]. Available from: https://www.astm.org/Standards/E2537.htm
82. International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use. Technical and regulatory considerations for pharmaceutical product lifecycle management. Q12. 2019 [homepage on the Internet]. [cited 2020 Dec 20]. Available from: https://database.ich.org/sites/default/files/Q12_Guideline_Step4_2019_1119.pdf
83. U.S. Food and Drug Administration. Statement from FDA Commissioner Scott Gottlieb, M.D. on new programs to promote the adoption of innovations in drug manufacturing that can improve quality and lower drug costs. 2019 [homepage on the Internet]. [cited 2020 Dec 20]. Available from: https://www.fda.gov/news-events/press-announcements/statement-fda-commissioner-scott-gottlieb-md-new-programs-promote-adoption-innovations-drug.
84. Hu YC. Pharmaceutical continuous manufacturing and recent regulatory status (Review) – Chinese. Chinese Journal of New Drugs. 2020;29(13):1464-8.
85. Richard M, Sciacca R. Continuous manufacturing in pharmaceuticals: Economic and policy issues. Bates White Economic Consulting. 2018 Oct.
86. Pollock J, Coffman J, Ho Sa V, Farid SS. Integrated continuous bioprocessing: economic, operational and environmental feasibility for clinical and commercial antibody manufacture. Biotechnol Prog. 2017;33(4):854-66.
87. Hammerschmidt N, Tscheliessnig A, Sommer R, Helk B, Jungbauer A, et al. Economics of recombinant antibody production processes at various scales: industry-standard compared to continuous precipitation. Biotechnol J. 2014;9(6):766-75.
88. Yang O, Qadan M, Ierapetritou M. Economic analysis of batch and continuous biopharmaceutical antibody production: a review. J Pharm Innov. 2019;14:1-19.
89. Walther J, Godawat R, Hwang C, Abe Y, Sinclair A, Konstantinov K. The business impact of an integrated continuous biomanufacturing platform for recombinant protein production. J Biotechnol. 2015;213:3-12.
90. Dream R, Herwig C, Pelletier E. Continuous manufacturing in biotech processes – challenges for implementation. 2018 Nov/Dec.
91. Brennan Z. FDA calls on manufacturers to begin switch from batch to continuous production. 2015 Apr 15.
92. Byrn S, Futran M, Thomas H, Jayjock E, Maron N, Meyer RF, et al. Achieving continuous manufacturing for final dosage formation: challenges and how to meet them May 20-21, 2014 Continuous Manufacturing Symposium. J Pharm Sci. 2015;104(3):792-802.
93. Engisch W, Muzzio F. Using residence time distributions (RTDs) to address the traceability of raw materials in continuous pharmaceutical manufacturing. J Pharm Innov. 2016;11(1):64-81.
94. Gao Y, Vanarase A, Muzzio F, Ierapetritou M. Characterizing continuous powder mixing using residence time distribution. Chem Eng Sci. 2011;66(3):417-25.
95. Vanarase AU, Osorio JG, Muzzio FJ. Effects of powder flow properties and shear environment on the performance of continuous mixing of pharmaceutical powders. Powder Technology. 2013;246:63-72.
96. International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use. Guidance for industry. ICH Q10: Pharmaceutical Quality System. 2008 [homepage on the Internet]. [cited 2020 Dec 20]. Available from: https://database.ich.org/sites/default/files/Q10%20Guideline.pdf
97. International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use. ICH Q9: Quality Risk Management. 2005 [homepage on the Internet]. [cited 2020 Dec 20]. Available from: https://database.ich.org/sites/default/files/Q9_Guideline.pdf
98. Escobar G. Working with a contract manufacturer: key considerations. BioPharm Int. 2008;(4):40-5.
99. Jacoby R, Pernenkil L, Harutunian S, Heim M, Sabad A. Advanced biopharmaceutical manufacturing: an evolution underway. 2015. Deloitte.
100. Teżyk M, Milanowski B, Ernst A, Lulek J. Recent progress in continuous and semi-continuous processing of solid oral dosage forms: a review. Drug Dev Ind Pharm. 2016;42(8):1195-214.
101. Johnson B, Persson J, Nielsen JT. Use of continuous plate reactor technology – reshaping reactions in pharmaceuticals as well as fine and specialty chemicals. Genet Eng Biotechnol News. 2008;28(9):50-1.
102. Short PL. Microreactors hit the major leagues. Chem Eng News. 2008;86(42):37-8.
103. D’Amore T, Yang Y.-p. Advances and challenges in vaccine development and manufacture. BioProcess International. 2019 Sep.
104. Rathore AS, Bhambure R, Ghare V. Process analytical technology (PAT) for biopharmaceutical products. Anal Bioanal Chem. 2010;398(1):137-54.
105. Park S, Kim M, Noh J, Chung H, Woo Y, Lee J, et al. Reliable and fast quantitative analysis of active ingredient in pharmaceutical suspension using Raman spectroscopy. Anal Chim Acta. 2007;593:46-53.
106. St-Onge L, Kwong E, Sabsabi M, Vadas EB. Rapid analysis of liquid formulations containing sodium chloride using laser-induced breakdown spectroscopy. J Pharm Biomed Anal. 2004;36:277-84.
107. Metz H, Mäder K. Benchtop-NMR and MRI–a new analytical tool in drug delivery research. Int J Pharm. 2008;364:170-5.
108. International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use. Final concept paper. ICH Q13: Continuous manufacturing of drug substances and drug products. 2018 [homepage on the Internet]. [cited 2020 Dec 20]. Available from: https://database.ich.org/sites/default/files/Q13%20Concept%20Paper.pdf
109. Srai J, Settanni E, Aulakh P. Evaluating the business case for continuous manufacturing of pharmaceuticals: a supply network perspective. In: Nagy Z, El Hagrasy A, Litster J, editors. Continuous pharmaceutical processing. AAPS Advances in the pharmaceutical sciences series, Vol 42. Springer, Cham. 2020. p. 477-512.
110. McWilliams JC, Allian AD, Opalka SM, May SA, Journet M, Braden TM, et al. The evolving state of continuous processing in pharmaceutical API manufacturing: a survey of pharmaceutical companies and contract manufacturing organizations. Org Process Res Dev. 2018;22(9):1143-66.
111. FDA awards $4.9 million to Rutgers-Led Research Consortium to expand development of continuous manufacturing in the pharmaceutical industry. News Wise. 2015 Oct 15.
112. Leuenberger H. New trends in the production of pharmaceutical granules: batch versus continuous processing. Eur J Pharm Biopharm. 2001;52(3):289-96.
113. Elder D, Savla R, Tindal S. Continuous manufacturing: regulatory and quality assurance challenges. Eur Pharm Rev. 2017 Aug 22.
114. Shanley A. Building a continuous processing culture. Pharm Tech. 2017;41(3):s24-s27.
115. Fiola MJ. Achieving a culture of quality in pharmaceutical biostatistics: one approach to achieving quality deliverables. Ther Innov Regul Sci. 2013;47(3):301-5.
116. Congdon K. Quality culture tips from Pharma Pros. Pharmaceutical Online. 2015 May 26.
117. Manzano T, Langer G. Getting ready for Pharma 4.0â„¢. Pharm Eng. 2018 Sep/Oct.
118. Roggo Y, Jelsch M, Heger P, Ensslin S, Krumme M. Deep learning for continuous manufacturing of pharmaceutical solid dosage form. Eur J Pharm Biopharm. 2020;153:95-105.
119. Government UK. Medicines & Healthcare Products Regulatory Agency. MHRA ‘GXP’ data integrity guidance and definitions. 2018 [homepage on the Internet]. [cited 2020 Dec 20]. Available from: https://www.gov.uk/government/news/mhra-gxp-data-integrity-definitions-and-guidance-for-industry
120. Chemometric methods applied to analytical data. 2020, European Pharmacopoeia Supplement 8.7.
121. Quartic.ai and Bright Path Labs will use AI for API continuous manufacturing. Pharm Tech. 2020.
122. Brettler D. Evolving risks in continuous manufacturing. Pharma Manufacturing. 2019.
|
Author for correspondence: Adjunct Associate Professor Sia Chong Hock, BSc (Pharm), MSc, Senior Consultant (Audit and Licensing) and Director (Quality Assurance), Health Products Regulation Group, Health Sciences Authority Singapore, 11 Biopolis Way, #11-01 Helios, Singapore 138667 |
Disclosure of Conflict of Interest Statement is available upon request.
Copyright © 2021 Pro Pharma Communications International
Permission granted to reproduce for personal and non-commercial use only. All other reproduction, copy or reprinting of all or part of any ‘Content’ found on this website is strictly prohibited without the prior consent of the publisher. Contact the publisher to obtain permission before redistributing.