GnRH agonists and antagonists in prostate cancer
Published on 2014/06/16
Generics and Biosimilars Initiative Journal (GaBI Journal). 2014;3(3):133-42.
Author byline as per print journal: Robert Janknegt, PharmD, PhD, Niels Boone, PharmD, Frans Erdkamp, MD, PhD, Victor Zambon, MD
|
Abstract: |
Submitted: 10 April 2014; Revised: 29 July 2014; Accepted: 11 August 2014; Published online first: 25 August 2014
Introduction
Prostate cancer
Prostate carcinoma is, after lung carcinoma, the most frequent form of cancer in men [1]. About 8,000 new patients are diagnosed with prostate cancer in The Netherlands each year [2]. The diagnosis of localized prostate cancer has increased considerably, probably because of the measurement of prostate specific antigen (PSA), which is useful in the detection of early stage prostate cancer [1]. A detailed description of the treatment of all stages of prostate cancer falls outside the scope of this manuscript. The reader is referred to the Dutch national guideline for a full overview of the treatment of prostate cancer [1].
Androgens stimulate the growth of both normal and cancerous prostate cells. Androgen deprivation therapy (ADT) is the primary treatment for patients with advanced prostate cancer [2]. Gonadotropin-releasing hormone (GnRH), also known as luteinising hormone release hormone (LHRH) is secreted by the hypothalamus and stimulates the hypophysis to secrete LH, follicle stimulating hormone (FSH) and adrenocorticotropic hormone (ACTH). LH activates the testes to produce testosterone. Chronic administration of GnRH agonists (analogues) blocks the secretion of LH, FSH and ACTH by the hypophysis. This results in a reduction of circulating testosterone levels. GnRH agonists increase survival as effectively as bilateral orchiectomy or treatment with oestrogens [2].
Androgen deprivation therapy (ADT) is a palliative and not a curative treatment of advanced or metastatic prostate cancer. It can normalize serum levels of PSA and can produce objective tumour responses. This antitumour activity can improve quality of life in patients with metastatic prostate cancer by reducing bone pain as well as the rates of complications, such as pathologic fracture, spinal cord compression, and ureteral obstruction. The duration of response to ADT for patients with metastatic disease is highly variable, and most prostate cancer patients eventually experience disease progression despite treatment. Patients who have progressed while on ADT are said to have castration-resistant disease [2].
Applications of GnRH agonists and GnRH antagonists
Patients with high-risk or locally advanced prostate cancer should be treated with external beam radiotherapy plus hormone treatment for at least two years.
Neoadjuvant GnRH agonists are recommended for four to six months in patients receiving radical radiotherapy for high-risk disease and should be considered in patients with intermediate-risk disease. Adjuvant hormonal therapy for two to three years is recommended for men receiving neo-adjuvant hormonal therapy and radical radiotherapy who are at high risk of prostate cancer mortality [1]. The drugs are indicated in the treatment of advanced or metastatic prostate cancer. GnRH agonists are the drugs of choice in metastatic prostate cancer, although a recent guideline from the European Society of Medical Oncology (ESMO) stated that antagonists could be an alternative [1]. Combined androgen depletion (GnRH agonists + ochiectomy) does not offer advantages over chemical or surgical castration only [1].
Guidelines for the treatment of prostate cancer do not specify a medicine of choice within the drug classes. There are no published tools available that could aid therapy choice. In this article the SOJA method was applied to both GnRH agonists and antagonists in order to make a transparent and rational selection of the most suitable medicines.
Methods
The SOJA method is a model for rational drug selection for formulary purposes [3]. See [2] for a detailed description of the methodology. The outcome of this study should be seen as the basis for discussions within formulary committees and not as the absolute truth. The present score is specific for the European situation.
The selection criteria and the relative weights that are assigned by the authors are shown in Table 1. For drugs included in this analysis, see Table 2.
Results
Efficacy
Improved overall survival should be the aim or all cancer treatment but this requires very large scale and long-term studies to establish. Also, both relapse-free survival and disease-free survival are used alternative endpoints in the judgement of clinical efficacy. Relapse-free survival is probably a more relevant endpoint than disease-free survival, because death unrelated to prostate cancer or its complications is included in the latter endpoint.
Outcomes that have been used in trials to establish the role of hormonal therapy in men with advanced prostate cancer include overall survival, measurable tumour response, changes in serum PSA, skeletal-related events, and quality of life (QoL). Complicating the interpretation of results, many studies were conducted prior to the routine use of serum PSA testing in screening and monitoring of disease and therefore these studies do not reflect typical contemporary patient populations or current practice patterns [4].
The prolonged natural history of advanced prostate cancer, its occurrence in older men who often have substantial comorbidity, and the heterogeneity of disease between patients complicate the use of overall survival as an endpoint in assessing response to treatment. The standard classifications of complete response, partial response, stable disease, and progressive disease are inadequate to evaluate response in most men with metastatic prostate cancer. Measurable disease is present in a small fraction of patients. Bone metastases are the most common site of disease, and bone involvement is difficult to measure objectively. Bone scan interpretation is variable, and there is a long healing time when lesions do respond to treatment [4].
PSA levels as a measure of efficacy
The appropriate use of serum PSA as a response endpoint for hormone therapy has not been well studied. The rate of PSA decline following initial hormone therapy relative to the rate of rise prior to initiation of hormone therapy is highly predictive of the time to prostate cancer-specific death [4]. The median survival of those with low PSA levels (< 0.2 ng/mL) was much longer than those with PSA levels of above 4 ng/mL [4].
Inclusion and exclusion criteria
In most cases, we have only used double-blind randomized studies to judge clinical efficacy of drugs included in SOJA analyses. The SOJA model is an instrument that enables users of the programme to determine, on the basis of agreed criteria, an order of merit for the various medicines available in a specific category [3]. This was not done in this SOJA score, because very few double-blind studies have been performed. For this reason, open, randomized phase III studies were included in the analysis. Non-randomized studies and studies comparing GnRH agonists with the addition of a drug such as flutamide or placebo were not included in the analysis, as these studies investigated the effects of the drug added to the GnRH agonist. Studies including a minimum of 25 patients per treatment arm were included in the analysis. Studies with short acting GnRH agonist formulations or nasal formulations were excluded. Similarly, studies in which hormonal treatment was not distinguished from orchiectomy in the same treatment arm were excluded, as well as studies that did not specify the GnRH agonist by name [5].
Direct comparative studies
Few direct comparative studies between GnRH (ant)agonists were identified. One retrospective study was excluded [6] as well as two other studies with a very small number of patients [7, 8] and one non-comparative study [9].
Abarelix versus leuprorelin
One study compared abarelix to leuprorelin. As could be expected testosterone surge was not seen in the abarelix group and did occur in the leuprorelin group [10].
Degarelix versus goserelin
Two studies compared degarelix and goserelin. Testosterone levels decreased more rapidly in the degarelix arm than in the goserelin arm, at eight weeks the levels were similar [11]. The effects of prostate volume and PSA levels were similar at 12 weeks [12]. At the same time point, more patients reported a > 3 point decrease in the International Prostate Symptom Score on degarelix than on goserelin: 36% vs 27% [12].
Degarelix versus leuprorelin
One study compared leuprorelin with degarelix 240 mg (n = 201). The testosterone response rates were comparable at one year. PSA levels declined more quickly in the degarelix group. The final reductions at 364 days were similar in the treatment groups [13–15].
Goserelin versus leuprorelin
A double-blind study compared goserelin 3.6 mg every 28 days (n = 540) and leuprorlin (n = 273) in patients with stage D2 (metastatic) prostate cancer. Both drugs were given in combination with either bicalutamide or (50 mg once daily) or flutamide 250 mg tid. The median follow-up was 160 weeks. The effects on time to progression and survival were similar [16].
Leuprorelin versus triptorelin
Two studies compared leuprorelin and triptorelin in patients with advanced prostate cancer. The effects on testosterone were identical. LH and PSA levels fell to a similar extent in both medicines [17, 18].
Studies with individual drugs
Buserelin
In one study buserelin depot was compared to polyestradiol phosphate (PEP). A more favourable effect of buserelin on disease progression was observed after three years of treatment [19]. This study is difficult to interpret because the comparator is not approved in The Netherlands.
Goserelin
Localized prostate cancer
Many studies were performed with goserelin. The medicine was studied as add-on to radiotherapy [20–44], showing lower PSA failure [28, 32, 34], increased five years disease-free survival [21, 31, 33, 36, 40] and 10 years [32] and lower degrees of local progression [23, 26, 33], better progression-free survival [26] and lower disease specific mortality at 10 years [23, 28, 33]. There was no effect on overall survival in the majority of the studies. Only one study showed an effect on overall survival at five and 10 years [37, 38].
Advanced prostate cancer
Six studies compared the monthly 3.6 mg and the 10.8 mg dose given every three months of goserelin in patients with advanced prostate cancer. The effects on testosterone levels were similar in the studies [45–50].
Other studies compared goserelin (3.6 mg monthly) to various medicines, such as polyestradiol phosphate (PEP) [51], diethylstilbestrol [52–54], bicalutamide [55–57] and orchiectomy [58–61]. Another study compared intermittent and continuous ADT [62].
Goserelin resulted in a longer time to progression [51], objective response rate [52, 53, 58] or no differences in clinically relevant endpoints [54–57, 59–62].
Metastatic prostate cancer
Several studies compared ADT with goserelin with or without flutamide with surgical orchiecomy in patients with metastatic prostate cancer [63–69], resulting in similar effects on objective response rates, time to disease progression and overall survival.
The EORTC 30853 study compared orchiectomy [n = 161] with a combination of goserelin (3.6 mg monthly) plus flutamide (250 mg tid orally, n = 163) in patients with metastatic prostate cancer. Significantly more favourable effects on objective progression and death from cancer were seen in the ADT group [70–74]. The time from objective progression to death was however longer in the orchiectomy group [72]. At longer follow-up (7.2 years) the advantages of ADT were maintained [75].
Other studies compared combinations of goserelin, flutamide and finasteride [76], goserelin versus extramustine [77], goserelin versus cyproterone acetate [78]. No differences were found in the first two studies, a longer time to progression was found for goserelin compared to cyproterone acetate [78].
Leuprorelin
Localized prostate cancer
Several studies were performed with leuprorelin: three versus eight months of neoadjuvant therapy with leuprorelin [79], as add-on to surgery [80]. The medicine was also studied as add-on to radiotherapy [20, 81, 82].
PSA was reduced compared to surgery alone. Positive surgical margins and lymph node involvement were seen more often in the group with surgery alone [80]. A higher overall survival was seen compared to radiotherapy alone [81]. Another study showed no positive effects on quality of life [82].
Advanced prostate cancer
One study investigated the effects of leuprorelin or oral bicalutamide on bone mineral density (BMD). The results were more favourable for bicalutamide [83].
Studies comparing one and three months formulations showed no relevant differences concerning effects on testosterone levels and PSA [84–87]. This was also the case for a comparison of three and six months formulations in a mixed population [88].
Leuprorelin prior to radical prostatectomy was compared to no pretreatment by a US study group. This study showed no differences in clinical relapse-free or PSA relapse-free survival rates between the groups [89, 90].
Triptorelin
Localized prostate cancer
One study compared preoperative triptorelin with no hormonal treatment in patients with localized prostate cancer. Triptorelin did not show favourable effects on postoperative PSA or skeletal events [91].
Advanced prostate cancer
One study compared triptorelin (+flutamide) with PEP. The primary endpoint was overall survival. No differences in mortality were observed at shorter or longer follow-up [92–94]. The 28 days and 3 months formulations showed similar effects on testosterone, LH and PSA levels [95]. Use of triptorelin prior to prostatectomy resulted in a lower rate than the control group, but there was no effect on progression-free survival [96–97].
Metastatic prostate cancer
Triptorelin was as effective as orchiectomy regarding effects on metastases and pain scores [98].
Although the levels of evidence were quite variable, no clinically meaningful differences were identified in clinical efficacy among buserelin, goserelin, leuprorelin and triptorelin in localized, advanced or metastatic prostate cancer. The clinical efficacy of goserelin and leuprorelin are much better documented than the other drugs.
It is not yet clear whether or not intermediate therapy with GnRH agonists is as safe and effective as continuous therapy [5].
All medicines are awarded 80%.
Safety
The incidence of severe adverse reactions was low for all compounds. Very few direct comparative studies between GnRH agonists and antagonists were identified. Agonists in general may induce depression, which can be severe. The incidence of severe adverse events was low to moderately high in most studies. The duration and size of most studies was insufficient to make firm statements concerning relative safety in the long term. There are no indications for major differences between the drugs concerning safety, with the exception of abarelix, which shows anaphylactic reactions at a higher rate than is the case with the other drugs.
Abarelix is awarded 60%, whereas the other medicines are awarded 70%.
Tolerability
Gonadotropin-releasing hormone (GnRH) agonists were associated with frequent, but harmless side effects. The side effects in direct comparative studies that are most relevant are summarized in Table 3.
The most common side effects result from the mechanism of action of the drugs, leading to impotence, decreased sexual drive and hot flushes. When GnRH agonists are given as monotherapy, testosterone surge may occur in the early phases of treatment.
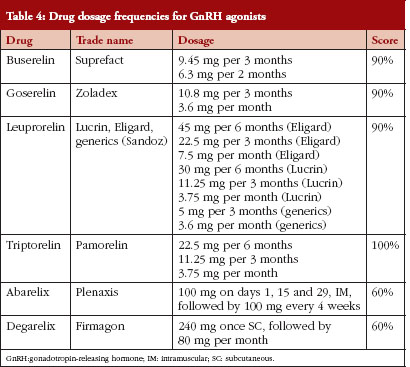
Dosage frequency
A low dosage frequency is convenient to the patient and may increase compliance with therapy. The highest score (100%) was awarded to the lowest dose frequency (every six months), the lowest score (20%) was awarded to the highest dose frequency (every week). Scores for different dosage frequecies are given in Table 4. Leuprorelin was awarded 90%, because the generic formulation was included in the analysis. Six months formulations of goserelin and leuprorelin are not available in The Netherlands, but these are approved in other European countries. The dosage frequency of the agonists is more favourable than the antagonists.
User-friendly dosage forms
A user-friendly dosage form which is easy to store and handle is convenient to the patient and the caregiver. User-friendly scores ranged from 30% for drugs stored at room temperature to 15% for drugs stored in a refrigerator, and 0% for drugs stored below 0°C. Drugs that required no reconstitution had a score of 30%, drugs that needed complicated reconstitution had a score of 10%. Ease of administration ranged from 40% for easy, to 10% for complex. Score for different drugs are shown in Table 5.
The hybrid generic implant formulation of leuprorelin was used for calculation of the score. Eligard is not ready for use and needs to be reconstituted and kept in the refrigirator.
All agonists are given subcutaneously in a depot formulation. No independent studies comparing the ease of use of the implants are available. The ease of administration is better for the antagonists, as no implant has to be injected.
Drug interactions
No specific studies were performed. There are almost no known interactions with any of the GnRH agonists. Buserelin and goserelin may lower glucose tolerance, which could lead to decreased efficacy of antidiabetic medication.
Special precautions
Data were collected from the summaries of product characteristics (SPCs) for each drug. The warnings and precautions of the GnRH agonists are summarized in Table 6.
More special precautions are applicable to abarelix and degarelix. These drugs are awarded 60%. Although there are differences in the SPCs of the GnRH agonists, it is unclear whether this reflects real differences between the drugs. These medicines are given a score of 70%.
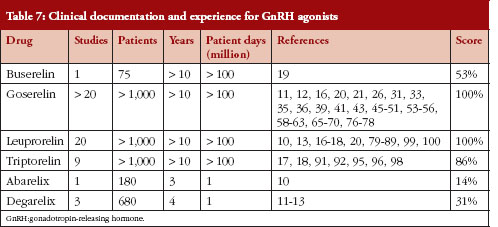
Documentation
The score for this criterion is divided into four sub-criteria: (1) number of randomized comparative studies; (2) number of patients in these studies; (3) number of years marketed; and (4) number of patient days worldwide.
The first two of these sub-criteria are indicative of the overall clinical documentation of the drugs in randomized controlled clinical studies. A large number of clinical studies and a large number of patients included in these studies leave no doubt about the clinical efficacy and safety of this drug in the studied population. The latter two criteria are indicative of the overall clinical experience with the drug. These sub-criteria may introduce a bias to the advantage of older drugs, but this is done intentionally. The safety of a newly introduced drug cannot be guaranteed from the results of clinical studies, in which only a relatively small number of patients were included and most patients at risk for the development of adverse reactions (e.g. patients with diminished renal function) were excluded. Both the number of patients that have been treated on a worldwide basis and the period that a certain drug has been available are of importance, as it may take time until adverse reactions occur. For a summary of these data, see Table 7.
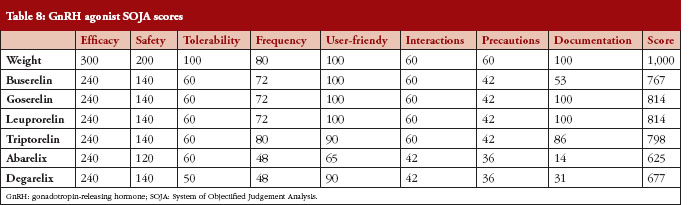
The overall SOJA score is presented in Table 8.
Discussion
There is currently no major need to make formulary choices within the GnRH agonists and antagonists in most countries. The drugs are usually not included in the hospital formulary because they are primarily used outside the hospital. In The Netherlands, many expensive drugs will be transferred to the hospital budget in January 2015. This will lead to discussions concerning formulary selection, because the cost of these drugs will be the responsibility of the hospital. Therefore, there is a need for tools to aid formulary choices. We have not included the criterion acquisition cost, to allow for a pre-selection only on quality aspects. Only the drugs with the highest scores will be considered as options for the treatment of patients with prostate cancer. After completion of the study, it turned out that the medicines in the present analysis would not be transferred to the hospital budget in 2015.
The weighting of the selection criteria reflects the opinion of the authors. Of course, such opinions are always open for debate. Therefore, all existing SOJA productions are available on the Internet (www.tablet.sojaonline.nl), allowing each user of the method to assign his/her own relative weight to each criterion, thereby calculating a personal score [101]. None of the SOJA productions is financially supported by pharmaceutical companies.
Goserelin and leuprorelin show the highest scores. The main advantage compared with buserelin and triptorelin is the better documentation for the treatment of prostate cancer. Because the differences in score between goserelin and leuprorelin (and possibly triptorelin) are limited, these drugs are acceptable as first-line therapy. Clearly the judgement of the authors concerning the properties of the medicines has an impact on the final outcomes. There are however few indications that there are clinically relevant differences between the agonists regarding clinical efficacy, safety and tolerability.
It should be noted that the studies with leuprorelin were performed with various formulations, whereas this was not the case for the other medicines. A specification of the applied formulation was only provided in a few studies: Lupron [17, 83], Enantone [18] and Sandoz generic formulation [87]. The 16 other studies did not specify the formulation, although the vast majority of studies used a dose of 7.5 mg per 28 days or 22.5 mg per three months [10, 13, 16, 20, 79, 81, 86, 99, 102]. The 3.75 mg or 11.25 strengths were used in two studies [80, 89].
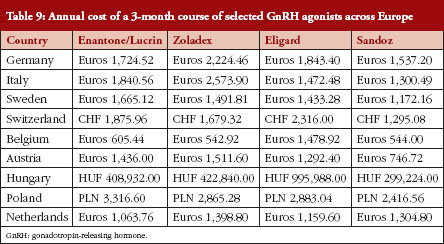
Acquisition cost plays a key role in the final selection of the drug of choice. The recent introduction of a generic leuprorelin implant formulation, which does not need reconstitution and can be stored outside of a refrigerator [87], and which is at least as effective as previously used leuprorelin formulations and was well tolerated in a relatively large group of patients (n = 818) [103] may be a good starting point for a renewed discussion on drug selection for the treatment of prostate cancer. Major cost savings might be applicable, because the acquisition cost of the various drugs has always been quite high. It seems likely that the need for a careful economic evaluation of drugs in oncology will increase throughout Europe.
Table 9 provides an overview of prices of the various agents in countries throughout Europe. The generic formulation is less expensive than the other medicines in most countries, with the interesting exception of The Netherlands. Prices are also quite different between countries, prices in Belgium are considerably lower than in other counties. Prices may be lower after negotiations between hospitals and companies.
The GnRH antagonists, degarelix and abarelix, show considerably lower scores than the GnRH agonists. Based on current data, these drugs should not be considered as first-line therapy for the treatment of prostate cancer. Their acquisition cost is also higher than those of (the already expensive) GnRH agonists. The 2013 guideline for the treatment of prostate carcinoma of the European Association of Urology assigned a limited place to GnRH antagonists: ‘Overall, this new family of agents seems appealing, but their advantages over GnRH agonists are far from proven. The use of GnRH antagonists is limited by a monthly formulation. Suppression of the initial flare-up with monotherapy is only clinically relevant in a few, symptomatic, metastatic patients’ [104].
Competing interests: Dr Rob Janknegt is the Deputy Editor-in-Chief and member of the Executive Editorial Board of the Generics and Biosimilars Initiative Journal (GaBI Journal). Niels Boone has nothing to declare. Dr Frans Erdkamp has participated in clinical trials funded by AstraZeneca. Dr Victor Zambon has nothing to declare.
Provenance and peer review: Not commissioned; externally peer reviewed.
Authors
Robert Janknegt, PharmaD, PhD, Hospital Pharmacist
Niels Boone, PharmD, Hospital Pharmacist
Frans Erdkamp, MD, PhD, Oncologist
Victor Zambon, MD, Urologist
Orbis Medisch Centrum, 1 Dr H van der Hoffplein, NL-6162 BG Sittard-Geleen, The Netherlands
References
1. Horwich A, Parker C, de Reijker T, et al. Prostate cancer: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2013;24 Suppl 6:vi106-14. doi:10.1093/annonc/mdt208 Epub 2013 Jun 27
2. Vereniging Integrale Kankercentra. Prostaatcarcinoom. Landelijke richtlijn 2007. Versie 1 [homepage on the Internet].[cited 2014 Sept 5]. Available from: www.oncoline.nl
3. Janknegt R, Steenhoek A. The System of Objectified Judgement Analysis (SOJA). A tool in rational drug selection for formulary purposes. Drugs. 1997;53(4):550-62.
4. Lee RJ, Smith MR. Initial hormone therapy of metastatic prostate cancer. Available from: www.UpToDate.com
5. Hussain M, Tangen CM, Berry DL, et al. Intermittend versus continuous androgen deprivation in prostate cancer. N Engl J Med. 2013;368(14):1314-24.
6. Fujii Y, Yonese J, Kawakami S, et al. Equivalent and sufficient effects of leuprolide acetate and goserelin acetate to suppress serum testosterone levels in patients with prostate cancer. BJU Int. 2008;101(9):1096-100.
7. Dias Silva É, Ferreira U, Matheus W, et al. Goserelin versus leuprolide in the chemical castration of patients with prostate cancer. Int Urol Nephrol. 2012;44(4):1039-44.
8. Anderson J, Al-Ali G, Wirth M, et al. Degarelix versus goserelin (+ antiandrogen flare protection) in the relief of lower urinary tract symptoms secondary to prostate cancer: results from a phase IIIb study (NCT00831233). Urol Int. 2013;90:321-8.
9. Trachtenberg J, Gittleman M, Steidle C, et al. A phase 3, multicenter, open label, randomized study of abarelix versus leuprolide plus daily antiandrogen in men with prostate cancer. J Urol. 2002;167(4):1670-4.
10. McLeod D, Zinner N, Tomera K, et al. A phase 3, multicenter, open-label, randomized study of abarelix versus leuprolide acetate in men with prostate cancer. Urology. 2001;58:756-61.
11. Axcrona K, Aaltomaa S, da Silva CM, et al. Androgen deprivation therapy for volume reduction, lower urinary tract symptom relief and quality of life improvement in patients with prostate cancer: degarelix vs goserelin plus bicalutamide. BJU Int. 2012;110(11):1721-8.
12. Mason M, Maldonado Pijoan X, Steidle C, et al. Neoadjuvant androgen deprivation therapy for prostate volume reduction, lower urinary tract symptom relief and quality of life improvement in men with intermediate- to high-risk prostate cancer: a randomised non-inferiority trial of degarelix versus goserelin plus bicalutamide. Clin Oncol (R Coll Radiol). 2013;25(3):190-6.
13. Klotz L, Boccon-Gibod L, Shore ND, et al. The efficacy and safety of degarelix: a 12-month, comparative, randomized, open-label, parallel-group phase III study in patients with prostate cancer. BJU Int. 2008;102(11):1531-8.
14. Tombal B, Miller K, Boccon-Gibod L, et al. Additional analysis of the secondary end point of biochemical recurrence rate in a phase 3 trial (CS21) comparing degarelix 80 mg versus leuprolide in prostate cancer patients segmented by baseline characteristics. Eur Urol. 2010;57(5):836-42.
15. Damber JE, Tammela TL, Iversen P, et al.The effect of baseline testosterone on the efficacy of degarelix and leuprolide: further insights from a 12-month, comparative, phase III study in prostate cancer patients. Urology. 2012;80(1):174-80.
16. Sarosdy MF, Schellhammer PF, Sharifi R, et al. Comparison of goserelin and leuprolide in combined androgen blockade therapy. Urology. 1998;52(1):82-8.
17. Heyns CF, Simonin MP, Grosgurin P, et al. Comparative efficacy of triptorelin pamoate and leuprolide acetate in men with advanced prostate cancer. BJU Int. 2003;92(3):226-31.
18. Kuhn JM, Abourachid H, Brucher P, et al. A randomized comparison of the clinical and hormonal effects of two GnRH agonists in patients with prostate cancer. Eur Urol. 1997;32(4):397-403.
19. Aro J, Ruutu M, Juusela H, et al. Polyestradiol phosphate (160 mg/month) or LHRH analog (buserelin depot) in the treatment of locally advanced or metastasized prostatic cancer. The Finnprostate Group. Ann Chir Gynaecol Suppl. 1993;206:5-8.
20. Jones CU, Hunt D, McGowan DG, et al. Radiotherapy and short-term androgen deprivation for localized prostate cancer. N Engl J Med. 2011;365(2):107-18.
21. Pilepich MV, Caplan R, Byhardt RW, et al. Phase III trial of androgen suppression using goserelin in unfavorable-prognosis carcinoma of the prostate treated with definitive radiotherapy: report of Radiation Therapy Oncology Group Protocol 85-31. J Clin Oncol. 1997;15(3):1013-21.
22. Lawton CA, Winter K, Murray K, et al. Updated results of the phase III Radiation Therapy Oncology Group (RTOG) trial 85-31 evaluating the potential benefit of androgen suppression following standard radiation therapy for unfavorable prognosis carcinoma of the prostate. Int J Radiat Oncol Biol Phys. 2001;49(4):937-46.
23. Pilepich MV, Winter K, Lawton CA, et al. Androgen suppression adjuvant to definitive radiotherapy in prostate carcinoma. Long-term results of phase III RTOG 85-31. Int J Radiat Oncol Biol Phys. 2005:61(5):1285-90.
24. Lawton CA, Winter K, Grignon D, et al. Androgen suppression plus radiation versus radiation alone for patients with stage D1/pathologic node-positive adenocarcinoma of the prostate: updated results based on national prospective randomized trial Radiation Therapy Oncology Group 85-31. J Clin Oncol. 2005;23(4):800-7.
25. Souhami L, Bae K, Pilepich M, et al. Impact of the duration of adjuvant hormonal therapy in patients with locally advanced prostate cancer treated with radiotherapy: a secondary analysis of RTOG 85-31. J Clin Oncol. 2009;27(13):2137-43.
26. Pilepich MV, Krall JM, al-Sarraf M, et al. Androgen deprivation with radiation therapy compared with radiation therapy alone for locally advanced prostatic carcinoma: a randomized comparative trial of the Radiation Therapy Oncology Group. Urology. 1995;45(4):616-23.
27. Pilepich MV, Winter K, John MJ, et al. Phase III radiation therapy oncology group (RTOG) trial 86-10 of androgen deprivation adjuvant to definitive radiotherapy in locally advanced carcinoma of the prostate. Int J Radiat Oncol Biol Phys. 2001;50(5):1243-52.
28. Roach M 3rd, Bae K, Speight J, et al. Short-term neoadjuvant androgen deprivation therapy and external-beam radiotherapy for locally advanced prostate cancer: long-term results of RTOG 8610. J Clin Oncol. 2008;26(4):585-91.
29. Shipley WU, Lu JD, Pilepich MV, et al. Effect of a short course of neoadjuvant hormonal therapy on the response to subsequent androgen suppression in prostate cancer patients with relapse after radiotherapy: a secondary analysis of the randomized protocol RTOG 86-10. Int J Radiat Oncol Biol Phys. 2002;54(4):1302-10.
30. Shipley WU, Desilvio M, Pilepich MV, et al. Early initiation of salvage hormone therapy influences survival in patients who failed initial radiation for locally advanced prostate cancer: a secondary analysis of RTOG protocol 86-10. Int J Radiat Oncol Biol Phys. 2006;64(4):1162-7.
31. Hanks GE, Pajak TF, Porter A, et al. Phase III trial of long-term adjuvant androgen deprivation after neoadjuvant hormonal cytoreduction and radiotherapy in locally advanced carcinoma of the prostate: the Radiation Therapy Oncology Group Protocol 92-02. J Clin Oncol. 2003;21(21):3972-8.
32. Horwitz EM, Bae K, Hanks GE, et al. Ten-year follow-up of radiation therapy oncology group protocol 92-02: a phase III trial of the duration of elective androgen deprivation in locally advanced prostate cancer. J Clin Oncol. 2008;26(15):2497-504.
33. Denham JW, Steigler A, Lamb DS, et al. Short-term androgen deprivation and radiotherapy for locally advanced prostate cancer: results from the Trans-Tasman Radiation Oncology Group 96.01 randomised controlled trial. Lancet Oncol. 2005;6(11):841-50.
34. Denham JW, Steigler A, Lamb DS, et al. Short-term neoadjuvant androgen deprivation and radiotherapy for locally advanced prostate cancer: 10-year data from the TROG 96.01 randomised trial. Lancet Oncol. 2011;12(5):451-9.
35. Laverdière J, Gomez JL, Cusan L, et al. Beneficial effect of combination hormonal therapy administered prior and following external beam radiation therapy in localized prostate cancer. Int J Radiat Oncol Biol Phys. 1997;37(2):247-52.
36. Bolla M, Gonzalez D, Warde P, et al. Improved survival in patients with locally advanced prostate cancer treated with radiotherapy and goserelin. N Engl J Med. 1997;337(5):295-300.
37. Bolla M, Colette L, Blank L, et al. Long-term results with immediate androgen suppression and external irradiation in patients with locally advanced prostate cancer (an EORTC study): a phase III randomised trial. Lancet. 2002;360(9327):103-6.
38. Bolla M, Van Tienhoven G, Warde P, et al. External irradiation with or without long-term androgen suppression for prostate cancer with high metastatic risk: 10-year results of an EORTC randomised study. Lancet Oncol. 2010;11(11):1066-73.
39. Crook J, Ludgate C, Malone S, et al. Report of a multicenter Canadian phase III randomized trial of 3 months vs. 8 months neoadjuvant androgen deprivation before standard-dose radiotherapy for clinically localized prostate cancer. Int J Radiat Oncol Biol Phys. 2004;60(1):15-23.
40. Crook J, Ludgate C, Malone S, et al. Final report of multicenter Canadian Phase III randomized trial of 3 versus 8 months of neoadjuvant androgen deprivation therapy before conventional-dose radiotherapy for clinically localized prostate cancer. Int J Radiat Oncol Biol Phys. 2009;73(2):327-33.
41. Witjes WP, Schulman CC, Debruyne FM. Preliminary results of a prospective randomized study comparing radical prostatectomy versus radical prostatectomy associated with neoadjuvant hormonal combination therapy in T2-3 N0 M0 prostatic carcinoma. The European Study Group on Neoadjuvant Treatment of Prostate Cancer. Urology. 1997;49(3 A Suppl):65-9.
42. Schulman CC, Debruyne FMJ, Forster G, et al. 4-year follow-up results of a European prospective randomized study on neoadjuvant hormonal therapy prior to radical prostatectomy in T2-3 N0M0 prostate cancer. European Study Group on Neoadjuvant Treatment of Prostate Cancer. Eur Urol. 2000;38:706-13.
43. Yee DS, Lowrance WT, Eastham JA, et al. Long-term follow-up of 3-month neoadjuvant hormone therapy before radical prostatectomy in a randomized trial. BJU Int. 2010;105(2):185-90.
44. Roach M 3rd, DeSilvio M, Lawton C, et al. Phase III trial comparing whole-pelvic versus prostate-only radiotherapy and neoadjuvant versus adjuvant combined androgen suppression: Radiation Therapy Oncology Group 9413. J Clin Oncol. 2003:21(10):1904-11.
45. Zinner NR, Bidair M, Centeno A, et al. Similar frequency of testosterone surge after repeat injections of goserelin (Zoladex) 3.6 mg and 10.8 mg: results of a randomized open-label trial. Urology. 2004;64(6):1177-81.
46. Fernandez del Moral P, Dijkman GA, Debruyne FM, et al. Three-month depot of goserelin acetate: clinical efficacy and endocrine profile. Dutch South East Cooperative Urological Group. Urology. 1996;48(6):894-900.
47. Wechsel HW, Zerbib M, Pagano F, et al. Randomized open labelled comparative study of the efficacy, safety and tolerability of leuprorelin acetate 1M and 3M depot in patients with advanced prostatic cancer. Eur Urol. 1996;30 Suppl 1:7-14; discussion 19-21.
48. Dijkman GA, Debruyne FM, Fernandez del Moral P, et al. A randomised trial comparing the safety and efficacy of the Zoladex 10.8-mg depot, administered every 12 weeks, to that of the Zoladex 3.6-mg depot, administered every 4 weeks, in patients with advanced prostate cancer. The Dutch South East Cooperative Urological Group. Eur Urol. 1995;27(1):43-6.
49. Debruyne FM, Dijkman GA, Lee DC, et al. A new long acting formulation of the luteinizing hormone-releasing hormone analogue goserelin: results of studies in prostate cancer. J Urol. 1996;155(4):1352-4.
50. Ishizuka O, Nishizawa O, Nishizawa S, et al. Comparison of efficacy and safety of 1- and 3-month luteinizing hormone-releasing hormone agonist depots as initial therapies for prostate cancer. Int J Clin Oncol. 2013;18(3):524-30.
51. Lukkarinen O, Kontturi M. Comparison of a long-acting LHRH agonist and polyoestradiol phosphate in the treatment of advanced prostatic carcinoma. An open prospective, randomized multicentre study. Scand J Urol Nephrol. 1994;28(2):171-8.
52. Emtage LA, George J, Boughton BJ, et al. Haemostatic changes during hormone manipulation in advanced prostate cancer: a comparison of DES 3 mg/day and goserelin 3.6 mg/month. Eur J Cancer. 1990;26(3):315-9.
53. Waymont B, Lynch TH, Dunn JA, et al. Phase III randomised study of zoladex versus stilboestrol in the treatment of advanced prostate cancer. Br J Urol. 1992;69(6):614-20.
54. Peeling WB. Phase III studies to compare goserelin (Zoladex) with orchiectomy and with diethylstilbestrol in treatment of prostatic carcinoma. Urology. 1989;33(5 Suppl):45-52.
55. Iversen P, Tyrrell CJ, Kaisary AV, et al. Casodex (bicalutamide) 150-mg monotherapy compared with castration in patients with previously untreated nonmetastatic prostate cancer: results from two multicenter randomized trials at a median follow-up of 4 years. Urology. 1998;51(3):389-96.
56. Boccardo F, Rubagotti A, Barichello M, et al. Bicalutamide monotherapy versus flutamide plus goserelin in prostate cancer patients: results of an Italian Prostate Cancer Project study. J Clin Oncol.1999;17(7):2027-38.
57. Boccardo F, Barichello M, Battaglia M, et al. Bicalutamide monotherapy versus flutamide plus goserelin in prostate cancer: updated results of a multicentric trial. Eur Urol. 2002;42(5):481-90.
58. Iversen P, Christensen MG, Friis E, et al. A phase III trial of zoladex and flutamide versus orchiectomy in the treatment of patients with advanced carcinoma of the prostate. Cancer.1990;66(5 Suppl):1058-66.
59. Turkes AO, Peeling WB, Griffiths K. Treatment of patients with advanced cancer of the prostate: phase III trial, zoladex against castration; a study of the British Prostate Group. J Steroid Biochem. 1987;27(1-3):543-9.
60. Soloway MS, Chodak G, Vogelzang NJ, et al. Zoladex versus orchiectomy in treatment of advanced prostate cancer: a randomized trial. Zoladex Prostate Study Group. Urology. 1991;37(1):46-51.
61. Vogelzang NJ, Chodak GW, Soloway MS, et al. Goserelin versus orchiectomy in the treatment of advanced prostate cancer: final results of a randomized trial. Zoladex Prostate Study Group. Urology. 1995;46(2):220-6.
62. de Leval J, Boca P, Yousef E, et al. Intermittent versus continuous total androgen blockade in the treatment of patients with advanced hormone-naive prostate cancer: results of a prospective randomized multicenter trial. Clin Prostate Cancer. 2002;1(3):163-71.
63. Iversen P. Zoladex plus flutamide vs. orchidectomy for advanced prostatic cancer. Danish Prostatic Cancer Group (DAPROCA). Eur Urol. 1990;18 Suppl 3:41-4.
64. Iversen P, Rasmussen F, Klarskov P, et al. Long-term results of Danish Prostatic Cancer Group trial 86. Goserelin acetate plus flutamide versus orchiectomy in advanced prostate cancer. Cancer. 1993;72(12 Suppl):3851-4.
65. Denis L, Smith PH, De Moura JL, et al. Orchidectomy vs. Zoladex plus flutamide in patients with metastatic prostate cancer. The EORTC GU Group. Eur Urol. 1990;18 Suppl 3:34-40.
66. Ryan PG, Peeling WB. U.K. trials of treatment for M1 prostatic cancer. The LH-RH analogue Zoladex vs. orchidectomy. Am J Clin Oncol. 1988;11 Suppl 2:S169-72.
67. Peeling WB. Phase III studies to compare goserelin (Zoladex) with orchiectomy and with diethylstilbestrol in treatment of prostatic carcinoma. Urology. 1989;33(5 Suppl):45-52.
68. Parmar H, Edwards L, Phillips RH, et al. Orchiectomy versus long-acting D-Trp-6-LHRH in advanced prostatic cancer. Br J Urol. 1987;59(3):248-54.
69. Kaisary AV, Tyrrell CJ, Peeling WB, Griffiths K. Comparison of LHRH analogue (Zoladex) with orchiectomy in patients with metastatic prostatic carcinoma. Br J Urol. 1991;67(5):502-8.
70. Keuppens F, Whelan P, Carneiro de Moura JL, et al. Orchidectomy versus goserelin plus flutamide in patients with metastatic prostate cancer (EORTC 30853). European Organization for Research and Treatment of Cancer—Genitourinary Group. Cancer. 1993;72(12 Suppl):3863-9.
71. Newling DW, Denis L, Vermeylen K. Orchiectomy versus goserelin and flutamide in the treatment of newly diagnosed metastatic prostate cancer. Analysis of the criteria of evaluation used in the European Organization for Research and Treatment of Cancer—Genitourinary Group Study 30853. Cancer. 1993;72(12 Suppl):3793-8.
72. Denis L, Robinson M, Mahler C, et al. Orchidectomy versus Zoladex(R) plus Eulexin(R) in patients with metastatic prostate cancer (EORTC 30853). J Steroid Biochem Moled Biol. 1990;37(6):951-9.
73. Denis LJ, Carnelro de Moura JL, Bono A, et al. Goserelin acetate and flutamide versus bilateral orchiectomy: a phase III EORTC trial (30853). EORTC GU Group and EORTC Data Center. Urology. 1993;42(2):119-30.
74. Rosendahl I, Kiebert GM, Curran D, et al. Quality-adjusted survival (Q-TWiST) analysis of EORTC Trial 30853: comparing goserelin acetate and flutamide with bilateral orchiectomy in patients with metastatic prostate cancer. European Organization for Research and Treatment of Cancer. Prostate. 1999;38(2):100-9.
75. Denis LJ, Keuppens F, Smith PH, et al. Maximal androgen blockade: final analysis of EORTC phase III trial 30853. EORTC Genito-Urinary Tract Cancer Cooperative Group and the EORTC Data Center. Eur Urol. 1998;33(2):144-51.
76. Kirby R, Robertson C, Turkes A, et al. Finasteride in association with either flutamide or goserelin as combination hormonal therapy in patients with stage M1 carcinoma of the prostate gland. International Prostate Health Council (IPHC) Trial Study Group. Prostate. 1999;40(2):105-14.
77. Kuhn MW, Weibbach L, Hinke A. Primary therapy of metastatic prostate carcinoma with depot gonadotropin-releasing hormone analogue goserelin versus estramustine phosphate. Urology. 1994;43(2 Suppl):61-7.
78. Thorpe SC, Azmatullah S, Fellows GJ, et al. A prospective, randomised study to compare goserelin acetate (Zoladex) versus cyproterone acetate (Cyprostat) versus a combination of the two in the treatment of metastatic prostatic carcinoma. Eur Urol. 1996;29(1):47-54.
79. Gleave ME, Goldenberg SL, Chin JL, et al. Randomized comparative study of 3 versus 8-month neoadjuvant hormonal therapy before radical prostatectomy: biochemical and pathological effects. J Urol. 2001;166(2):500-6.
80. Prezioso D, Lotti T, Polito M, et al. Neoadjuvant hormone treatment with leuprolide acetate depot 3.75 mg and cyproterone acetate, before radical prostatectomy: a randomized study. Urol Int. 2004;72(3):189-95.
81. D’Amico AV, Manola J, Loffredo M, et al. 6-month androgen suppression plus radiation therapy vs radiation therapy alone for patients with clinically localized prostate cancer: a randomized controlled trial. JAMA. 2004;292(7):821-7.
82. Denham JW, Wilcox C, Joseph D, et al. Quality of life in men with locally advanced prostate cancer treated with leuprorelin and radiotherapy with or without zoledronic acid (TROG 03.04 RADAR): secondary endpoints from a randomised phase 3 factorial trial. Lancet Oncol. 2012;13(12):1260-70.
83. Smith MR, Goode M, Zietman AL, et al. Bicalutamide monotherapy versus leuprolide monotherapy for prostate cancer: effects on bone mineral density and body composition. J Clin Oncol. 2004;22(13):2546-53.
84. Fornara P, Jocham D. Clinical study results of the new formulation leuprorelin acetate three-month depot for the treatment of advanced prostate carcinoma. Urol Int. 1996;56 Suppl 1:18-22.
85. Wechsel HW, Zerbib M, Pagano F, et al. Randomized open labelled comparative study of the efficacy, safety and tolerability of leuprorelin acetate 1M and 3M depot in patients with advanced prostatic cancer. Eur Urol. 1996;30 Suppl 1:7-14; discussion 19-21.
86. Soloway MS, Sharifi R, Wajsman Z, et al. Randomized prospective study comparing radical prostatectomy alone versus radical prostatectomy preceded by androgen blockade in clinical stage B2 (T2bNxM0) prostate cancer. The Lupron Depot Neoadjuvant Prostate Cancer Study Group. J Urol. 1995;154(2 Pt 1):424-8.
87. Geiges G, Schapperer E, Thyroff-Friesinger U, et al. Clinical development of two innovative pharmaceutical forms of leuprorelin acetate. Ther Adv Urol. 2013;5(1):3-10.
88. Tunn UW, Wiedey K. Safety and clinical efficacy of a new 6-month depot formulation of leuprorelin acetate in patients with prostate cancer in Europe. Prostate Cancer Prostatic Dis. 2009;12(1):83-7.
89. Homma Y, Akaza H, Okada K, et al. Early results of radical prostatectomy and adjuvant endocrine therapy for prostate cancer with or without preoperative androgen deprivation. The Prostate Cancer Study Group. Int J Urol. 1999;6(5):229-39.
90. Homma Y, Akaza H, Okada K, et al. Radical prostatectomy and adjuvant endocrine therapy for prostate cancer with or without preoperative androgen deprivation: Five-year results. Int J Urol. 2004;11(5):295-303.
91. Hellström M, Häggman M, Pedersen K, et al. A 3-year follow-up of patients with localized prostate cancer operated on with or without pre-treatment with GnRH-agonist triptorelin. Br J Urol. 1996;78(3):432-6.
92. Hedlund PO, Henriksson P. Parenteral estrogen versus total androgen ablation in the treatment of advanced prostate carcinoma: effects on overall survival and cardiovascular mortality. The Scandinavian Prostatic Cancer Group (SPCG)-5 Trial Study. Urology. 2000;55(3):328-33.
93. Hedlund PO, Ala-Opas M, Brekkan E, et al. Parenteral estrogen versus combined androgen deprivation in the treatment of metastatic prostatic cancer. Scandinavian Prostatic Cancer Group (SPCG) Study No. 5. Scand J Urol Nephrol. 2002;36(6):405-13.
94. Hedlund PO, Damber JE, Hagerman I, et al. Parenteral estrogen versus combined androgen deprivation in the treatment of metastatic prostatic cancer: part 2. Final evaluation of the Scandinavian Prostatic Cancer Group (SPCG) Study No. 5. Scand J Urol Nephrol. 2008;42(3):220-9.
95. Teillac P, Heyns CF, Kaisary AV, et al. Pharmacodynamic equivalence of a decapeptyl 3-month SR formulation with the 28-day SR formulation in patients with advanced prostate cancer. Horm Res. 2004;62(5):252-8.
96. Aus G, Abrahamsson PA, Ahlgren G, et al. Hormonal treatment before radical prostatectomy: a 3-year followup. J Urol. 1998;159(6):2013-6.
97. Aus G, Abrahamsson PA, Ahlgren G, et al. Three-month neoadjuvant hormonal therapy before radical prostatectomy: a 7-year follow-up of a randomized controlled trial. BJU Int. 2002;90(6):561-6.
98. Parmar H, Phillips RH, Lightman SL, et al. Randomised controlled study of orchidectomy vs long-acting D-Trp-6-LHRH microcapsules in advanced prostatic carcinoma. Lancet. 1985;2(8466):1201-5.
99. Iversen P, Karup C, van der Meulen E, et al. Hot flushes in prostatic cancer patients during androgen-deprivation therapy with monthly dose of degarelix or leuprolide. Prostate Cancer Prostatic Dis. 2011;14(2):184-90.
100. Williams G, Lindsay S, Bowsher WG. Randomised crossover trial to assess the tolerability of LHRH analogue administration. Prostate Cancer Prostatic Dis. 2003;6(2):187-9.
101. Brenninkmeijer R, Janknegt R. Application of SOJA and InforMatrix in practice: interactive web and workshop tools. Expert Opin Pharmacother. 2007;8 Suppl 1:S49-55.
102. Sharifi R, Browneller R; Leuprolide Study Group. Serum testosterone suppression and potential for agonistic stimulation during chronic treatment with monthly and 3-month depot formulations of leuprolide acetate for advanced prostate cancer. J Urol. 2002;168(3):1001-4.
103. Gravel P, Samland D, Löffler M, et al. Two innovative pharmaceutical forms of leuprorelin: results from 818 patients with advanced prostate cancer. Adv Ther. 2013;30(3):271-85.
104. European Association of Urology. Heidenreich A, Bastian PJ, Bellmunt J, et al. Guidelines on prostate cancer. March 2013 [homepage on the Internet]. 2013 Mar 6 [cited 2014 Sep 5]. Available from: http://www.uroweb.org/gls/pdf/09_Prostate_Cancer_LR.pdf
|
Author for correspondence: Rob Janknegt, PhamD, PhD, Hospital Pharmcist, Clinical Pharmacologist, Orbis Medisch Centrum, 1 Dr H van der Hoff plein, NL-6162 BG Sittard-Geleen, The Netherlands |
Copyright © 2014 Pro Pharma Communications InternationalDisclosure of Conflict of Interest Statement is available upon request.
Permission granted to reproduce for personal and non-commercial use only. All other reproduction, copy or reprinting of all or part of any ‘Content’ found on this website is strictly prohibited without the prior consent of the publisher. Contact the publisher to obtain permission before redistributing.








You have performed a great job on this article. It’s very precise and highly qualitative. Nobody can stop to admire you. Lots of appreciation.