Importance of manufacturing consistency of the glycosylated monoclonal antibody adalimumab (Humira®) and potential impact on the clinical use of biosimilars
Published on 2016/06/29
Generics and Biosimilars Initiative Journal (GaBI Journal). 2016;5(2):70-3.
Author byline as per print journal: Professor Paul J Declerck, PhD; Paul W Tebbey, PhD
|
Abstract: |
Submitted: 5 May 2016; Revised: 23 June 2016; Accepted: 24 June 2016; Published online first: 7 July 2016
Introduction
Biological therapeutics are highly sensitive to formation of structural variants during manufacturing processes. These structural variants can affect clinical performance. Here, we review important recently published technical data on adalimumab consistency and manufacturing experience from a clinical perspective.
Adalimumab
Adalimumab (Humira®, AbbVie, Inc) is a recombinant human immunoglobulin (Ig) G1 anti-tumour necrosis factor-α (TNF-α) monoclonal antibody originally approved for the treatment of rheumatoid arthritis in the US in 2002, and subsequently approved for the management of Crohn’s disease, ulcerative colitis, plaque psoriasis, psoriatic arthritis, ankylosing spondylitis, juvenile idiopathic arthritis, and hidradenitis suppurativa [1]. Since its approval in the US, many other countries have approved its use for multiple indications [2, 3].
Complex nature of biologicals
As is the case for many biologicals, adalimumab is a large glycoprotein that is a heterogenous mixture of structural isoforms [4–6]. Glycosylation, the addition of sugar residues by the host organism during manufacturing process, is a major post-translational protein modification resulting in different structural isoforms [7, 8]. In addition to glycosylation, enzymatic cleavage of C-terminal lysines contributes to the heterogeneity of monoclonal antibodies [9]. The formation and nature of these structural variants are highly sensitive to manufacturing processes, and changes in any of the manufacturing steps may lead to differences in the specific isoforms that are present.
Differences in glycosylation profile and cleavage of C-terminal lysines can affect the tertiary (i.e. the overall 3-dimensional shape of the protein) and quaternary structure (i.e. interaction of protein subunits) of a biological agent [10]. Thus, it is important to characterize the key quality attributes, e.g. physicochemical and structural features, of a biological drug substance and constantly monitor these during production, using robust analytical methods to help ensure product consistency, including the pattern and percentage of each structural isoform.
Consistency of adalimumab manufacturing
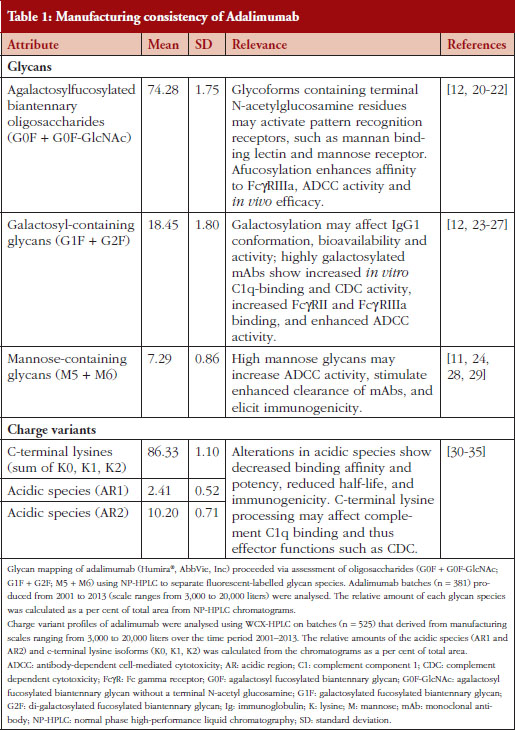
A recent product quality analysis evaluated 544 total batches of adalimumab manufactured between 2000 and 2013 [6]. Molecular charge, as measured by the presence of acidic species and C-terminal lysine variants, and glycosylation pattern were the key properties used to demonstrate the comparability and consistency of the production process, see Table 1 [6]. The quantitation of the sum and overall lysine variant profile demonstrated consistency between the batches over the course of 13 years [6]. Additionally, a high degree of consistency for the sum of lysine variants was observed between five different bioreactor sizes used for production [6]. Comparison of the identity and quantity of adalimumab oligosaccharides profile from 381 different batches indicated a high level of consistency between the batches over time and throughout the production scale changes, see Table 1 [6]. Furthermore, the consistency of the charged species and glycosylation patterns of adalimumab was supported by biological data, demonstrating that its interaction with and intrinsic binding affinity for soluble TNF-α ligand remained stable during this period [6].
Importance of consistency of adalimumab manufacturing
It has been clearly demonstrated that the manufacturing process for adalimumab produced a consistent product over an extended (> 10 years) period of time [6], even as necessary changes to the manufacturing processes and production scale were introduced. It is paramount for manufacturers of biological products to maintain tight control of the drug substance and its production to ensure effective and safe use in patients. In the case of adalimumab, the need for a high level of consistency and product quality may be magnified, in part, because of the diverse patient populations being treated, and the underlying etiologies of the different chronic immune-mediated inflammatory diseases for which adalimumab is indicated.
Variations in glycosylation pattern or other post-translational modifications can result in subtle changes in the conformation of a biological that can potentially alter its solubility, stability, efficacy, or immunogenicity [8, 10]. Studies have demonstrated that variations in glycosylation can lead to significant changes in the circulating levels of the biological subsequent to altered pharmacokinetics, and alter its distribution to specific tissues and organs [7]. For example, therapeutic IgGs containing high-mannose glycans in the Fc region are cleared more rapidly in humans than other glycan forms [11]. Furthermore, some residues, such as galactose-containing glycans, can induce conformational changes that expose portions of the molecule previously hidden from the immune system and possibly lead to an antigenic response [8, 12].
Immunogenicity is a primary area of safety for biologicals and is often carefully monitored [13]. Immunogenic response results in the development of non-neutralizing (sometimes termed binding) or neutralizing anti-drug antibodies against the biological product. Non-neutralizing antibodies bind biological molecules on sites unrelated to target binding; however, non-neutralizing antibodies can reduce drug bioavailability through increased clearance, indirectly decreasing efficacy [14]. Neutralizing antibodies, on the other hand, bind biologicals on sites that interfere with target binding, reducing or sometimes halting efficacy [14]. Immunogenic response can have a number of outcomes ranging from adverse events, such as hypersensitivity reactions, i.e. reactions to drug administration, to reduced serum levels and neutralized activity of the biological; in other cases, it has no apparent effect on efficacy or safety of the product [9].
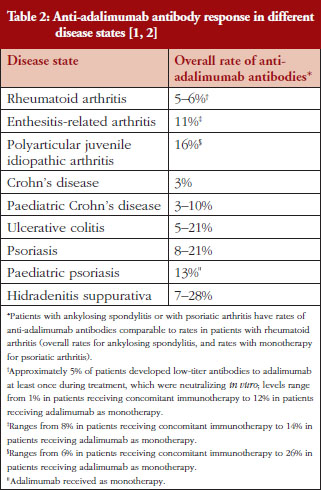
The presence of anti-adalimumab antibodies after treatment varied between 3% and 28% among different disease states, see Table 2 [1, 2, 15–17]. Because the measured incidence of anti-drug and neutralizing antibodies is highly dependent on the assay type and because multiple factors (such as assay sensitivity and specificity, methodology, disease state, sample handling, and timing of sample collection) affect the results, comparison of the incidence of antibodies between studies or between different biologicals is misleading [14]. Nevertheless, presence of anti-adalimumab antibodies, even at moderate concentration, can impact patients’ response to treatment. In studies of rheumatoid arthritis and Crohn’s disease, anti-adalimumab antibodies were associated with lower adalimumab serum levels [16, 17]. Furthermore, a higher percentage of treatment non-responders had anti-adalimumab antibodies compared with treatment responders in one study [16]. Because adalimumab is used for the treatment of patients with chronic immune-mediated inflammatory diseases, it may be important to establish and maintain a long-term immunologic equilibrium in patients treated with adalimumab. By providing a consistent product with key attributes maintained within a narrow window of variability from batch to batch, the risk of potential changes in the antigenicity of adalimumab over time is minimized.
Changes to a tightly controlled manufacturing process that may lead to variations in epitopes could disrupt the immunologically tolerated state of a patient to their initial therapeutic. Likewise, switching a stable patient to a related but different molecule, as would be the case when using a biosimilar of the initial therapeutic made by a different manufacturer, may increase antigenicity. Well-established mechanisms of tolerance include clonal deletion, receptor editing, clonal anergy, blockade of memory response, and competitive tolerance [18]. Overall, immune responses are unpredictable and immunological response to treatment with biological therapies, such as adalimumab, can vary greatly between individual patients and disease states. Furthermore, patients can develop anti-drug antibodies with clinical impact ranging from no effects to secondary loss of response. The immune response to adalimumab is a dynamic process in which anti-drug antibodies can develop and then disappear over time. Whether a stable patient who undergoes a non-medical switch between an initial therapeutic and its biosimilar (which would expose the immune system to distinct antigens) can maintain or subsequently regain tolerance is unknown. Appropriate investigation is recommended because of the paucity of available clinical data that pertain to the phenomenon of switching [19]. Importantly, data regarding the use of multiple biosimilar molecules in a single patient are also lacking.
Conclusions
The comparison of over 500 batches of adalimumab from 2000 to 2013 demonstrated that the key physicochemical and functional quality attributes of adalimumab have remained within a narrow range during this long time frame. Thus, patients who have been or are currently treated with adalimumab have received a consistent product during the course of their treatments, have developed immune tolerance to the antigens consistently delivered, and ultimately have achieved stability in terms of clinical response.
Acknowledgements
AbbVie funded these studies, contributed to their design, and was involved in the collection, analysis and interpretation of the data, and in the writing, review and approval of the publication. Medical writing support was provided by Patrick Little, PhD, and Maria Hovenden, PhD, of Complete Publication Solutions, LLC (North Wales, PA, USA).
Funding source: AbbVie, Inc funded the analyses.
Competing interests: Professor Paul Declerck participated at advisory board meetings for AbbVie, Amgen and Hospira and is on the Speakers’ Bureau of AbbVie, Celltrion, Hospira, Merck Serono and Roche. Dr Paul W Tebbey is an employee of AbbVie, Inc and therefore receives a salary and may own AbbVie stock or stock options.
Provenance and peer review: Not commissioned; externally peer reviewed.
Authors
Professor Paul Declerck, PhD
Department of Pharmaceutical and Pharmaceutical Sciences, KU Leuven, University of Leuven, O & N II, 49 Herestraat, BE-3000 Leuven, Belgium.
Paul W Tebbey, PhD
AbbVie, Inc, US Medical Affairs, Biotherapeutics, AP32-2, 1 N Waukegan Road, North Chicago, IL 60064, USA.
References
1. Humira® adalimumab. North Chicago, IL: AbbVie Inc. 2015 [homepage on the Internet]. [cited 2016 Jun 23]. Available from: https://www.humira.com/?cid=ppc_ppd_ggl_franchise_brand_2015_humira_Phrase_64X1790908
2. European Medicines Agency. Humira summary of product characteristics. 2015 [homepage on the Internet]. [cited 2016 Jun 23]. Available from: http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Product_Information/human/000481/WC500050870.pdf
3. AbbVie Corporation. Humira product monograph. 2015 [homepage on the Internet]. [cited 2016 Jun 23]. Available from: http://docplayer.net/334609-Product-monograph-humira-adalimumab-40-mg-in-0-8-ml-sterile-solution-50-mg-ml-subcutaneous-injection.html
4. Ramanan S, Grampp G. Drift, evolution, and divergence in biologics and biosimilars manufacturing. BioDrugs. 2014;28(4):363-72.
5. Sekhon BS, Saluja V. Biosimilars: an overview. Biosimilars. 2011;1:1-11.
6. Tebbey PW, Varga A, Naill M, Clewell J, Venema J. Consistency of quality attributes for the glycosylated monoclonal antibody Humira® (adalimumab). MAbs. 2015;7(5):805-11.
7. Sola RJ, Griebenow K. Glycosylation of therapeutic proteins: an effective strategy to optimize efficacy. BioDrugs. 2010;24(1):9-21.
8. Revers L, Furczon E. An introduction to biologics and biosimilars. Part II: Subsequent entry biologics: biosame or biodifferent? Can Pharm J. 2010;143(4):184-91.
9. Chirino AJ, Mire-Sluis A. Characterizing biological products and assessing comparability following manufacturing changes. Nat Biotechnol. 2004;22(11):1383-91.
10. Dorner T, Strand V, Castaneda-Hernandez G, et al. The role of biosimilars in the treatment of rheumatic diseases. Ann Rheum Dis. 2013;72(3):322-8.
11. Goetze AM, Liu YD, Zhang Z, et al. High-mannose glycans on the Fc region of therapeutic IgG antibodies increase serum clearance in humans. Glycobiology. 2011;21(7):949-59.
12. Houde D, Peng Y, Berkowitz SA, Engen JR. Post-translational modifications differentially affect IgG1 conformation and receptor binding. Mol Cell Proteomics. 2010;9(8):1716-28.
13. Gils A, Vande Casteele N, Poppe R, et al. Development of a universal anti-adalimumab antibody standard for interlaboratory harmonization. Ther Drug Monit. 2014;36(5):669-73.
14. Shankar G, Pendley C, Stein KE. A risk-based bioanalytical strategy for the assessment of antibody immune responses against biological drugs. Nat Biotechnol. 2007;25(5):555-61.
15. van de Putte LB, Atkins C, Malaise M, et al. Efficacy and safety of adalimumab as monotherapy in patients with rheumatoid arthritis for whom previous disease modifying antirheumatic drug treatment has failed. Ann Rheum Dis. 2004;63(5):508-16.
16. Bartelds GM, Wijbrandts CA, Nurmohamed MT, et al. Clinical response to adalimumab: relationship to anti-adalimumab antibodies and serum adalimumab concentrations in rheumatoid arthritis. Ann Rheum Dis. 2007;66(7):921-6.
17. Baert F, Kondragunta V, Lockton S, et al. Antibodies to adalimumab are associated with future inflammation in Crohn’s patients receiving maintenance adalimumab therapy: a post hoc analysis of the Karmiris trial. Gut. 2016 Jul;65(7):1126-31.
18. Stewart JJ, Agosto H, Litwin S, et al. A solution to the rheumatoid factor paradox: pathologic rheumatoid factors can be tolerized by competition with natural rheumatoid factors. J Immunol. 1997;159(4):1728-38.
19. Ebbers HC, Muenzberg M, Schellekens H. The safety of switching between therapeutic proteins. Expert Opin Biol Ther. 2012;12(11):1473-85.
20. Ward E, Mittereder N, Kuta E, et al. A glycoengineered anti-CD19 antibody with potent antibody-dependent cellular cytotoxicity activity in vitro and lymphoma growth inhibition in vivo. Br J Haematol. 2011;155(4):426-37.
21. Herbst R, Wang Y, Gallagher S, et al. B-cell depletion in vitro and in vivo with an afucosylated anti-CD19 antibody. J Pharmacol Exp Ther. 2010;335(1):213-22.
22. Kanda Y, Yamada T, Mori K, et al. Comparison of biological activity among nonfucosylated therapeutic IgG1 antibodies with three different N-linked Fc oligosaccharides: the high-mannose, hybrid, and complex types. Glycobiology. 2007;17(1):104-18.
23. Krapp S, Mimura Y, Jefferis R, Huber R, Sondermann P. Structural analysis of human IgG-Fc glycoforms reveals a correlation between glycosylation and structural integrity. J Mol Biol. 2003;325(5):979-89.
24. Liu L. Antibody glycosylation and its impact on the pharmacokinetics and pharmacodynamics of monoclonal antibodies and Fc-fusion proteins. J Pharm Sci. 2015;104(6):1866-84.
25. Hodoniczky J, Zheng YZ, James DC. Control of recombinant monoclonal antibody effector functions by Fc N-glycan remodeling in vitro. Biotechnol Prog. 2005;21(6):1644-52.
26. Thomann M, Schlothauer T, Dashivets T, et al. In vitro glycoengineering of IgG1 and its effect on Fc receptor binding and ADCC activity. PloS one. 2015;10(8):e0134949.
27. Kurogochi M, Mori M, Osumi K, et al. Glycoengineered monoclonal antibodies with homogeneous glycan (M3, G0, G2, and A2) using a chemoenzymatic approach have different affinities for FcgammaRIIIa and variable antibody-dependent cellular cytotoxicity activities. PloS one. 2015;10(7):e0132848.
28. Brady LJ, Velayudhan J, Visone DB, et al. The criticality of high-resolution N-linked carbohydrate assays and detailed characterization of antibody effector function in the context of biosimilar development. MAbs. 2015;7(3):562-70.
29. Alessandri L, Ouellette D, Acquah A, et al. Increased serum clearance of oligomannose species present on a human IgG1 molecule. MAbs. 2012;4(4):509-20.
30. Harris RJ, Kabakoff B, Macchi FD, et al. Identification of multiple sources of charge heterogeneity in a recombinant antibody. J Chromatogr B Biomed Sci Appl. 2001;752(2):233-45.
31. Liu H, Ponniah G, Zhang HM, et al. In vitro and in vivo modifications of recombinant and human IgG antibodies. MAbs. 2014;6(5):1145-54.
32. Scallon BJ, Tam SH, McCarthy SG, Cai AN, Raju TS. Higher levels of sialylated Fc glycans in immunoglobulin G molecules can adversely impact functionality. Mol Immunol. 2007;44(7):1524-34.
33. Raju TS, Lang SE. Diversity in structure and functions of antibody sialylation in the Fc. Curr Opin Biotechnol. 2014;30:147-52.
34. Vlasak J, Bussat MC, Wang S, et al. Identification and characterization of asparagine deamidation in the light chain CDR1 of a humanized IgG1 antibody. Anal Biochem. 2009;392(2):145-54.
35. van den Bremer ET, Beurskens FJ, Voorhorst M, et al. Human IgG is produced in a pro-form that requires clipping of C-terminal lysines for maximal complement activation. MAbs. 2015;7(4):672-80.
|
Author for correspondence: Professor Paul Declerck, PhD, Department of Pharmaceutical and Pharmaceutical Sciences, KU Leuven, University of Leuven, O & N II, 49 Herestraat, BE-3000 Leuven, Belgium |
Disclosure of Conflict of Interest Statement is available upon request.
Copyright © 2016 Pro Pharma Communications International
Permission granted to reproduce for personal and non-commercial use only. All other reproduction, copy or reprinting of all or part of any ‘Content’ found on this website is strictly prohibited without the prior consent of the publisher. Contact the publisher to obtain permission before redistributing.