Reference pricing systems in Europe: characteristics and consequences
Published on 2012/07/02
Generics and Biosimilars Initiative Journal (GaBI Journal). 2012;1(3-4):127-31.
Author byline as per print journal: Pieter Dylst, PharmD; Professor Steven Simoens, MSc, PhD; Professor Arnold G Vulto, PharmD, PhD
|
Introduction: A reference pricing system is a system that establishes a common reimbursement level or reference price for a group of interchangeable medicines, i.e. the reference group. This article provides an overview of the different characteristics of the different reference pricing systems in Europe. Additionally, the impact of reference pricing on price competition, generic medicine use, pharmaceutical expenditure and health outcome will be discussed. |
Submitted: 16 May 2012; Revised: 8 August 2012; Accepted: 15 August 2012; Published online first: 17 August 2012
Introduction
A reference pricing system is a system that establishes a reimbursement level or reference price for a group of interchangeable medicines. If a medicine is priced above the reference price, the patient pays the difference between the price of the medicine and the reference price, in addition to any other co-payments, e.g. prescription fee, percentage co-payment [1].
Unlike its name suggests, a reference pricing system is not a pricing system, but in fact a reimbursement system. A reference pricing system sets a common reimbursement level, i.e. reference price, for a group of medicines, thereby generating savings for the third-party payer. Manufacturers are in principle free to set prices, although medicines priced above the reference price incur an additional patient co-payment and generic medicines in some countries, e.g. Belgium, need to be priced below the reference price in order to be reimbursed.
Reference pricing can help governments to contain public pharmaceutical expenditure as it controls the reimbursement level of medicines. A reference pricing system may also promote generic medicine use because originator medicines priced above the level of the reference price are likely to lose market share as a result of the additional patient co-payment.
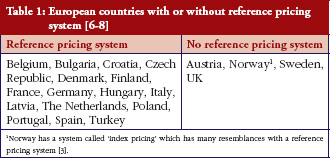
Many European countries have already installed a reference pricing system, see Table 1. Sweden had adopted a reference pricing system in 1993 but abandoned this in 2002 [2]. In Norway, reference pricing applied from 1993 until the end of 2000. In 2003, the Norwegian government installed a system called ‘index pricing’ to a set of off-patent medicines, which has many resemblances with a reference pricing system [3, 4]. Reference pricing is in many European countries combined with other policies such as prescribing by international non-proprietary name or generics substitution, as this combination of policies seems to positively influence each other [5].
This article describes the characteristics of the different reference pricing systems in Europe. Also, the impact of reference pricing on price competition, generic medicine use, pharmaceutical expenditure and health outcome will be discussed. To this effect, a literature review and survey were carried out. The survey was used to document the current status of reference pricing systems in Europe. Survey data were collected from member associations of the European Generic medicines Association in the context of their 2011 survey of European drug retail markets [6].
Reference price methods in Europe
Table 2 gives an overview of the different methods used by European countries to set reference prices. A country may employ one or a combination of method(s) to establish reference prices. The reference price is generally calculated as a function of market prices of medicines. The medicines which are taken into account for the calculation of these reference prices can differ between countries, see Table 2.
The different methods of setting reference prices need to be considered in the context of price competition and the volume of generic medicine use. Some European countries set the reference price at the average price level of (generic) medicines in the reference group. If accompanied by incentives to stimulate demand for generic medicines, generic medicine companies have an incentive to compete, thereby driving down (reference) prices of medicines.
Once the generic medicines market reaches a minimum level of development—for instance, a generic drug market share by volume of 40%—the reference price can be further reduced and set at the level of the lowest priced (generic) medicine. For instance, the high generic medicines market share in combination with reference prices based on the lowest priced medicines in Poland support price competition between companies and maximize savings from generic medicines use. However, other countries such as Italy and Spain have set the reference price based on the lowest priced medicine in the absence of strong incentives to stimulate demand for generic medicines, thereby undermining the development of the generic medicines market [9].
Conversely, in countries with developing generic medicines markets, setting the reference price at a higher level to encourage generic medicines market entry can be introduced as a temporary measure to boost the generic medicines market until it reaches a more mature level of development [9]. For instance, when the reference pricing system was first introduced in Portugal in 2003, the reference price was established at the level of the most expensive generic medicine. This approach increased the number of generics competitors and competition in the generic medicines market [10].
This recommendation reflects the approach used in Germany, where the reference price is calculated as a function of medicine prices and the number of generics competitors. Higher reference prices are awarded in reference groups with fewer generics competitors, thereby stimulating market entry of generic medicines companies. Conversely, reference prices are reduced and price competition is stimulated in established markets, but not to the extent that it becomes economically unviable for generic medicines companies to remain on the market [11].
In Belgium, the reference price is set at 69% (imposed by the government) of the price of the originator medicine on the day that the patent expires. This method has the benefit of guaranteeing savings to health insurance funds, but has in general not generated price reductions of generic medicines below the reference price [12].
Reference group
In a reference pricing system, equivalent medicines are put together in a reference group as defined by:
- active substance, i.e. medicines with the same active substance (ATC-level 5)
- pharmacological class, i.e. medicines with chemically-related active substances that are pharmacologically equivalent (ATC-level 4) or
- therapeutic class, i.e. medicines that have a comparable therapeutic effect (ATC-level 3).
The term ‘generic reference pricing’ refers to a level 1 reference group, whereas ‘therapeutic reference pricing’ relates to level 2 and 3 reference groups [13].
The methods for establishing reference groups in European countries are outlined in Table 3. Countries can implement a combination of these methods to group medicines in reference groups.

The majority of countries group medicines by active substance, see Table 2. Such a method may lead to ‘re-allocation of demand’ away from off-patent medicines included in the reference pricing system towards patented medicines with a similar therapeutic indication that do not fall under the reference pricing system [9].
Re-allocation of demand occurred in Italy in the case of ranitidine: the falling market share of originator ranitidine following the advent of generic medicines was offset by increasing sales of patented medicines with the same therapeutic indication, e.g. omeprazole and its derivatives. Estimates suggest that re-allocation of demand was responsible for an increase in public pharmaceutical expenditure by 3.1% in 2003 [14].
Following the patent expiry of omeprazole in The Netherlands, a higher percentage of patients switched to another proton pump inhibitor (such as pantoprazole and esomeprazole, which are not included in the reference pricing system) than before the patent expiry of omeprazole [15].
In the Valencian region of Spain, the inclusion of fluoxetine in the reference pricing system and the market entry of generics competitors resulted in a shift away from fluoxetine to other antidepressants, e.g. escitalopram, venlafaxine, which were not included in the reference pricing system [16].
In countries that define reference groups by pharmacological class or by therapeutic class, see Table 3, the heterogeneity of medicines within the reference group increases. As a result, physicians may be incited to prescribe a specific medicine for financial reasons, i.e. avoidance of patient co-payment, rather than for clinical reasons, i.e. effectiveness, safety, drug–drug interaction profile [9].
Differences exist between European countries with respect to the consideration of dosage equivalence when establishing reference groups [13]. If there is no differentiation between dosages, e.g. 10 mg, 20 mg and 40 mg of the same active substance are included in the same reference group; 1 mg of active substance will be the cheapest in the highest dosage formulation. Thus, patients have an incentive to switch to the highest dosage of the medicine with the lowest co-payment. Alternatively, reference groups may be defined based on the defined daily dose of medicines. However, one defined daily dose of a specific medicine may not be therapeutically equivalent with one defined daily dose of another medicine in the same reference group. Defined daily doses of medicines also might change over time. World Health Organization therefore stresses that the defined daily dose methodology should not be misused for pricing and reimbursement decisions [17].
Hungary already applied a reference pricing system by active substance since 1997 and introduced a reference pricing system by therapeutic class for, amongst other medicine classes, statins in 2003. Statins were put together in one reference group based on their mechanism of action, without taking into account differences in pharmacologic profile, safety and effectiveness between individual products. As the reference price was determined based on the price per mg, the reference pricing system financially promoted the use of higher-dosed statins such as atorvastatin 40 mg and fluvastatin 80 mg. As a result, the majority of patients switched to higher-dosed statins and the anticipated reduction in the average price of prescribed statins did not materialize [18].
Reference pricing and price competition in Europe
What is the impact of a reference pricing system on price competition? The literature suggests that the introduction of a reference pricing system reduces prices of all medicines that are included in the system [19, 20]. Obviously, price reductions tend to be larger for originator medicines than for generic medicines. Also, greater price reductions have been witnessed in markets where generic medicine competition already occurred prior to the introduction of a reference pricing system [19].
The European experience with respect to price reductions below the reference price is mixed: such price competition has occurred in some countries, e.g. Lithuania [20], but not in others, e.g. Slovenia, Spain [21]. Conversely, some medicines that were originally priced below the reference price actually increased their prices to the level of the reference price following the implementation of a reference pricing system in The Netherlands [22] and in Spain [23]. Therefore, in addition to setting reference prices as a function of the prices of existing medicines, some countries have established a fixed minimum price difference between generic and originator medicines, e.g. Finland, Portugal, Spain [6].
The number of generic medicine competitors within a reference pricing system appears to be associated with the extent of competition, although this impact occurs mainly through discounting to the distribution chain rather than through price competition. Larger discounts have been observed when there are more generic medicine competitors [20, 24].
Finally, as compared with other mechanisms to regulate prices such as price caps i.e. a system where the regulator sets a maximum price that can be charged for a medicine, a Norwegian study reports that a reference pricing system stimulates generics competition to a greater extent and leads to lower prices than price cap regulation [3].
In Germany, medicine prices dropped by 10–26% in the first years following the introduction of the reference pricing system in 1989. This price reduction was greater for originator medicines that faced more generic medicines competition. However, price reductions were counterbalanced by an increase in prices of medicines that were not included in the reference pricing system [25, 26]. In Norway, the implementation of a system called ‘index pricing’ to a set of off-patent medicines in 2003, which has many resemblances with reference pricing, reduced prices of originator medicines by 18% and prices of generic medicines by 8% [3]. In Sweden, medicines covered by the reference pricing system saw a drop in prices by 19% following the introduction of the system [27].
Reference pricing and generic medicine use
In general, the implementation of a reference pricing system has been accompanied by an increase in the use of medicines priced at or below the reference price [13]. However, a literature review indicated that a reference pricing system does not aid generic medicines use if:
- prices of originator medicines fall to the level of the reference price
- the originator company launches new dosages and/or new formulations
- physicians switch to patented medicines with a similar therapeutic indication that do not fall under the reference pricing system (re-allocation of demand) [19].
In Germany, therapeutic reference pricing applies to the group of statins since 2005. As the manufacturer of atorvastatin claimed that atorvastatin was superior to other statins in terms of side effects and drug interaction profile, the manufacturer kept the price of atorvastatin above the reference price. As a result, the market share of atorvastatin declined from 33.3% prior to the reference pricing system to 4.8% in 2006. This volume shifted mainly towards simvastatin to which no additional patient co-payment was applicable as its price was below the reference price [28].
Reference pricing and pharmaceutical expenditure in Europe
A literature review has indicated that reference pricing systems generate savings in the short term, but that savings are probably limited to the one-off impact of the introduction of the reference pricing system and that savings may be substantial at the level of individual medicines, but not necessarily at the level of total pharmaceutical expenditure [19]. Figure 1 shows how the introduction of a reference pricing system may impact long-term pharmaceutical expenditure [29].

The limited scope for savings is to be expected for at least two reasons [29]. First, the coverage of reference pricing systems tends to be limited to off-patent medicines. Patented medicines are included in the reference pricing system by therapeutic class in some countries only. Second, although a reference pricing system may reduce medicine prices and may affect the substitution of new more expensive medicines for older less expensive medicines, it does not affect other drivers of increasing pharmaceutical expenditure such as medicines use by ageing populations, the introduction of expensive biotechnology medicines and orphan medicines, and the transformation from acute to chronic diseases.
Reference pricing and health outcome
A potential concern of therapeutic reference pricing is that switches in medicine use in response to the reference pricing system may adversely affect patient health outcomes and thereby increase the use (and costs) of other healthcare services. There are few studies that explore the impact of reference pricing systems on health and they suffer from methodological limitations. Nonetheless, the available evidence suggests that there is no association between reference pricing systems and health outcomes [13, 19].
A related issue is whether a reference pricing system influences access to health (care) of patient groups with a different socio-economic status. For instance, are patients with a lower socio-economic status more likely to buy costly originator medicines (incurring an additional patient co-payment) in a reference pricing system, thereby placing a financial burden on patient groups with a lower ability to pay? Very few studies have investigated this issue, but a recent Belgian study showed that patients with a lower socio-economic status tended to buy the cheapest (generic) medicines within a reference group and incurred lower medication costs than patients with a higher socio-economic status, thereby laying to rest potential socio-economic equity concerns related to reference pricing systems [8].
Conclusion
Reference pricing is used by many European countries as one element of governments’ strategies to contain public healthcare expenditure. It puts pressure on pharmaceutical companies to compete with the reference priced product but also reduces competition beyond this reference price. It also makes patients sensitive of drug prices, as an increased use of drugs priced at or below the reference price level was observed. The introduction of reference pricing generates a once-only setback of expenditures but does not affect the overall growth rate of health expenditure in the long term. No association between reference pricing and health outcome has been observed.
For patients
Over the last decades, European healthcare budgets have been increasing. Therefore, governments have implemented reference pricing, amongst other measures, in order to contain the pharmaceutical budget. In the reference pricing system, a common reimbursement level is set for a group of comparable and interchangeable medicines. The difference between this reimbursement level and the actual price of the medicine is paid by the patient, a so-called co-payment. This provides patients with a financial incentive to opt for the least expensive medicine. Reference pricing seems to reduce prices of medicines, increases the use of cheaper medicines, i.e. without co-payment, generates savings for healthcare budgets in the short term and does not seem to have a negative influence on the health of patients.
Competing interests: Professor Steven Simoens holds the EGA Chair ‘European policy towards generic medicines’. The authors have no conflicts of interest that are directly relevant to the content of this manuscript. This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Provenance and peer review: Commissioned; externally peer reviewed.
The paper is abstracted based on the presentation at the 24th Annual EuroMeeting, Drug Information Association, 28 March 2012, Copenhagen, Denmark.
Co-authors
Professor Steven Simoens, MSc, PhD, Research Centre for Pharmaceutical Care and Pharmacoeconomics, Katholieke Universiteit Leuven, Onderwijs en Navorsing 2, PO Box 521, 49 Herestraat, BE-3000 Leuven, Belgium
Professor Arnold G Vulto, PharmD, PhD, Deputy Head Hospital Pharmacy, Professor of Hospital Pharmacy and Practical Therapeutics, Erasmus University Medical Center, PO Box 2040, NL-3000 CA Rotterdam, The Netherlands
References
1. Folino-Gallo P, Muscolo L, Vogler S, Morak S. PHIS Glossary: Glossary for pharmaceutical policies/systems developed in the Pharmaceutical Health Information Systems (PHIS) Project. PHIS/AIFA/GÖG; July 2009 [update 2011 Apr].
2. Vogler S, Habl C, Leopold C, Rosian-Schikuta I, de Joncheere K, Thomsen TL. PPRI Report. Vienna, Austria: Commissioned by European Commission, Directorate-General Health and Consumer Protection and Austrian Federal Ministry of Health, Family and Youth; 2008.
3. Brekke K, Grasdal A, Holmås TH. Regulation and pricing of pharmaceuticals: Reference pricing of price cap regulation? Eur Econ Rev. 2009;53:170-85.
4. Håkonsen H, Horn AM, Toverud E-L. Price control as a strategy for pharmaceutical cost containment – What has been achieved in Norway in the period 1994–2004? Health Policy. 2009;90(2):277-85.
5. Vogler S. The impact of pharmaceutical pricing and reimbursement policies on generics uptake: implementation of policy options on generics in 29 European countries—an overview. Generics and Biosimilars Initiative Journal (GaBI Journal). 2012;1(2):93-100. doi:10.5639/gabij.2012.0102.020
6. European Generic medicines Association. 2011 Market Review. Brussels, Belgium: European Generic medicines Association; 2011.
7. Vogler S, Habl C, Bogut M, Voncina L. Comparing pharmaceutical pricing and reimbursement policies in Croatia to the European Union Member States. Croat Med J. 2011;52:183-97.
8. Vrijens F, Van de Voorde C, Farfan-Portet M-I, le Polain M, Lohest O. The reference price system and socio-economic differences in the use of cheap medicines. Brussels, Belgium: KCE; 2010. Report No.: 126A.
9. Simoens S, De Coster S. Sustaining generic medicines markets in Europe. Journal of Generic Medicines. 2006;3(4):257-68.
10. Portela C. Reference pricing system and competition: case study from Portugal. Croat Med J. 2009;50(5):429-39.
11. Stargardt T, Schreyögg J, Busse R. [Pharmaceutical reference pricing in Germany: definition of therapeutic groups, price setting through regression procedure and effect]. Gesundheitswesen. 2005;67(7):468-77.
12. Simoens S, De Bruyn K, Bogaert M, Laekeman G. Pharmaceutical policy regarding generic drugs in Belgium. Pharmacoeconomics. 2005;23(8):755-66.
13. Dylst P, Vulto A, Simoens S. The impact of reference-pricing systems in Europe: a literature review and case studies. Expert Rev Pharmacoecon Outcomes Res. 2011;11(6):729-37.
14. Garattini L, Ghislandi S. Off-patent drugs in Italy. A short-sighted view? Eur J Health Econ. 2006;7:79-83.
15. Klok RM, Boersma C, Oosterhuis I, Visser ST, De Jong-Van den Berg LTW, Postma MJ. Switch patterns before and after patent expiry of omeprazole: a case study in The Netherlands. Aliment Pharmacol Ther. 2006;23(11):1595-600.
16. Ubeda A, Cardo E, Sellés N, Broseta R, Trillo JL, Fernández-Llimós F. Antidepressant utilization in primary care in a Spanish region. Impact of generic and reference-based pricing policy (2000–2004). Soc Psychiatry Psychiatr Epidemiol. 2007;42(3):181-8.
17. World Health Organization Collaborating Centre for Drug Statistics and Methodology. Use of ATC/DDD. Oslo, Norway: WHOCC; 2012.
18. Kaló Z, Muszbek N, Bodrogi J, Bidló J. Does therapeutic reference pricing always result in cost-containment? The Hungarian evidence. Health Policy. 2007;80(3):402-12.
19. Galizzi MM, Ghislandi S, Miraldo M. Effects of reference pricing in pharmaceutical markets: a review. Pharmacoeconomics. 2011;29(1):17-33.
20. Garuolienè K, Godman B, Gulbinovic J, Wettermark B, Haycox A. European countries with small populations can obtain low prices for drugs: Lithuania as a case history. Expert Rev Pharmacoecon Outcomes Res. 2011;11(3):343-9.
21. Puig-Junoy J. Impact of European pharmaceutical price regulation on generic price competition: a review. Pharmacoeconomics. 2010;28(8):649-63.
22. Danzon P, Ketcham J. Reference pricing of pharmaceuticals for Medicare: evidence from Germany, the Netherlands and New Zealand. Cambridge, MA: National Bureau of Economic Research; 2004.
23. Puig-Junoy J. The impact of generic reference pricing interventions in the statin market. Health Policy. 2007;84(1):14-29.
24. Puig-Junoy J. Do higher-priced generic medicines enjoy a competitive advantage under reference pricing? Appl Health Econ Health Policy. 2012; in press.
25. Kanavos P, Costa-Font J, Seeley E. Competition in off-patent drug markets: Issues, regulation and evidence. Economic Policy. 2008;23(55):499-544.
26. Pavcnik N. Do pharmaceutical prices respond to potential patient out-of-pockets expenses? Rand J Econ. 2002;33(3):469-87.
27. Andersson K, Petzold M, Sonesson C, Lönnroth K, Carlsten A. Do policy changes in the pharmaceutical reimbursement schedule affect drug expenditure? Interrupted time series analysis of cost, volume and cost per volume trends in Sweden 1986–2002. Health Policy. 2006;79(2):231-43.
28. Stargardt T. The impact of reference pricing on switching behaviour and healthcare utilization: the case of statins in Germany. Eur J Health Econ. 2010;11:267-77.
29. Dylst P. The impact of reference-pricing systems in Europe. 24th Annual EuroMeeting, DIA, 2012 March 28, Copenhagen, Denmark.
|
Author for correspondence: Pieter Dylst, PharmD, Research Centre for Pharmaceutical Care and Pharmacoeconomics, Katholieke Universiteit Leuven, Onderwijs en Navorsing 2, PO Box 521, 49 Herestraat, BE-3000 Leuven, Belgium |
Disclosure of Conflict of Interest Statement is available upon request.
Permission granted to reproduce for personal and non-commercial use only. All other reproduction, copy or reprinting of all or part of any ‘Content’ found on this website is strictly prohibited without the prior consent of the publisher. Contact the publisher to obtain permission before redistributing.
Related article
Reference price systems: stakeholder dialogue and involvement
 [
[ [
[

