Clinical trials for trastuzumab biosimilars
Published on 2020/04/29
|
Abstract: |
Submitted: 26 March 2020; Revised: 2 April 2020; Accepted: 2 April 2020; Published online first: 15 April 2020
Roche’s originator trastuzumab biological (Herceptin) was approved by the US Food and Drug Administration (FDA) in September 1998 and by the European Medicines Agency (EMA) in August 2000 [1]. The patents on Herceptin expired in Europe in July 2014 and in the US in June 2019 [1].
Trastuzumab is a monoclonal antibody that binds to and inactivates the human epidermal growth factor receptor 2 (HER2)/neu receptor. In some cancers, notably certain types of breast cancer, HER2 is overexpressed and causes cancer cells to reproduce. Trastuzumab is therefore used to treat HER2 positive (HER2+) breast cancers.
EMA developed the first guidelines for the approval of biosimilars via an abbreviated registration process during 2005 to 2006, and since then EMA has developed many general and specific guidelines for biosimilars [2]. In the US, a legal framework for approving biosimilars was established in 2009, via the Biologics Price Competition and Innovation Act of 2009 (BPCI Act). Since then, FDA has issued several guidance documents on biosimilars [3].
In order to gain regulatory approval for biosimilars a package of data that includes analytical, non-clinical and clinical data is required. A comprehensive comparison of the quality attributes of the biosimilar with the reference product using state-of-the-art methods must support the conclusion that the products are biosimilar on a structural and functional level [2, 3]. However, even with robust analytical data, the biosimilar can be approved only with additional non-clinical and clinical data. Hence the need for clinical trials.
Clinical trials aim to resolve any uncertainties that may remain following non-clinical development regarding the similarity of the proposed biosimilar with the reference product. Pharmacokinetic and pharmacodynamic studies form the backbone of early clinical development and serve to inform phase III clinical development. Factors to be considered in clinical development include study population, design, endpoint, sample size, duration and analytical methods.
To demonstrate biosimilarity for regulatory approval, clinical trials of biosimilars should therefore use sensitive endpoints with the aim of ensuring that there are no clinically meaningful differences in efficacy, safety or immunogenicity.
Many companies have developed trastuzumab biosimilars [4]. These include major players such as South Korean biotechnology company Celltrion, which gained European Commission (EC) approval for its trastuzumab biosimilar Herzuma (CT-P6) in February 2018 [5] and FDA approval in December 2018 [6].
Other major players who have completed or are carrying out clinical trials for candidate biosimilars include US-based biotech giant Amgen, Korea-based Samsung Bioepis (Samsung and Biogen’s joint venture), as well as collaborations such as that between Japan-based Daiichi Sankyo and UK-based AstraZeneca.
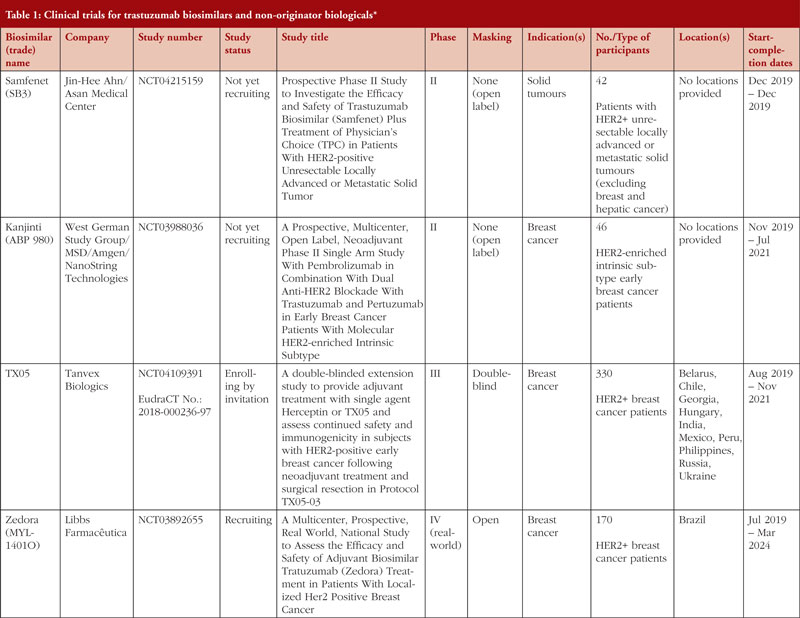
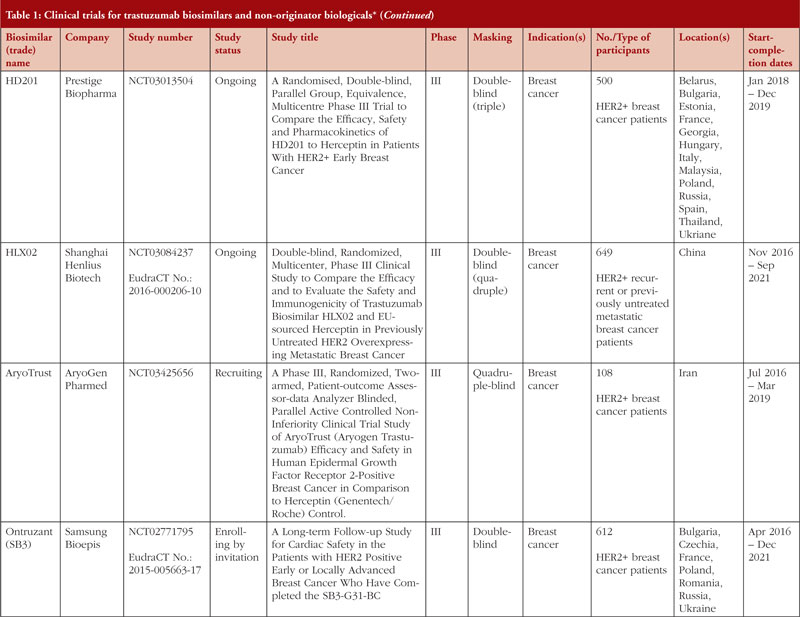
Clinical trials being carried out by these companies include those listed in Table 1. As of 30 March 2020, 19 clinical trials relevant to trastuzumab biosimilar development covering 13 different candidate biosimilars and non-originator biologicals were identified by a search of ClinicalTrials.gov and the EU Clinical Trials Register using the search terms ‘trastuzumab’ AND ‘biosimilar’. The searches were carried out on 26 March 2020 and included all clinical trials recorded in the databases until that date.
Editor’s comment
European Medicines Agency regulatory requirements ensure the same high standards of quality, safety and efficacy for biosimilars as for originator biologicals, and also include a rigorous comparability exercise with the reference product but they are not universally accepted by regulatory bodies outside of the European Union (EU). It should be noted that non-originator biologicals approved only outside of Europe might not have been authorized if they had been subjected to the strict regulatory processes required for approval of biosimilars in the EU.
Competing interests: None.
Provenance and peer review: Commissioned; internally peer reviewed.
Michelle Derbyshire, PhD, GaBI Online Editor
References
1. Derbyshire M. Patent expiry dates for biologicals: 2018 update. Generics and Biosimilars Initiative Journal (GaBI Journal). 2019;8(1):24-31. doi:10.5639/gabij.2019.0801.003
2. GaBI Online – Generics and Biosimilars Initiative. EU guidelines for biosimilars [www.gabionline.net]. Mol, Belgium: Pro Pharma Communications International; [cited 2020 Apr 2]. Available from: www.gabionline.net/Guidelines/EU-guidelines-for-biosimilars
3. GaBI Online – Generics and Biosimilars Initiative. US guidelines for biosimilars [www.gabionline.net]. Mol, Belgium: Pro Pharma Communications International; [cited 2020 Apr 2]. Available from: www.gabionline.net/Guidelines/US-guidelines-for-biosimilars
4. GaBI Online – Generics and Biosimilars Initiative. Biosimilars of trastuzumab [www.gabionline.net]. Mol, Belgium: Pro Pharma Communications International; [cited 2020 Apr 2]. Available from: www.gabionline.net/Biosimilars/General/Biosimilars-of-trastuzumab
5. GaBI Online – Generics and Biosimilars Initiative. EC approval for trastuzumab biosimilar Herzuma [www.gabionline.net]. Mol, Belgium: Pro Pharma Communications International; [cited 2020 Apr 2]. Available from: www.gabionline.net/Biosimilars/News/EC-approval-for-trastuzumab-biosimilar-Herzuma
6. GaBI Online – Generics and Biosimilars Initiative. FDA approves trastuzumab biosimilar Herzuma [www.gabionline.net]. Mol, Belgium: Pro Pharma Communications International; [cited 2020 Apr 2]. Available from: www.gabionline.net/Biosimilars/News/FDA-approves-trastuzumab-biosimilar-Herzuma
Disclosure of Conflict of Interest Statement is available upon request.
Copyright © 2020 Pro Pharma Communications International
Permission granted to reproduce for personal and non-commercial use only. All other reproduction, copy or reprinting of all or part of any ‘Content’ found on this website is strictly prohibited without the prior consent of the publisher. Contact the publisher to obtain permission before redistributing.