Challenges in the manufacture, storage, distribution and regulation of traditional and novel vaccines
Published on 2022/02/28
Generics and Biosimilars Initiative Journal (GaBI Journal). 2022;11(1):13-24.
Author byline as per print journal: Adjunct Associate Professor Sia Chong Hock1, BSc (Pharm), MSc; Adelia Pheh1, BSc (Pharm) (Hon); Vimal Sachdeva2, MSc; Associate Professor Chan Lai Wah1, BSc (Pharm) (Hon), PhD
|
Abstract: |
Submitted: 11 January 2022; Revised: 11 February 2022; Accepted: 13 February 2022; Published online first: 25 February 2022
Introduction
Each year, millions of people worldwide receive vaccines to protect themselves from infectious diseases that may otherwise be fatal [1]. Today, a majority of infants are subject to specific vaccines as part of post-birth care [2]. From the well-known influenza and chickenpox vaccines with a relatively longer history of use [3] to the more recently developed COVID-19 vaccines [4], there is a myriad of different types of vaccines in use today. Traditionally, vaccines directly mimic a milder form of the infection to stimulate antibody production in the body of a healthy individual. But, beyond this, there are now novel vaccines that leverage on different technologies to improve their stability and efficacy [5].
Despite the large number of vaccines available, newer vaccines are still being introduced to the regional and global markets [6, 7]. A major reason is the presence of many existing and emerging diseases that still lack a proper preventive vaccine [8] alongside the ever-evolving variants of such diseases, as in the case of the COVID-19 virus which has evolved from the alpha, beta, delta and into the omicron variants of concern. Additionally, the formulations of many current vaccines are also being improved to lengthen their short-lived protection or to expand their coverage of the disease, including the emerging strains of viruses. The potential benefits of improved formulations are very promising. However, there had been an overall drop in the number of vaccines introduced globally in the recent years [2] due largely to challenges in ensuring the safety of vaccines [9].
Vaccines are of paramount importance to the control of infections [5] and studies have shown that they can drastically reduce the rate of infections [10, 11]. They confer immunity against specific potentially fatal diseases such as smallpox, diphtheria, tuberculosis, hepatitis, influenza and COVID-19 [12]. In fact, vaccines are the primary prevention method against many diseases [13] and are administered to healthy individuals as prophylaxis. While there are overwhelming evidence that vaccines can prevent diseases and save lives, vaccines are never completely safe [14] and side effects are inevitable [15].
Being biological in nature, the manufacture, storage, distribution and handling of vaccines require strict temperature control to maintain their quality [16]. They are considered cold chain products as vaccines have components that are very sensitive to temperature changes [17]. The safety, quality and efficacy of vaccines may be significantly compromised if they are not handled under appropriate conditions [18] at any stage of their product life cycle, from manufacture to distribution to their administration to individuals. Across the world, regulatory authorities adopt specific national and international standards to assure the safety, quality and efficacy of vaccines. It is of paramount importance that there are adequate regulatory oversight to ensure that vaccines remain safe and efficacious when they are administered to individuals at the point of use [19].
Although many studies have identified various challenges in the formulation of novel vaccines, few have addressed the challenges in the manufacture and quality assurance of such vaccines, post-formulation [20-23], as well as the challenges that are common to both traditional and novel vaccines.
Hence, this manuscript aims to provide a better understanding of the evolution of vaccines and identify the challenges in vaccine manufacture, storage, distribution and their regulation. This manuscript also intends to evaluate whether existing national and international regulatory frameworks for vaccines are sufficient to address these challenges, and to propose improvements.
Classification of vaccines
There are many different types of vaccines, and their key differences may form the basis for the need of specific control measures during manufacturing and regulatory control. Regardless of its classification, a vaccine is scientifically defined as a pharmacological compound for improving immunity to a specific disease [24, 25]. All vaccines have a general mechanism of action where the body recognizes the vaccine’s components (associated with the disease pathogen) as foreign antigens and thereby stimulates antibody production against the specific antigens. Active immunity is acquired [26] as future exposure to the same pathogen would trigger memory cells to begin a chain of signals leading to suppression and removal of the pathogen [27]. Vaccines may be classified according to the types of pathogenic component that it contains [28-30]. Vaccines can also be broadly classified as traditional or novel. Table 1 summarises some common vaccines according to class, type and composition.
Traditional vaccines
Traditional vaccines were the earliest developed vaccines [59]. Generally, they contain whole pathogens or pathogenic subunits [60] which are directly recognized by the body’s immune cells [61, 62]. Whole pathogen vaccines are the oldest vaccines with many studies supporting their efficacy [63-65], and they may be further classified into live attenuated or inactivated vaccines. Live attenuated vaccines contain modified whole bacteria or viruses with decreased virulence, sufficient to induce an immune response but not cause disease. Such vaccines are occasionally unsuitable for immunocompromised patients due to the risk of reversion to its virulent state [66] and in such cases, inactivated vaccines may be used instead. Inactivated vaccines contain whole bacteria or viruses that have been chemically or heat-killed and are hence unable to replicate. One example of such vaccine often used for children is the Inactivated Polio Vaccine (IPV) administered to pre-school children.
Another class of traditional vaccines comprises subunit vaccines which are acellular. The recombinant vaccines consist of bacterial or viral protein fragments as the antigen for immune cell recognition [67]. Bacterial toxins are also used in vaccines, but they are often inactivated to form toxoids that can trigger an immune response without causing disease. Vaccines using such toxoids are known as toxoid vaccines. Studies have shown that some bacterial polysaccharides used in vaccines are more efficacious in inducing an immune response when conjugated to proteins such as diphtheria or tetanus toxoid proteins due to the toxoid’s high affinity for immune cell recognition. Hence, conjugate vaccines such as the Haemophilus-Influenzae type b vaccine, containing polysaccharides conjugated to the tetanus toxoid, have been developed as well.
Apart from toxoids, newer technology allows a non-infectious component of bacterial outer cell membrane known as outer membrane vesicle (OMV) to be used as an antigen in OMV vaccines. An example of a licensed OMV vaccine is the Bexsero® vaccine used in the US against the Type B meningococcal virus which causes meningitis and sepsis [68]. Another newer traditional vaccine is the virus-like particle (VLP) vaccine which contain naturally occurring or chemically synthesised VLPs as the antigen of interest. In fact, VLP vaccines can also be manufactured with different antigens from multiple pathogens incorporated together.
While vaccines can be separated into the above-mentioned classes according to their characteristics, distinct classification is occasionally impractical such as in the case of combination vaccines. One combination vaccine is the Infanrix Hexa® 6-in-1 vaccine used in the UK [69] containing both inactivated viruses and recombinant viral proteins. The single combination traditional vaccine offers protection against six diseases, namely diphtheria, tetanus, pertussis, polio, influenza B and hepatitis B [70].
Novel vaccines
Traditional approaches to conferring immunity may be ineffective for chronic or newer infections that require more specific focus on certain antigens [71]. Hence, new methods of delivering pathogenic antigens have been developed. Novel vaccines are a recent development and they rely on pathogenic nucleic material or other alternative vector delivery systems instead of the specified pathogen [72]. The most well-known novel vaccines are nucleic acid-based vaccines that use genetic material of the pathogen, such as mRNA and DNA, to elicit an immune response [60, 73]. mRNA vaccines contain lipid enveloped mRNA of the pathogen that are ultimately translated by human cells to produce pathogenic proteins that act as immune cell antigens. The Pfizer BioNTech® and Moderna® vaccines against COVID-19 are the two most recent examples. DNA vaccines contain bacterial or viral DNA, which do not require the protection of any lipid membranes due to its higher stability relative to mRNA. The pathogenic DNA undergoes additional transcription to mRNA before embarking on a pathway similar to mRNA [74].
Live attenuated vaccines are the most widely used traditional vaccines due to the better-established balance between their immune effect and safety [61]. They are also relatively long lasting [75]. On the other hand, mRNA vaccines make up the biggest group of novel vaccines to date [76], having undergone the most extensive research and development among the novel vaccines [77, 78]. For simplicity, this review will focus on live attenuated vaccines and mRNA vaccines as examples of traditional and novel vaccines, respectively.
Vaccine manufacture
The manufacture of vaccines is an elaborate process chain involving many well-coordinated steps [79]. Depending on the composition of a vaccine, the complexity of the steps may differ. It is also more complex to manufacture combined vaccines, e.g. MMR vaccine, than single vaccines. Figure 1 shows the different levels of complexity in the manufacture of different types of traditional vaccines.
Depending on the type of traditional vaccines, the complexity and need for additional steps will vary. However, traditional vaccines tend to have relatively less complicated steps compared to novel vaccines which require a more precise coordination of steps. However, there are general manufacturing steps that are common to most vaccines, both traditional and novel, as summarised in Figure 2.
The first step in the manufacture of traditional vaccines is to generate the antigen used to stimulate antibody production. This antigen is specific to each vaccine and will require specific production conditions. Most traditional vaccines require the growth of a pathogen, such as viruses or bacteria, as the antigen. These pathogens are commonly grown in various cell cultures. Eggs and mammalian cells are most commonly used for viruses. The candidate vaccine virus is injected into these eggs/cells that are later incubated to allow virus replication. Some methods use a chemical bioreactor to provide a favourable environment for growth of bacteria. The manufacture of novel vaccines vary significantly as replication of pathogenic genetic material is required instead.
For mRNA or DNA vaccines, replication of pathogenic DNA is the first step of its manufacture. The biosynthesis of DNA begins when plasmids containing specific viral DNA are inserted into bacterial cells such as Escherichia coli (E.coli), and these genetically modified bacteria are allowed to replicate in bioreactors. For mRNA vaccines, additional transcription of the DNA to mRNA is completed using specific enzymes and chemicals. Novel vaccines using viral vectors will require an additional step as the DNA plasmid will need to be inserted into non-pathogenic vector viral cells before replication.
In traditional vaccines manufacture, the resultant antigen has to be isolated from the culture medium and purified. For most vaccines, this begins with the separation of the pathogen from its cell culture medium. From here, each vaccine type will require specific additional steps. Live attenuated vaccines will require reduction in the pathogen’s virulence through multiple sequential cell cultures or chemical processes to decrease virulence [79]. One example is the Bacillus Calmette-Guerin (BCG) vaccine, where live strains of the bacterium, namely, Mycobacterium bovis (M. bovis) [82] are weakened and used to confer immunity against tuberculosis [83]. The pathogens used in inactivated vaccines undergo inactivation by heat [84], chemicals like hydrogen peroxide [85] or ultraviolet (UV) radiation [86]. Subunit vaccines will require physical disruption of whole pathogens to obtain the specific antigens needed, such as viral proteins or bacterial toxins.
Thereafter, the isolated antigens undergo multiple purification steps, including filtration, chromatography, clarification and concentration [87]. Simple purification methods exploit particle-size difference, where hollow fibres or flat screens are used to filter out antigens of a specific size. The liquid containing the antigens can be flushed in a direction parallel to the filter, known as tangential flow filtration, to ensure continuous filtration and better recovery of the antigens. When the size difference between contaminants and antigens are significantly less distinct, high affinity chromatography is a common method of purification [88]. The antigens and other components are separated based on their ionic charges or hydrophobic interactions instead. For nucleic acid-based novel vaccines, DNA and mRNA have to be isolated and purified.
After purification, the next step is to formulate the vaccine by incorporating the relevant components [89]. Antigens may be combined with an adjuvant to intensify the immune response triggered [90]. Stabilizers, such as surfactants [91], are added to extend the shelf life of vaccines. Some multi-dose vaccine formulations include preservatives to prevent unwanted microbial contamination [92]. All the above-mentioned steps are normally carried out in a segregated cleanroom of the manufacturing facility as an aseptic environment is required to prevent unwanted microbial contamination.
Finally, the manufactured vaccines are filled into sterile depyrogenated vials in an aseptic environment. The freshly manufactured vaccines are sealed with sterile stoppers together with an outer cap to enhance the physical protection against contamination. After filling, the vials are clearly labelled. Upon completion, the sealed, labelled vaccine vials will undergo strict testing and inspection using specialised equipment to ensure container-closure integrity and to eliminate any defects that may compromise the vaccine’s quality. Throughout the entire manufacturing process, the raw materials and products are to be kept strictly at their respective optimal temperatures.
Vaccine storage, transport and distribution
Once manufactured, the vaccines are stored within the manufacturing facility at their recommended temperature until they are ready for distribution. The storage, transport and distribution of vaccines are constantly managed under temperature-regulated environments. This is because high temperature can cause denaturation of the vaccine antigen and adjuvants. Hence, post-manufacture handling of vaccines involves a collective and continuous monitoring programme known as the vaccine cold-chain management [93] as all vaccines are cold chain products (CCP). The management of CCPs require high quality temperature control within a stringent temperature range, commonly at 2°C‒8°C for most traditional vaccines [94]. Some traditional vaccines, such as the hepatitis B vaccine and diphtheria vaccine, are prone to freeze damage [95, 96]. Under freezing temperature, the vaccines experience potency loss. Administration of such freeze-damaged vaccines can result in an increased risk of adverse effects such as sterile abscesses [97]. On the other hand, novel vaccines, especially nucleic acid-based vaccines, need to be stored at sub-zero temperatures in order to maintain their potency. The mRNA or DNA in such vaccines are highly susceptible to enzymatic damage and hence ultra-low temperatures are necessary to minimize enzymatic activity and any genetic material damage. Therefore, vaccines have to be kept within the appropriate temperature ranges that are specific to the individual vaccines. Table 2 summarises some optimal temperatures of the different types of vaccines.
The actual storage temperature of a vaccine will vary as it travels from the manufacturing facility to the destination country and vaccination centre. The Pfizer-BioNTech COVID-19 vaccine may be stored long term in ultracold freezers at a temperature of between -90°C and -60°C. The vaccines arrive at the warehouses of destination countries in thermal shippers packed with dry ice, see Figure 3. These thermal shippers maintain the vaccines at the required ultra-cold temperature of between -90°C and -60°C. At the destination country, the temperature monitoring device in the thermal shipper is checked to assure that there are no temperature excursions during transportation.
The vaccines are transferred to higher temperatures in a step-wise manner before use. Before mixing with the diluent for administration, the vaccine may be stored in a pharmaceutical refrigerator between 2°C and 8°C for up to 1 month (31 days). Upon mixing with the diluent (sterile saline), the reconstituted vaccines can be left at room temperature (2°C to 25°C) for up to 6 hours. No refreezing of such vaccines is allowed after reconstitution. Any remaining unused vaccine must be discarded after 6 hours [98, 99].
Regulatory controls
The inherent need for scientifically sound vaccine regulation is acknowledged by regulatory authorities, both globally and nationally [100]. All countries should have an organization that is legally responsible for vaccine regulatory actions. Vaccines are a distinct class of biological products which need to be subject to specific regulations due to their unique characteristics. Since there is currently no single biopharmaceutical products classification system that clearly defines vaccines and their respective scope of regulation [101], national regulatory authorities (NRAs) across different nations have set their own regulations. Table 3 summarises some vaccine-producing countries and the names of their NRAs.
Apart from NRAs, there are other international organizations (IO) that aid in the harmonization of vaccine regulation. Although not defined as a regulatory agency (RA), these IOs are important in vaccine control as they act as a benchmark organization for regulation by setting standards recognized by most countries. These IOs set guidelines which form the basis of regulations enforced by the NRAs. The World Health Organization (WHO) is one IO that is a key player in vaccine regulation by its provision of harmonized standards for NRAs. While different NRAs have slightly varying standards, most vaccines are regulated similarly in accordance with WHO or internationally standardized guidelines [103].
Regulatory control of vaccines begins with the development of the vaccine where multiple clinical trials are required before it is eventually licensed for manufacture. This review focuses on post-licensure regulations relating to vaccines manufacture and post-manufacture handling.
All procedures involving vaccine handling are bound by specific regulatory requirements, and details set by the respective NRAs. Firstly, vaccine manufacturers require a licence to operate, regardless of their country of origin [104]. The licence is granted under the condition that a set of standard manufacturing procedures is established and only this approved set of procedures is permitted at the specific manufacturing facility. In the US, vaccine manufacturers are also required to have a functional department reporting any proposed changes to the US Food and Drug Administration (FDA), Centre for Biologics Evaluation and Research (CBER). Strictly no digression of the standard procedures, raw materials or equipment will be condoned by FDA until it is approved by CBER [105]. Vaccine manufacturers are also required to complete and produce all necessary documentations for inspection at all times [105]. FDA stipulates all manufacturing information and documentations required for the Biologics License Application (BLA) [106].
In general, vaccine manufacture is strictly required to be performed in cleanrooms, that are specifically designed to allow for sterile manufacture of products in accordance with good manufacturing practices (GMP) guidelines [107]. These GMP guidelines have been prepared by WHO and other NRAs and IOs, e.g. US FDA, EMA and PIC/S, and they specify precise measures required to ensure the manufacture of safe and good-quality vaccines. A specific portion of the guidelines is used internationally as a reference for individual countries to set their national GMP requirements in vaccine manufacturing facilities [108]. This means that the cleanrooms have to maintain a certain GMP grade before they can operate. Figure 4 shows some examples of cleanroom grades for the different stages of vaccine manufacturing.
Additionally, the US Centres for Disease Control and Prevention (CDC) will assign the biosafety levels (BSLs) to the vaccine-related facilities after assessment of the level of precautionary measures required. Different vaccine types require different BSLs for its cleanrooms and related facilities. For example, some cell-culture-based Influenza vaccines are assigned BSL 2 due to its large-scale open nature, while other Influenza vaccines are assigned BSL 4 due to the virus’ highly virulent nature [110]. According to the BSL assigned, the amount of safety controls implemented will differ, such as the compulsory use of different personal protective equipment (PPE) or specific training required. Table 4 shows a summary of the different BSL and the respective considerations required.
Airlocks and airflow hoods are necessary to ensure unidirectional air flow and to maintain sterility of the environment. The walls of the facility have to be specially designed and environmental monitoring is mandatory. The facility also needs to be kept at the optimal temperature for manufacture by employing heating, ventilation and air conditioning (HVAC) systems [112]. The workers in the manufacturing facility are also regulated. They need to be dressed in the appropriate PPE and undergo necessary training. The complex nature of vaccine processing and handling necessitate timely inspections of the vaccine facilities and its procedures based on WHO or other international GMP standards.
Additionally, the International Organization for Standardization (ISO) has developed a harmonized standard, namely the ISO classification for cleanrooms and controlled areas, as a standardization of quality assurance across industries, including health care. It is widely used in many countries including the US and European Union (EU) where vaccine cleanrooms are subjected to ISO classifications [113] according to its particulate content as shown in Table 5. Each cleanroom used for handling vaccines has a specific ISO class which determines its respective controls. There are 9 ISO classes according to the particulate level in the air. In cleanrooms, classes 5 to 8 are the most commonly required and their characteristics are summarised in Table 5. Lower ISO classes have more stringent requirements. For example, the areas have a lower maximum concentration of particulates and hence require higher rates of airflow to maintain the air quality [115].
Challenges, safety and quality issues and possible solutions
Despite stringent regulation and post-market surveillance of the vaccine industry, challenges in the manufacture of safe, efficacious and good quality vaccines still prevail.
Currently, there is no single harmonized regulatory system that defines the standards for manufacture, storage and distribution of vaccines across the world, resulting in subjectivity of controls [101]. Scientifically, vaccines have been defined to include biological preparations which are administered to confer immunity against specific diseases [24, 25]. Without a standardized universal definition of vaccines, there are differences in how regulatory authorities and manufacturers emphasize control on vaccines. For instance, RAs tend to focus on safe vaccines and need for vaccines to contain adjuvants that would ensure its quality [116-119]. On the other hand, manufacturers may tend to emphasize on the overall efficacy of vaccines in reducing disease rate or severity [120]. This variation in emphasis may pose complications and challenges in the regulations and manufacture of vaccines.
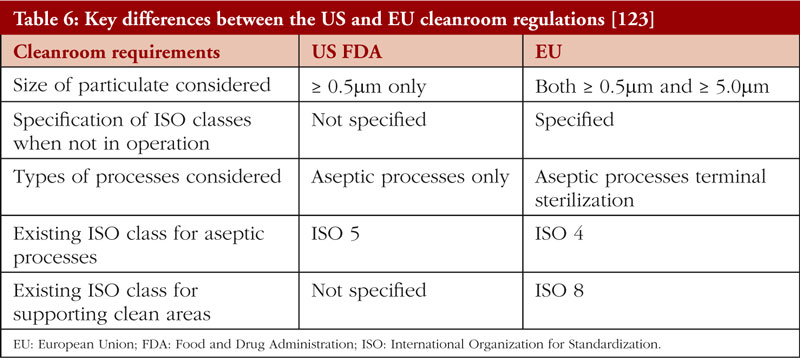
The lack of a standardised or internationally harmonized framework can lead to differing frameworks and therefore variations in terms of regulatory control. For example, currently, the two dominant regulatory frameworks are the EU GMP Guidance Annex 1: Manufacturing of Sterile Medicinal Products [121], and the US FDA Guidance for Industry: Sterile Drug Products Produced by Aseptic Processing Current Good Manufacturing Practice [122]. Some key differences in the US and EU regulations are summarised in Table 6.
In comparison with US regulations, the EU GMP Guidance has a larger and more stringent scope for cleanroom requirements [124]. Additionally, the EU has set different requirements for cleanrooms during and after operation, respectively. On the other hand, the US FDA has no specifications for cleanroom particulate levels when manufacturing processes are not ongoing. While the EU has specified that cleanroom requirements apply to both aseptic processes and terminal sterilization, the US FDA Guidance specifies only aseptic processes, and does not mention terminal sterilization. Although Annex 1 of the EU GMP Guidance has recently been revised in 2020 to widen its scope [125], the harmonization of standards between the two major jurisdictions remains a more desirable solution.
In 2021, a US vaccine manufacturing plant by the name of Emergent BioSolutions (EBS) had its production operations suspended due to contamination of its vaccine products. The single facility was used concurrently for the manufacture of two different COVID-19 vaccines, one by Johnson & Johnson (JNJ) and the other by AstraZeneca (AZ), leading to a mix-up of distinct starting materials required for each vaccine [126]. JNJ is a company based in the US while AZ is a company based in Belgium; however, both vaccines are manufactured in the same US facility, namely EBS. Although both vaccines involved are novel vaccines using similar viral vectors, the respective vectors used were non-identical and incompatible. It does not help that there are differing cross-jurisdictional regulatory requirements between the US and Belgium.
Additionally, it was discovered that the facility at EBS had previously been found to have a substandard documentation of procedures and training of staff involved [127]. While existing regulations are in place, subsequent review of these regulations and their enforcement are equally crucial. This unfortunate event also points to a need for global convergence and harmonization of international standards for vaccine manufacture and regulation.
Currently, many developing countries still lack a functioning framework for regulating vaccines. In fact, as of 2020, a significant 73% of WHO Member States do not have a mature system for optimal regulation of vaccines [128]. Developing countries also face additional challenges in maintaining the quality of vaccines due to the lack of funding and resources. To be approved as a functional NRA by WHO, the regulatory body must be able to perform regulatory actions such as assuring standards for vaccine licensure and conducting regular inspections of facilities, at least a maturity level of 3 and above [103]. This includes having a national laboratory solely for testing and evaluating the efficacy of vaccine in the country. This poses a challenge to developing countries which are already experiencing a strain on their overall regulatory resources.
With the COVID-19 pandemic driving the need for safer and more efficacious vaccines, new types of vaccines are expected to emerge in the near future. With this evolution, there are also challenges that are bound to arise in the manufacture and quality assurance of both traditional and novel vaccines. The manufacture of traditional whole vaccines is labour and time intensive, which poses the risk of pathogenic shift or drift as the vaccines undergo manufacture. Also, subunit vaccines face the challenge of thorough purification as they contain antigens of relatively smaller sizes. This can limit the degree of purification possible and make it harder for the manufacture of safe vaccines.
Likewise, mRNA vaccines face specific challenges to their novelty. Since most novel vaccines are relatively new, there is still lack of optimization at many stages of their manufacture, which may compromise the quality of vaccines manufactured. The complexity of mRNA vaccines also adds to the challenge of requiring more intricate quality assurance systems that are able to assure the vaccine’s quality at every stage of manufacture [80].
The recent rise in adverse events globally due to the use of poor-quality vaccines suggests the possible inadequacy of current regulatory frameworks and presents opportunity for refinement. In 2013, a batch of Gardasil HPV vaccine was recalled due to contamination with the vaccine glass shards [129], suggesting poor GMP compliance and inadequate enforcement of regulations.
WHO has shown much effort in harmonizing regulatory frameworks with regular review of regulatory guidelines. However, international harmonization of vaccine regulations is the way forward as this would allow cross-border use of all vaccines approved according to such an internationally harmonized regulatory framework for vaccines.
Conclusion
Challenges in the manufacture, storage, distribution and supply chain management, and associated regulation of vaccines are expected to continue. Vaccines have become a crucial weapon in the global war against pandemics. Our reliance on vaccines during the COVID-19 pandemic has clearly illustrated the critical importance of vaccines in the war against this elusive infection. The COVID-19 pandemic has also highlighted the importance of both the industry and the authority to work closely together to assure that safe, good quality and efficacious products are available at vaccination centres and points of use that are located at each and every nook and corner of the world. In a more positive light, the COVID-19 pandemic has presented opportunities for collaboration amongst NRAs, IOs and the industry in vaccine manufacture, storage, distribution, handling, regulation and international convergence of standards. Globally, NRAs should strive towards an internationally harmonized regulatory framework that will facilitate the approval and use of vaccines, whether traditional or novel, across national borders.
Competing interests: None
Provenance and peer review: Not commissioned; externally peer reviewed.
Authors
Adjunct Associate Professor Sia Chong Hocka 1, BSc (Pharm), MSc
Adelia Pheha 1, BSc (Pharm) (Hons)
Vimal Sachdeva2, MSc
Associate Professor Chan Lai Wah1, BSc (Pharm) (Hons), PhD
1Department of Pharmacy, National University of Singapore, 18 Science Drive 4, Singapore 117543
2Technical Officer (Senior GMP Inspector), WHO/HQ/MHP/ RPQ/PQT/INS, 20 Avenue Appia, Geneva CH-1211, Switzerland
References
1. World Health Organization. Immunization and vaccine-preventable communicable diseases [homepage on the Internet]. [cited 2022 Feb 11]. Available from: https://www.who.int/data/gho/data/themes/immunization
2. World Health Organization. Immunization coverage. 2021 [homepage on the Internet]. [cited 2022 Feb 11]. Available from: https://www.who.int/news-room/fact-sheets/detail/immunization-coverage
3. World Health Organization. Regional Office for Europe. Types of seasonal influenza vaccine [homepage on the Internet]. [cited 2022 Feb 11]. Available from: https://www.euro.who.int/en/health-topics/communicable-diseases/influenza/vaccination/types-of-seasonal-influenza-vaccine
4. World Health Organization. Coronavirus disease (COVID-19): vaccines. 2022 [homepage on the Internet]. [cited 2022 Feb 11]. Available from: https://www.who.int/news-room/q-a-detail/coronavirus-disease-(covid-19)-vaccines
5. Plotkin SA. Vaccines: past, present and future. Nat Med. 2005;11(4 Suppl):S5-11.
6. World Health Organization. Strategic Advisory group of Experts on Immunization. The Global Vaccine Action Plan 2011–2020. Review and lessons learned. 2019 [cited 2022 Feb 11]. Available from: https://apps.who.int/iris/bitstream/handle/10665/329097/WHO-IVB-19.07-eng.pdf
7. Taylor K, Nguyen A, Stéphenne J. The need for new vaccines. Vaccine. 2009;27 Suppl 6:G3-8.
8. Girard MP. [Vaccines for the future]. Ann Pharm Fr. 2009;67(3):203-12.
9. Kanesa-thasan N, Shaw A, Stoddard JJ, Vernon TM. Ensuring the optimal safety of licensed vaccines: a perspective of the vaccine research, development, and manufacturing companies. Pediatrics. 2011;127 Suppl 1:S16-22.
10. Greenwood B. The contribution of vaccination to global health: past, present and future. Philos Trans R Soc Lond B Biol Sci. 2014;369(1645):20130433.
11. Global routine vaccination coverage, 2009. MMWR Morb Mortal Wkly Rep. 2010;59(42):1367-71.
12. Rümke HC. [BCG: an almost 100-year-old vaccine]. Ned Tijdschr Geneeskd. 2020;164:D5146.
13. World Health Organization. Regional Office for Europe. EPHO5: Disease prevention, including early detection of illness [homepage on the Internet]. [cited 2022 Feb 11]. Available from: https://www.euro.who.int/en/health-topics/Health-systems/public-health-services/policy/the-10-essential-public-health-operations/epho5-disease-prevention,-including-early-detection-of-illness2
14. Moro PL, Haber P, McNeil MM. Challenges in evaluating post-licensure vaccine safety: observations from the Centers for Disease Control and Prevention. Expert Rev Vaccines. 2019;18(10):1091-101.
15. Children’s Hospital of Philadelphia. Vaccine safety: are vaccines safe? [homepage on the Internet]. [cited 2022 Feb 11]. Available from: https://www.chop.edu/centers-programs/vaccine-education-center/vaccine-safety/are-vaccines-safe
16. Petrović V, Seguljev Z, Gajin B. Maintaining the cold chain for vaccines. Med Pregl. 2005;58(7-8):333-41.
17. Purssell E. Reviewing the importance of the cold chain in the distribution of vaccines. Br J Community Nurs. 2015;20(10):481-6.
18. Guidelines for maintaining and managing the vaccine cold chain. MMWR Morb Mortal Wkly Rep. 2003;52(42):1023-5.
19. Casto DT, Brunell PA. Safe handling of vaccines. Pediatrics. 1991;87(1):108-12.
20. Kochhar S. Scientific challenges and opportunities in developing novel vaccines for the emerging and developing markets: New Technologies in Emerging Markets, October 16th-18th 2012, World Vaccine Congress, Lyon. Hum Vaccin Immunother. 2013;9(4):928-31.
21. Gupta SS, Nair GB, Arora NK, Ganguly NK. Vaccine development and deployment: opportunities and challenges in India. Vaccine. 2013;31 Suppl 2:B43-53.
22. Hoft DF, Brusic V, Sakala IG. Optimizing vaccine development. Cell Microbiol. 2011;13(7):934-42.
23. Maslow JN. Challenges and solutions in the development of vaccines against emerging and neglected infectious diseases. Hum Vaccin Immunother. 2019;15(10):2230-4.
24. Ginglen JG, Doyle MQ. Immunization. StatPearls. Treasure Island (FL): StatPearls Publishing Copyright © 2021, StatPearls Publishing LLC.; 2021.
25. Czochor J, Turchick A. Introduction. Vaccines. Yale J Biol Med. 2014;87(4):401-2.
26. Baxter D. Active and passive immunity, vaccine types, excipients and licensing. Occup Med (Lond). 2007;57(8):552-6.
27. Kang SM, Compans RW. Host responses from innate to adaptive immunity after vaccination: molecular and cellular events. Mol Cells. 2009;27(1):5-14.
28. U.S. Department of Health & Human Services. Vaccine types [homepage on the Internet]. [cited 2022 Feb 11]. Available from: https://www.hhs.gov/immunization/basics/types/index.html
29. News Medical. What are the different types of vaccines? [homepage on the Internet]. [cited 2022 Feb 11]. Available from: https://www.news-medical.net/health/What-are-the-Different-Types-of-Vaccines.aspx
30. The Immunisation Advisory Centre. Types of vaccines [homepage on the Internet]. [cited 2022 Feb 11]. Available from: https://www.immune.org.nz/vaccines/vaccine-development/types-vaccines.
31. Military Health System (MHS). Types of vaccines [homepage on the Internet]. [cited 2022 Feb 11]. Available from: https://www.health.mil/Military-Health- Topics/Health-Readiness/Immunization-Healthcare/Clinical-Consultation- Services/Types-of-Vaccines
32. Watson BM, Laufer DS, Kuter BJ, Staehle B, White CJ, Starr SE. Safety and immunogenicity of a combined live attenuated measles, mumps, rubella, and varicella vaccine (MMR(II)V) in healthy children. J Infect Dis. 1996;173(3):731-4.
33. White SJ, Boldt KL, Holditch SJ, Poland GA, Jacobson RM. Measles, mumps, and rubella. Clin Obstet Gynecol. 2012;55(2):550-9.
34. Roth GA, Fee E. Images of health. Smallpox: the first vaccine. Am J Public Health. 2011;101(7):1217.
35. Gershon AA. Live-attenuated varicella vaccine. Infect Dis Clin North Am. 2001;15(1):65-81, viii.
36. Jones T. Varivax (Merck & Co). Curr Opin Investig Drugs. 2002;3(1):54-7.
37. Heinsbroek E, Ruitenberg EJ. The global introduction of inactivated polio vaccine can circumvent the oral polio vaccine paradox. Vaccine. 2010;28(22):3778-83.
38. Gröschel DH, Hornick RB. Who introduced typhoid vaccination: Almroth Write or Richard Pfeiffer? Rev Infect Dis. 1981;3(6):1251-4.
39. Plotkin SA. Rabies vaccine prepared in human cell cultures: progress and perspectives. Rev Infect Dis. 1980;2(3):433-48.
40. Jarząb A, Skowicki M, Witkowska D. [Subunit vaccines–antigens, carriers, conjugation methods and the role of adjuvants]. Postepy Hig Med Dosw (Online). 2013;67:1128-43.
41. Sanofi Pasteur Inc. Adacel® (Tdap vaccine) package insert. 2017.
42. Centers for Disease Control and Prevention. The Pink Book – Diphtheria. 2021 [homepage on the Internet]. [cited 2022 Feb 11]. Available from: https://www.cdc.gov/vaccines/pubs/pinkbook/dip.html#:~:text=Diphtheria%20toxoid%20was%20developed%20in,routinely%20used%20in%20the%201940s
43. Adkins JC, Wagstaff AJ. Recombinant hepatitis B vaccine: a review of its immunogenicity and protective efficacy against hepatitis B. BioDrugs. 1998;10(2): 137-58.
44. Pomfret TC, Gagnon JM, Jr., Gilchrist AT. Quadrivalent human papillomavirus (HPV) vaccine: a review of safety, efficacy, and pharmacoeconomics. J Clin Pharm Ther. 2011;36(1):1-9.
45. Institute of Medicine Committee to Assess the safety, efficacy of the anthrax V. In: Joellenbeck LM, Zwanziger LL, Durch JS, Strom BL, editors. The anthrax vaccine: is it safe? Does It work? Washington (DC): National Academies Press (US) Copyright 2002 by the National Academy of Sciences.
46. Miyazaki C. [Hib vaccine]. Nihon Rinsho. 2008;66(10):1985-9.
47. Goldblatt D. Conjugate vaccines. Clin Exp Immunol. 2000;119(1):1-3.
48. Ledwith M. Pneumococcal conjugate vaccine. Curr Opin Pediatr. 2001;13(1):70-4.
49. Pizza M, Bekkat-Berkani R, Rappuoli R. Vaccines against meningococcal diseases. Microorganisms 2020;8(10):1521. [cited 2022 Feb 11]. Available from: https://doi.org/10.3390/microorganisms8101521.
50. Petousis-Harris H. Impact of meningococcal group B OMV vaccines, beyond their brief. Hum Vaccin Immunother. 2018;14(5):1058-63.
51. Nooraei S, Bahrulolum H, Hoseini ZS, Katalani C, Hajizade A, Easton AJ, et al. Virus-like particles: preparation, immunogenicity and their roles as nanovaccines and drug nanocarriers. Nanobiotechnology. 2021;19(1):59.
52. Deng F. Advances and challenges in enveloped virus-like particle (VLP)-based vaccines. J Immunolog Sci. 2018;2:36-41.
53. Park JW, Lagniton PNP, Liu Y, Xu RH. mRNA vaccines for COVID-19: what, why and how. Int J Biol Sci. 2021;17(6):1446-60.
54. U.S. Food and Drug Administration. FDA approves first COVID-19 vaccine 2021 [homepage on the Internet]. [cited 2022 Feb 11]. Available from: https://www.fda.gov/news-events/press-announcements/fda-approves-first- covid-19-vaccine
55. Albrecht MT, Eyles JE, Baillie LW, Keane-Myers AM. Immunogenicity and efficacy of an anthrax/plague DNA fusion vaccine in a mouse model. FEMS Immunol Med Microbiol. 2012;65(3):505-9.
56. Dolzhikova IV, Tokarskaya EA, Dzharullaeva AS, Tukhvatulin AI, Shcheblyakov DV, Voronina OL, et al. Virus-Vectored Ebola Vaccines. Acta Naturae. 2017;9(3):4-11.
57. U.S. Food and Drug Administration. First FDA-approved vaccine for the prevention of Ebola virus disease, marking a critical milestone in public health preparedness and response. 2019 [homepage on the Internet]. [cited 2022 Feb 11]. Available from: https://www.fda.gov/news-events/press-announcements/first-fda-approved-vaccine-prevention-ebola-virus-disease-marking-critical-milestone-public-health
58. Knoll MD, Wonodi C. Oxford-AstraZeneca COVID-19 vaccine efficacy. Lancet 2021;397(10269):72-4.
59. Plotkin S. History of vaccination. Proc Natl Acad Sci U S A. 2014;111(34):12283-7.
60. Wang YB, Wang LP, Li P. Perspectives on novel vaccine development. Pol J Vet Sci. 2018;21(3):643-9.
61. Minor PD. Live attenuated vaccines: historical successes and current challenges. Virology. 2015;479-480:379-92.
62. Murdin AD, Barreto L, Plotkin S. Inactivated poliovirus vaccine: past and present experience. Vaccine. 1996;14(8):735-46.
63. Carr S, Allison KJ, Van De Velde LA, Zhang K, English EY, Iverson A, et al. Safety and immunogenicity of live attenuated and inactivated influenza vaccines in children with cancer. J Infect Dis. 2011;204(10):1475-82.
64. Coelingh K, Olajide IR, MacDonald P, Yogev R. Efficacy and effectiveness of live attenuated influenza vaccine in school-age children. Expert Rev Vaccines. 2015;14(10):1331-46.
65. Vaughan AM, Wang R, Kappe SH. Genetically engineered, attenuated whole-cell vaccine approaches for malaria. Hum Vaccin. 2010;6(1):107-13.
66. Hershman-Sarafov M, Potasman I. [Immunizations in immunocompromised hosts–principles and updated recommendations]. Harefuah. 2015;154(12):786-90, 804, 803.
67. Gavi. The Vaccine Alliance. What are protein subunit vaccines and how could they be used against COVID-19? [homepage on the Internet]. [cited 2022 Feb 11]. Available from: https://www.gavi.org/vaccineswork/what-are-protein-subunit-vaccines-and-how-could-they-be-used-against-covid-19
68. Oxford Vaccine Group. Vaccine Knowledge Project. MenB Vaccine (Meningococcal B Vaccine). 2019 [homepage on the Internet]. [cited 2022 Feb 11]. Available from: https://vk.ovg.ox.ac.uk/vk/menb-vaccine
69. Infanrix Hexa, Powder and Suspension for Suspension for Injection in a Pre-Filled Syringe, Package Leaflet.
70. Oxford Vaccine Group. Vaccine Knowledge Project. 6-in-1 Vaccine. 2019 [homepage on the Internet]. [cited 2022 Feb 11]. Available from: https://vk.ovg.ox.ac.uk/vk/6-in-1-vaccine
71. Pardi N, Hogan MJ, Weissman D. Recent advances in mRNA vaccine technology. Curr Opin Immunol. 2020;65:14-20.
72. Patil SU, Shreffler WG. Novel vaccines: technology and development. J Allergy Clin Immunol. 2019;143(3):844-51.
73. Delany I, Rappuoli R, De Gregorio E. Vaccines for the 21st century. EMBO Mol Med. 2014;6(6):708-20.
74. What is a DNA Vaccine? : News Medical Net; [cited 2022 Feb 11]. Available from: https://www.news-medical.net/health/What-is-a-DNA-based-vaccine.aspx.
75. Fay EJ, Langlois RA. MicroRNA-attenuated virus vaccines. Noncoding RNA. 2018;4(4):25.
76. Maruggi G, Zhang C, Li J, Ulmer JB, Yu D. mRNA as a transformative technology for vaccine development to control infectious diseases. Mol Ther. 2019;27(4):757-72.
77. Zhang C, Maruggi G, Shan H, Li J. Advances in mRNA vaccines for infectious diseases. Front Immunol. 2019;10:594.
78. Pardi N, Hogan MJ, Porter FW, Weissman D. mRNA vaccines – a new era in vaccinology. Nat Rev Drug Discov. 2018;17(4):261-79.
79. Speck D. [Specific aspects of vaccine manufacturing]. Ann Pharm Fr. 2009;67(3): 213-8.
80. Ulmer JB, Valley U, Rappuoli R. Vaccine manufacturing: challenges and solutions. Nat Biotechnol. 2006;24(11):1377-83.
81. Gomez PL, Robinson JM. Vaccine manufacturing. Plotkin’s Vaccines. 2018:51-60.e1.
82. Tran V, Liu J, Behr MA. BCG vaccines. Microbiol Spectr. 2014;2(1):MGM2- 0028-2013.
83. Ritz N, Hanekom WA, Robins-Browne R, Britton WJ, Curtis N. Influence of BCG vaccine strain on the immune response and protection against tuberculosis. FEMS Microbiol Rev. 2008;32(5):821-41.
84. Wigginton KR, Kohn T. Virus disinfection mechanisms: the role of virus composition, structure, and function. Curr Opin Virol. 2012;2(1):84-9.
85. Abd-Elghaffar AA, Ali AE, Boseila AA, Amin MA. Inactivation of rabies virus by hydrogen peroxide. Vaccine. 2016;34(6):798-802.
86. Inagaki H, Saito A, Sugiyama H, Okabayashi T, Fujimoto S. Rapid inactivation of SARS-CoV-2 with deep-UV LED irradiation. Emerg Microbes Infect. 2020;9(1):1744-7.
87. Batavia Biosciences. Downstream process development [homepage on the Internet]. [cited 2022 Feb 11]. Available from: https://www.bataviabiosciences.com/viral-vaccines/process-development/downstream-process-development/
88. Zhao M, Vandersluis M, Stout J, Haupts U, Sanders M, Jacquemart R. Affinity chromatography for vaccines manufacturing: finally ready for prime time? Vaccine. 2019;37(36):5491-503.
89. Kino Y. [Vaccine excipients]. Nihon Rinsho. 2008;66(10):1933-7.
90. Bastola R, Noh G, Keum T, Bashyal S, Seo JE, Choi J, et al. Vaccine adjuvants: smart components to boost the immune system. Arch Pharm Res. 2017;40(11):1238-48.
91. Ascarateil S, Dupuis L. Surfactants in vaccine adjuvants: description and perspectives. Vaccine. 2006;24 Suppl 2:S2-83-5.
92. Khandke L, Yang C, Krylova K, Jansen KU, Rashidbaigi A. Preservative of choice for Prev(e)nar 13™ in a multi-dose formulation. Vaccine. 2011;29(41): 7144-53.
93. Lloyd J, Cheyne J. The origins of the vaccine cold chain and a glimpse of the future. Vaccine. 2017;35(17):2115-20.
94. Storing vaccines at the right temperatures – NuAire [cited 1 Mar 2022] Available from: https://www.nuaire.com/-/media/Project/Nuaire/Resources/infographic/vaccine-storage-temperature.pdf
95. Chen D, Tyagi A, Carpenter J, Perkins S, Sylvester D, Guy M, et al. Characterization of the freeze sensitivity of a hepatitis B vaccine. Hum Vaccin. 2009;5(1):26-32.
96. Xue H, Yang B, Kristensen DD, Chen D. A freeze-stable formulation for DTwP and DTaP vaccines. Hum Vaccin Immunother. 2014;10(12):3607-10.
97. World Health Organization. Aide-memoire for prevention of freeze damage to vaccines. 2019 [homepage on the Internet]. [cited 2022 Feb 11]. Available from: https://apps.who.int/iris/handle/10665/69673
98. Centers for Disease Control and Prevention. Pfizer-BioNTech COVID-19 vaccine storage and handling summary. 2021 [homepage on the Internet]. [cited 2022 Feb 11]. Available from: https://www.cdc.gov/vaccines/covid-19/info-by-product/pfizer/downloads/Pfizer_PED_StorageHandling_Summary.pdf
99. Pfizer. Pfizer-BioNTech COVID-19 vaccine US distribution fact sheet. 2021 [homepage on the Internet]. [cited 2022 Feb 11]. Available from: https://www.pfizer.com/news/articles/covid_19_vaccine_u_s_distribution_fact_sheet
100. Baylor NW, Marshall VB. Regulation and testing of vaccines. Vaccines. 2013:1427-46.
101. GS1. Product classification in healthcare. 2015 [homepage on the Internet]. [cited 2022 Feb 11]. Available from: https://www.gs1.org/sites/default/files/docs/healthcare/product_classification_in_healthcare.pdf
102. World Health Organization. Product eligibility under the COVAX Facility 2020 [homepage on the Internet]. [cited 2022 Feb 11]. Available from: https://extranet.who.int/pqweb/sites/default/files/documents/Product-Eligibility_COVAX-Facility_Dec2020_0.pdf
103. World Health Organization. Regulatory agencies. 2021 [homepage on the Internet]. [cited 2022 Feb 11]. Available from: https://extranet.who.int/pqweb/vaccines/regulatory-agencies
104. World Health Organization. Prequalification of medical products (IVDs, medicines, vaccines and immunization devices, vector control). 2021 [homepage on the Internet]. [cited 2022 Feb 11]. Available from: https://extranet.who.int/pqweb/vaccines/manufacturers
105. Finn TM. U.S. FDA requirements for human vaccine product safety and potency testing. Procedia in Vaccinology. 2011;5:137-40.
106. U.S. Food and Drug Administration. Biologics License Applications (BLA) Process (CBER). 2021 [homepage on the Internet]. [cited 2022 Feb 11]. Available from: https://www.fda.gov/vaccines-blood-biologics/development-approval-process-cber/biologics-license-applications-bla-process-cber
107. World Health Organization. Environmental monitoring of clean rooms in vaccine manufacturing facilities. 2012 [homepage on the Internet]. [cited 2022 Feb 11]. Available from: https://www.who.int/immunization_standards/vaccine_quality/env_monitoring_cleanrooms_final.pdf
108. World Health Organization. Good manufacturing practices [homepage on the Internet]. [cited 2022 Feb 11]. Available from: https://www.who.int/teams/health-product-and-policy-standards/standards-and-specifications/gmp
109. Cleanroom Technology. Vaccine manufacturing facilities and cleanrooms explained. 2021 [homepage on the Internet]. [cited 2022 Feb 11]. Available from: https://www.cleanroomtechnology.com/news/article_page/Vaccine_ manufacturing_facilities_and_cleanrooms_explained/176581
110. World Health Organization. WHO biosafety risk assessment and guidelines for the production and quality control of novel human influenza candidate vaccine viruses and pandemic vaccines. 2017 [homepage on the Internet]. [cited 2022 Feb 11]. Available from: https://www.who.int/biologicals/vaccines/INFLUENZA_BIOSAFETY_GL_draft_1_1st_PC_22_Nov_2017_TZ.pdf
111. Public Health Emergency. Science Safety Security. Biosafety level requirements. 2019 [homepage on the Internet]. [cited 2022 Feb 11]. Available from: https://www.phe.gov/s3/BioriskManagement/biocontainment/Pages/BSL-Requirements.aspx
112. World Health Organization. Environmental monitoring of clean rooms in vaccine manufacturing facilities. 2012 [homepage on the Internet]. [cited 2022 Feb 11]. Available from: https://www.who.int/immunization_standards/vaccine_quality/env_monitoring_cleanrooms_final.pdf
113. International Organization for Standardization. ISO 14644-1 Cleanrooms and associated controlled environment. 2015 [homepage on the Internet]. [cited 2022 Feb 11]. Available from: https://www.iso.org/obp/ui/#iso:std:iso: 14644:-1:ed-2:v1:en
114. Terra Universal. FS209E and ISO cleanroom standards [homepage on the Internet]. [cited 2022 Feb 11]. Available from: https://www.terrauniversal.com/blog/fs209e-and-iso-cleanroom-standards/
115. The Federal Standard 209E Connect 2 Cleanrooms [cited 1 Mar 2022]. Available from: https://www.connect2cleanrooms.com/knowledge-base/glossary/federal-standard-209?___store=EN&___from_store=EN
116. World Health Organization. Vaccines and immunization [homepage on the Internet]. [cited 2022 Feb 11]. Available from: https://www.who.int/health-topics/vaccines-and-immunization#tab=tab_1
117. Australian Government. Department of Health. Vaccines overview [homepage on the Internet]. [cited 2022 Feb 11]. Available from: https://www.tga.gov.au/vaccines-overview
118. European Commission. Vaccination. Overview [homepage on the Internet]. [cited 2022 Feb 11]. Available from: https://ec.europa.eu/health/vaccination/overview_en
119. Health Sciences Authority. HSA grants interim authorisation for Moderna COVID-19 vaccine in Singapore [homepage on the Internet]. [cited 2022 Feb 11]. Available from: https://www.hsa.gov.sg/announcements/press-release/hsa-grants-interim-authorisation-for-moderna-covid-19-vaccine-in-singapore
120. Pfizer. Coronavirus COVID-19 vaccine updates [homepage on the Internet]. [cited 2022 Feb 11]. Available from: https://www.pfizer.com/science/coronavirus/vaccine/rapid-progress
121. EU GMP Annex 1 Revision: Manufacture of Sterile Medicinal Products (Draft) [homepage on the Internet]. [cited 2022 Feb 11]. Available from: https://www.gmp-compliance.org/files/guidemgr/2020_annex1ps_sterile_medicinal_products_en.pdf
122. U.S. Food and Drug Administration. Guidance for Industry. Sterile drug products produced by aseptic processing current good manufacturing practice. 2004 [homepage on the Internet]. [cited 2022 Feb 11]. Available from: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/sterile-drug-products-produced-aseptic-processing-current-good-manufacturing-practice
123. Regulatory Affairs Professionals Society. FDA and EU GMP Annex 1 differences in cleanroom specifications. 2019 [homepage on the Internet]. [cited 2022 Feb 11]. Available from: https://www.raps.org/news-and-articles/news-articles/2019/7/fda-and-eu-gmp-annex-1-differences-in-cleanroom-sp
124. Grazal JG, Earl DS. EU and FDA GMP regulations: overview and comparison. Qual Assur. 1997;2:55-60.
125. Thomas F. Unlocking the key changes to annex 1. Pharm Technol Eur. 2020;32(10):42-3.
126. Johnson & Johnson COVID-19 vaccine manufacturing halted at US plant that had contamination issue CNA2021. Channel News Asia.
127. Swaine J, Rowland C. Emergent plant that ruined Johnson & Johnson vaccine doses had prior FDA violations. The New York Times. 2021 Apr 1. Available from: https://www.washingtonpost.com/business/2021/04/01/emergent-jj-vaccine- plant-inspection/
128. World Health Organization. Strengthening NRAS. 2021 [homepage on the Internet]. [cited 2022 Feb 11]. Available from: https://vaccine-safety-training.org/strengthening-nras.html
129. Centers for Disease Control and Prevention. Voluntary recall of one lot of Gardasil HPV vaccine (2013) [homepage on the Internet]. Available from: https://www.cdc.gov/vaccinesafety/concerns/history/gardasil-recall-faq.html#anchor_1595011429613
|
Author for correspondence: Adjunct Associate Professor Sia Chong Hock, BSc (Pharm), MSc, Department of Pharmacy, National University of Singapore, 18 Science Drive 4, Singapore 117543 |
Disclosure of Conflict of Interest Statement is available upon request.
Copyright © 2022 Pro Pharma Communications International
Permission granted to reproduce for personal and non-commercial use only. All other reproduction, copy or reprinting of all or part of any ‘Content’ found on this website is strictly prohibited without the prior consent of the publisher. Contact the publisher to obtain permission before redistributing.