Non-medical switching of biologicals/biosimilars: Canada, Europe and the US – a webinar report
Published on 2023/03/02
Generics and Biosimilars Initiative Journal (GaBI Journal). 2023;12(1):23-36
Author byline as per print journal: Michael S Reilly, Esq; Gail Attara; Ralph D McKibbin, MD, FACP, FACG, AGAF; Professor Philip J Schneider, MS, FASHP, FASPEN, FFIP
|
Introduction: Biosimilars are now key players in the global drugs market offering potentially more affordable treatment options with similar safety and efficacy. However, there are concerns about non-medical switching practices of originator biologicals/biosimilars in different regions. A webinar was held to discuss non-medical switching practices and to explore the importance of safeguarding the physician–patient relationship. |
Submitted: 19 December 2022; Revised: 20 January 2023; Accepted: 21 January 2023; Published online first: 3 February 2023
Introduction
In joint effort with the Alliance for Safe Biologic Medicines (ASBM), the Generics and Biosimilars Initiative (GaBI) organized and hosted the second in a series of webinars on biosimilars entitled ‘Non-medical switching of biologicals/biosimilars: Canada, Europe and the US’. This webinar set out to give an overview of switching, substitution and interchangeability practices of originator biologicals/biosimilars in different regions and to explore the importance of safeguarding the physician–patient relationship.
Biologicals offer significant benefits and enhanced quality of life for patients with chronic diseases, such as rheumatoid arthritis, multiple sclerosis, inflammatory bowel disease and cancer. However, finding a working treatment regimen may take a long time and can involve trial and error for each individual. Biosimilars of originator biological drugs have been on the market for 16 years since their first launch in 2006 in Europe. At the time of the webinar, there were 84 approvals in the EU [1] and 34 in the US [2].
In many cases, biosimilar drugs can offer a more affordable treatment option with similar safety and efficacy. However, it is argued that government policies that force biosimilar switching for non-medical (non-medical switching refers to the switching of a patient’s medicine, often at the behest of a third party, for reasons other than the patient’s health and safety) and strictly economic reasons may have potential to ultimately affect patients’ treatment and their well-being, create mistrust in the healthcare system, undermine competition, increase total healthcare costs, and act as a barrier to innovation and future access to new biological drugs for patients.
Non-medical switching should consider two governing principles: 1) the physician–patient relationship must be protected to promote the best outcomes; and 2) patients stabilized on a biological therapy should not be forced to undergo a non-medical switch.
The webinar, and this follow-up article, discuss the non-medical switching practices in Canada, Europe and the US. The expert panelists provided a detailed overview of policies in the different regions and discussed the merits and concerns about non-medical switching practices. They also explored the importance of safeguarding the physician–patient relationship, through education and knowledge transfer.
Methods
In this online event, held on 20 July 2022 [3], the contributors discussed non-medical switching practices of biologicals, including biosimilars, in different regions and explored the importance of safeguarding the physician–patient relationship. The discussion highlighted that improved information and protecting the physician–patient relationship in medical switching will encourage smooth uptake of biosimilars.
The webinar was moderated by Dr Steven Stranne, JD, and partner at Foley Hoag LLP, who has a unique perspective as a physician and lawyer. Presentations were given by: Mr Michael S Reilly, Esq, Executive Director of the ASBM; Ms Gail Attara, President and CEO of the Gastrointestinal Society, Canada; Dr Ralph D McKibbin, Past President of the Pennsylvania Society of Gastroenterology and the Digestive Disease National Coalition; Dr Leah Christl, Executive Director, Global Biosimilars Regulatory Affairs and Regulatory and R&D Policy, Amgen; Dr Durhane Wong-Rieger, President and CEO of the Canadian Organization for Rare Disorders; and Professor Philip J Schneider, Professor of the Ohio State University College of Pharmacy. The presenters were also joined by two panelists, Ms Jaymee Maaghop, Outreach Coordinator at the Gastrointestinal Society, Canada, and Ms Urvashi Rathod, Project Officer at the International Federation on Ageing, Canada.
Overall, the panel was made up of academic clinicians patient advocates, industry specialist, policymakers and regulatory experts from Canada and the US. They shared their experience and knowledge of medical switching and principles and guidance on interchangeability of originator biologicals/biosimilars, highlighting specific concerns about, and the importance of a multi-stakeholder approach to protecting the physician–patient relationship in the practice of non-medical switching of originator biologicals/biosimilars.
Learning objectives
The overall learning objectives of the webinar were outlined as follows:
- To gain an insight on practice of switching originator biologicals/biosimilars in Europe and the US
- To learn the current state of non-medical switching policies of originator biologicals/biosimilars in Canada
- To hear the physicians’ concerns and experience of non-medical switching of originator biologicals/biosimilars
- To understand the principles and guidance of interchangeability of originator biologicals/biosimilars in the US
- To recognize patients’ concerns in non-medical switching of originator biologicals/biosimilars
- To evaluate the role of healthcare providers (physicians, health policymakers, pharmacists, nurses) and patients in non-medical switching of originator biologicals/biosimilars
- To identify the important elements and educational needs on non-medical switching of originator biologicals/biosimilars practices
- To produce and publish a meeting report in GaBI Journal covering recommen dations and informational programmes on non-medical switching policies of originator biologicals/biosimilars.
Results
Expert speaker presentations
There were a variety of expert speaker presentations followed by Q&A and an in-depth panel discussion. The presentations are downloadable [3].
Overview of originator biologicals/biosimilars switching policy in Europe and the US
Mr Michael Reilly noted that the first webinar in the series [4] examined several factors which led to the highly successful biosimilar programmes in Europe and the US. Europe is widely acknowledged as a global leader in biosimilars for successfully developing a robust and sustainable biosimilars programme. Many countries seek to emulate Europe’s success and ‘catch-up’.
White paper recommendations
The white paper entitled ‘Policy Recommendations for a Sustainable Biosimilars Market: Lessons from Europe’ [5] identifies several ‘Must-Have’ principles, critical for countries to achieve biosimilar success:
- Physicians should have the freedom to choose between off-patent originator biologicals and available biosimilars and to act in the best interest of their patients based on scientific evidence and clinical experience.
- Tenders should be designed to include multiple value-based criteria beyond price, e.g. education, services, available dose strengths, and provide a sufficient broad choice (multi-winner tenders versus single-winner tenders) to ensure continuity of supply and healthy competition.
- A level playing field between all participating manufacturers is the best way to foster competition; mandatory discounts which place artificial downward pressure on manufacturers do not engender a sustainable market environment.
Mr Reilly emphasized that Europe’s collaborative patient-based approach is key to the success of biosimilars. There are regular multi-stakeholder consultations held by the European Commission in Brussels and these consultations are transparent, open and inclusive. Education of patients to build trust in biosimilars has been a priority, as has establishing patient buy-in on biosimilar policy.
European physician survey and non-medical switching
According to survey findings presented at the European Society of Medical Oncology 2019 Congress in Barcelona, Spain, 27 September to 1 October 2019 [6], European physicians oppose non-medical switching. The survey highlighted that those European physicians took great pride in their approach, which is very patient focused. Notably, despite having overwhelming confidence in biosimilars, they are strongly opposed (73%) to non-medical switching. In addition, in nearly every European country, physicians and patients choose freely among multiple approved products (originator and several biosimilars) and all are reimbursed. Many countries encourage physicians to prescribe the cheapest medicine, typically a biosimilar, to new (bio-naïve) patients. Even in Norway with a national tendering system, physicians retain prescription choice among all available products [7].
Mr Reilly explained that non-medical switching is permitted in some Eastern European countries although patients and physicians do retain some autonomy:
- Estonia: Permitted, patient can refuse and pay price difference out-of-pocket.
- Latvia: Non-bio-naïve patients can refuse and pay cost difference; the physician can prevent substitution. Others must use cheapest product.
- Poland: Permitted, pharmacists are to discuss with patient.
Denmark’s unique approach
Denmark has a unique approach to non-medical switching. The country has adopted a ‘winner takes all’ tendering process under which, following a transparent process, only the winning product will be reimbursed, and patients are not given a choice regarding a switch. Although the approach may seem radical, it is facilitated by the fact that Denmark has a small population (5 million versus 38 million in Canada, where forced switching policies have been adopted, see later section on ‘Current state of non-medical switching policies of originator biologicals/biosimilars in Canada’) that is ethnically homogenous (98% White), while Canada is more ethnically diverse. It is also the only country in Europe that has every patient covered by Digitized Medical Records, which Canada does not have.
Biosimilars in the US
The US is now catching up with Europe in terms of the approval of biosimilars [8]. At the time of the webinar the US had approved two interchangeable biosimilars (meaning they can be automatically substituted at the pharmacy level subject to state laws [7]). In addition, there could soon be upwards of five biosimilars available for some originators in the US. This increased competition results in patients having access to more affordable alternative treatments.
Mr Reilly advised that in the US there is no government policy mandating non-medical switching. As in Europe, the thriving markets, high uptake rates (and savings) are due to many products competing for market share. In addition, all 50 States permit automatic substitution for ‘interchangeable’ biosimilars (those for which additional data, including switching studies have been gathered) [7].
Canada’s forced non-medical switch ing policy
In contrast to Europe and the US, several jurisdictions in Canada have implemented non-medical forced switching policies.
Mr Reilly noted that these have shown to [9]:
- Needlessly exclude physicians and patients from the treatment-decision process
- Contribute to Canada lagging behind Europe and the US in terms of biosimilars use
- Prioritize short-term savings over long-term growth and sustainability.
Canadian patient groups have been strongly opposed to these policies and vocal about them. In addition, the Canadian Association of Gastroenterologists issued statements opposing the forced-switching policies in place in Alberta and British Columbia. The Quebec heath technology assessment body, (Institut National d’excellence en Santé et en Services Sociaux, INESSS), published a report, Safety of switching biologics and their interchangeability, in May 2020 that says, ‘Non-medical switching in patients being treated with a reference biologic is generally not accepted by learned societies and the consulted clinicians’ [10].
Further presentations from patient organizations examine the Canadian patient experience of biosimilars. These organizations, the Canadian Organization for Rare Diseases and the Gastrointestinal Society, are part of a biosimilars working group, which includes ASBM, that is bringing the patient perspective to biosimilars policy discussions.
Current state of non-medical switching policies of originator biologicals/biosimilars in Canada
This presentation was given by Ms Gail Attara, Gastrointestinal Society, Canada via a pre-recorded video.
Ms Attara gave a brief overview of the healthcare system in Canada, noting that there is universal medical healthcare for physician visits and hospital use but not for drugs, adding that there is tightly managed, variable by jurisdiction, limited publicly funded drug coverage. The downside to this is that, when a biological is changed, this usually leads to changes in the patient’s healthcare team.
Switching and substitution
The Gastrointestinal Society in Canada has been active in the field of biosimilars since 2013 and has produced patient information pamphlets and a video [11]. These contain information defining:
- Medical Switch: Medication is changed by a physician for reasons related to efficacy or tolerability.
- Non-Medical Switch: Medication is chan ged, with or without physician consent, for reasons other than clinical necessity. May be driven by cost or convenience.
Regarding substitution, a pharmacist:
- May substitute a biosimilar for its reference product.
- Is subject to local (provincial) pharmacy laws and regulations.
- Carries out ‘automatic’ or ‘involuntary’ substitution if performed without physician’s involvement.
2015 IBD patients Survey
In 2015, the Gastrointestinal Society hosted a survey to help understand inflammatory bowel disease (IBD) patients’ opinions and outlooks regarding biosimilars (then called subsequent entry biologicals, or SEBs in Canada). There were 354 respondents. The results were presented at the European Crohn’s and Colitis Organization (ECCO) conference in Amsterdam in 2015 [12].
The IBD patients survey [n = 354] revealed that:
- 95% of respondents thought it was very (78%) or somewhat (17%) important that the physician has the sole authority to decide, together with the patient, the most suitable biological medicine to use to treat their disease.
- 94% of respondents thought it was very (75%) or somewhat (19%) important concerning if the pharmacist or government/private insurance plan made the determination which biological (innovator or biosimilar) to dispense to a patient on initiation of treatment.
- 96% of respondents thought it was very or somewhat concerning if the pharmacist, government drug plan, or private insurance plan made the determination of which biological (innovator or biosimilar) to dispense to a patient during treatment, including maintenance therapy (switch medicines without telling a patient).
Biosimilars Working Group Canada
The Biosimilars Working Group was set up in Canada to ensure patient outcomes and considerations are at the centre of health policy discussions regarding decisions over biological treatment. The Working Group aims to create evidence-based educational materials for patients and healthcare professionals, which are hosted at www.biosimilaroptions.ca. Members of the working group include:
- Alliance for Safe Biologic Medicines
- Canadian Council of the Blind
- Canadian Organization for Rare Disorders
- Canadian Society of Intestinal Research
- Crohn’s and Colitis Canada
- Gastrointestinal Society
- HS (hidradenitis suppurativa) Heroes
- International Federation on Ageing
- MedAccess BC
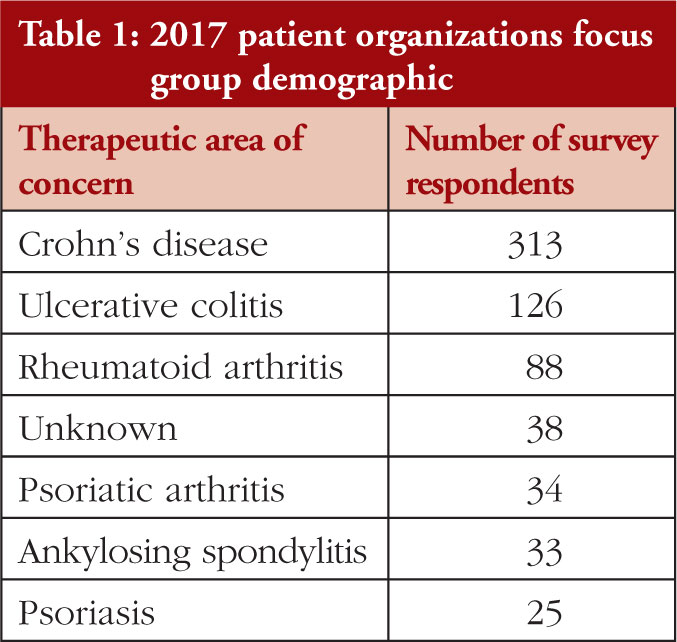
2017 Patient Organizations Focus Group
In 2017, five patient organizations across therapeutic areas came together to conduct several cross-country focus groups [13]. This included:
- Canadian Arthritis Patient Alliance
- Canadian Psoriasis Network
- Crohn’s and Colitis Canada
- Gastrointestinal Society
- The Arthritis Society (project lead)
The survey demographic is demonstrated in Table 1.
When discussing forced switching, the key findings with regards to patients were:
- many patients experience a high degree of emotional impact from disease, treatment decision process, delays
- this was the first time for biological switching in Canada and ‘trial-and-error’ approach to treatments amplified anxiety
- overall, patients have a poor understanding of biosimilars, coupled with poor understanding of healthcare system in general
- patients are opposed to forced non-medical switching
- patients recognize role of biosimilars for new patients
- patients are anxious for more clinical studies regarding impact of switching
- patients are concerned that switching would reopen access and coverage questions.
The Canadian perspective
Health Canada’s definitions related to interchangeability and switching of biological [14] are as follows:
- Interchangeability: ‘Health Canada’s auth orization of a biosimilar is not a declaration of equivalence to the reference biologic drug. The authority to declare two products as interchangeable rests with each province and territory according to its own rules and regulations’.
- Switching: ‘Patients and healthcare providers can have confidence that biosimilars are effective and safe for each of their authorized indications. No differences are expected in efficacy and safety following a change in routine use between a biosimilar and its reference biological drug in an authorized indication’.
There were also various statements released related to switching that were highlighted by national health technology bodies, including Heath Canada:
- In 2016: ‘Health Canada recommends that a decision to switch a patient being treated with a reference biologic drug to a biosimilar should be made by the treating physician in consultation with the patient and taking into account available clinical evidence and any policies of the relevant jurisdiction’.
- In 2019, Canada’s Drug and Health Technology Agency (CADTH): ‘Decisions about switching are generally made by individual patients and their practitioners based on the available clinical evidence,’ [15].
- In 2020, INESSS, (Quebec only): ‘This report identifies certain populations or biologics for which very little or no data are available regarding the safety of biologics switch, and the significant concerns that clinicians have about non-medical switching’, [16].
The Canadian Association of Gastroenterology stated that it does not recommend non-medical switching from originator to biosimilar in patients with stable disease and are doing well on the originator and does not recommend automatic substitution [17].
The Canadian Rheumatology Association advised that there must be [17]:
- ‘respectful and informed conversation between the rheumatologist and patient prior to any transitioning/changing from an originator biological to a biosimilar’
- ‘no substitution without an informed consultation by the patient with the prescribing rheumatologist prior to any treatment change’.
Ms Attara stressed that these statements are all in contrast to the non-medical forced-switching policies.
Canada’s non-medical switching policies
In Canada, non-medical switching policies had been adopted in a few provinces and one territory in Canada, see Table 2.
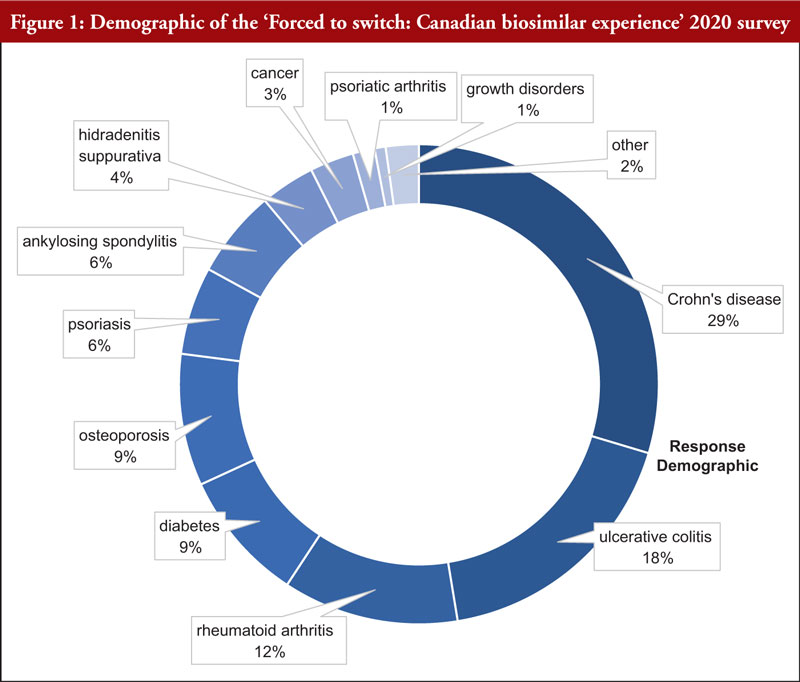
Forced to switch survey 2020 Canada
Ms Attara then presented the results from the ‘Forced to Switch: Canadian Biosimilar Experience’ 2020 survey [18]. Figure 1 shows the demographic of respondents of the survey in terms of disease.
The survey gathered 145 responses bet ween 13 November 2019 and 6 February 2020. A number of issues were uncovered and 53 of these were classified as:
- 18 delays with biosimilar dose, required additional Remicade® dose
- 24 lack of communication from biosimilar patient support programme
- 9 had no biosimilar dose available at scheduled infusion date
- 2 lack of coverage issues for biosimilar.
Overall issues with the policies had implications for:
Mental health
- 66% of online survey respondents experience mental health related conditions, e.g. insomnia, anxiety disorders, mood disorders
- majority of phone and email participants said they were stressed, anxious, depressed, and worried due to non-medical switch
- beginning of COVID-19 pandemic.
Timing and consultation
- delays in communications, accidental refills for originator biological, administrative backlog
- lack of appropriate notice to switch.
It was also evident that there was a lack of biosimilar education regarding forced-switching and the reasoning behind this.
In addition, there were also issues related to:
Patient support programmes
- exclusivity contracts (limited to 2019–2020)
- accessibility (capacity and availability, geographic location).
Special authority
- high request denial rates regarding physician requests for patients to remain on their treatment regimen: Alberta: 0.25% approved, British Columbia: 1% approved.
No exceptions for
- British Columbia’s non-medical switch
- some children with temporary special authority until November 2020
- Alberta: non-medical switch excluded paediatrics and pregnant women.
In conclusion, most survey respondents were familiar with biological medications. Compared to other inflammatory conditions, inflammatory bowel disease sufferers (Crohn’s disease and ulcerative colitis) only have a few options with varied mechanisms of action. Here, efficacy, safety and doctor’s recommendation are the top three factors for selecting biosimilar treatment. Overall, patients maintain that they, along with their physician, should have the sole authority to decide the best biological medicine for their disease.
Biosimilars in Alberta and BC survey 2021
The 2021 ‘Biosimilars in Alberta and BC’ survey results were discussed [19]. Direct quotes from respondents included comments such as: ‘positive, so far, so good, and same as original’, ‘first injection didn’t sting like the originator biologic, which was a nice change’, and ‘the biosimilar injection hurts more than the biologic’. The side effects reported ranged from moderate to severe, and included diarrhoea, headache, nausea, fatigue, respiratory issues, and skin and pouch infections; and there were varied experiences from person to person.
Overall, it was too soon to tell regarding the non-medical switch policies related to Humira® adalimumab biosimilars at the time of the survey. The majority of respondents were not confident in their ongoing care and felt they were not given enough time to switch.
Alberta extended their switching deadline during the pandemic. In both provinces, most respondents had extra communications with their healthcare team to facilitate the switch, including telephone calls (57%), in-person appointments (31%), virtual appointments (20%), and email communications (20%). However, there were difficulties with scheduling appointments, requiring additional testing, and challenges with coordinating the biosimilar product in a different healthcare setting, e.g. from infusion clinic to in-hospital care. All in all, inadequate coordination was reported, and this was confounded by COVID-19.
Gastrointestinal Society Canada
Ms Attara stressed the position of the Gastrointestinal Society Canada that a fair and equitable biosimilars policy is to set one price for reimbursement for all originator biologicals and their respective biosimilars. Originator companies could lower their prices to compete and thereby governments would not need to force patients to switch their medications. The Society believes that this is the most viable option and encourages all Canadian jurisdictions to implement such a policy so that patients do not have needless suffering. This ‘lowest cost alternative’ form of pricing policy is widespread in Canada for brand-name and generic drug products and gives governments the cost-savings they desire. Regrettably, Canadian jurisdictions will not adopt this. In view of this government hardline stance, the Gastrointestinal Society developed three documents that include minimal exception criteria that jurisdictions can adopt. These exceptions include a longer timeframe for switching children and elderly patients, those with severe disease and high-risk patients, and pregnant patients.
US physicians’ concerns/experience of non-medical switching of biologicals/biosimilars
Dr Ralph D McKibbin, gastroenterologist, outlined the results of a 2021 ASBM survey of US physicians [20]. The survey examined:
- Knowledge about biologicals and biosimilars/approval process
- Confidence in biosimilars: their safety and efficacy
- Substitution and switching: when and how? Who decides? What data should be required?
- Product identification: how best to differentiate between a biological and its various biosimilars
- Reimbursement policies: which products should be covered and why?
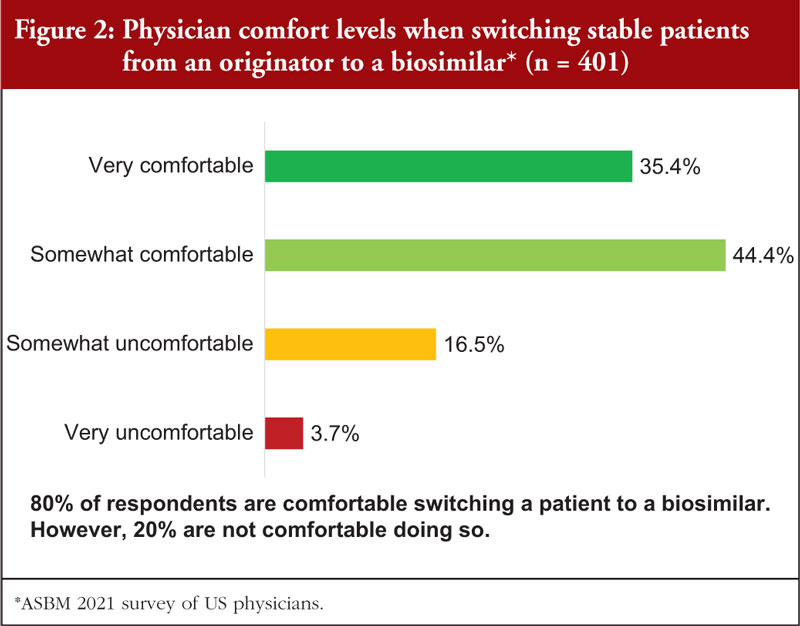
Overall, the survey highlighted that physician confidence in and comfort with biosimilars is high and the vast majority (~90%) of physicians have no concerns with prescribing biosimilars to new patients. However, they are less comfortable with non-medical switching.
US physicians are generally comfortable with non-medical switching if they are leading the switch, 80% of respondents are comfortable switching a patient to a biosimilar, see Figure 2.
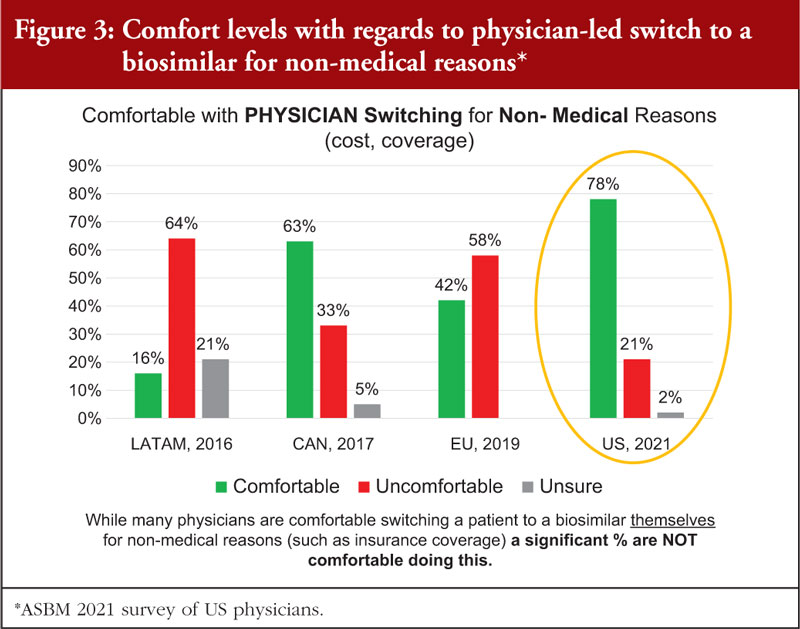
Globally, physicians are divided on physician-led non-medical switching. While many physicians are comfortable switching a patient to a biosimilar themselves for non-medical reasons such as insurance coverage, a significant percentage are NOT comfortable doing this, see Figure 3.
Dr McKibbin noted that there are many different reasons for concern over non-medical switching. In many cases, a patient goes through several rounds of trial and error with their physician to find the right treatment that works for them. This process often takes several years and is the basis of the doctor–patient relationship.
Opposition to third-party non-medical switching
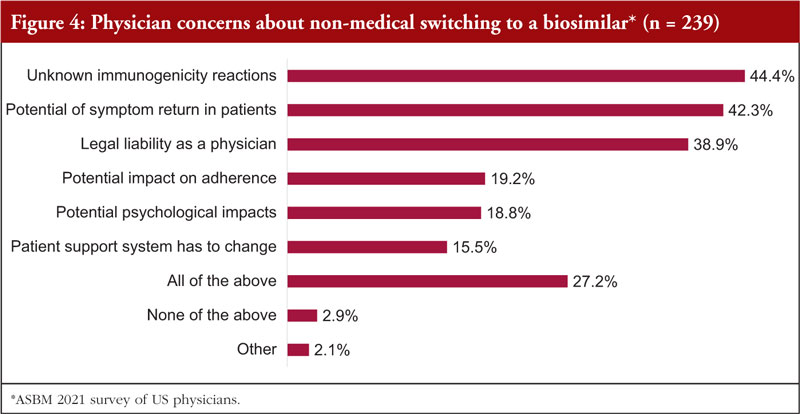
The survey revealed that majority (about 60%) of US physicians are not comfortable with third-party non-medical switching for a patient who is stable on their current treatment. This was due to several concerns that are highlighted in Figure 4.
Survey data revealed that globally, it is very important, or critical, to physicians that they are able to prevent third-party substitution that they do not feel is medically appropriate. In addition, pharmacists want ‘more data’ before there is automatic substitution at the pharmacy level.
For example, a 2016 Australian survey [21], revealed that the majority (53%) of Australian physicians wanted clinical trials showing no loss of safety or efficacy switching between originator to biosimilar. In addition, 81% wanted switching studies before permitting automatic substitution between biosimilars. The 2017 Canadian survey [22] revealed that more than 80% of physicians wanted switching studies to be carried out before automatic substitution was permitted.
In both Australia and Canada, where forced switching policies have been adopted, there has been opposition from physician organizations.
US’s interchangeable designation
In the US, a class of interchangeable biosimilars was created and is given this designation following the collection of additional data. This has been supported by US physician groups.
The US Food and Drug Administration (FDA) Interchangeability Guidance (drafted 2017, finalized 2019) states that ‘Interchangeable biosimilars’ may be substituted at the pharmacy without physician involvement [7]. The additional required data may include:
- Identification and analysis of critical quality attributes
- Identification of analytical differences and an analysis of the potential clinical impact of such differences
- An analysis of the mechanism of action in each condition of use
- An analysis of differences in ex pected pharmacokinetics (PK) and biodistribution in different patient populations
- An analysis of differences in expected immunogenicity risk
- An analysis of differences in expected toxicity
- Information on factors that could affect safety or efficacy.
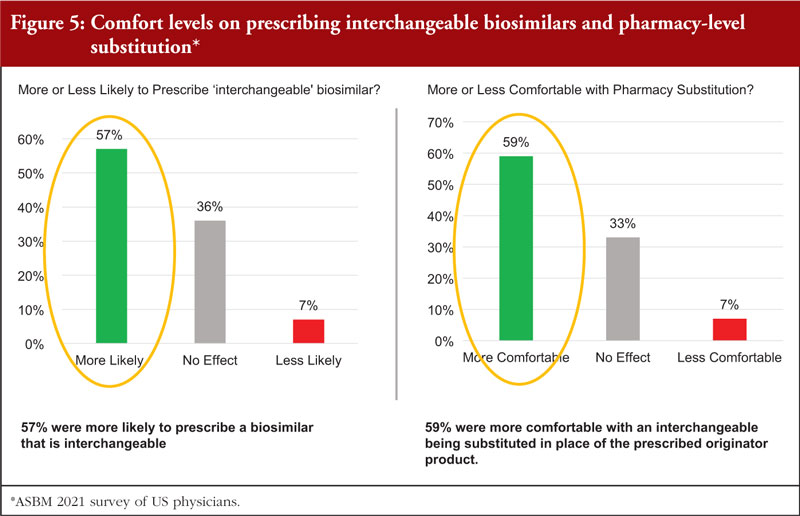
Following the introduction of the interchangeable designation, physicians have revealed that they are more comfortable with prescribing and substitution of these biosimilars, see Figure 5.
According to Dr McKibbin, it seems that the additional data requirements for an ‘interchangeable’ biosimilar show great promise in addressing physician concerns about switching and for building confidence in biosimilars.
Interchangeability of biologicals/biosimilars: US perspective
Amgen’s Dr Leah Christl first outlined the definitions of pharmacy substitution, switching and interchangeability.
Pharmacy substitution: A practice where one drug is dispensed in place of another at the pharmacy level, without consulting the prescribers [23, 24].
Switching: Physician may elect to prescribe one medicine in place of another with the same therapeutic intent [24]. In Europe, in the context of biosimilars, the term ‘switching’ is used synonymously with the term ‘interchangeable’ [25].
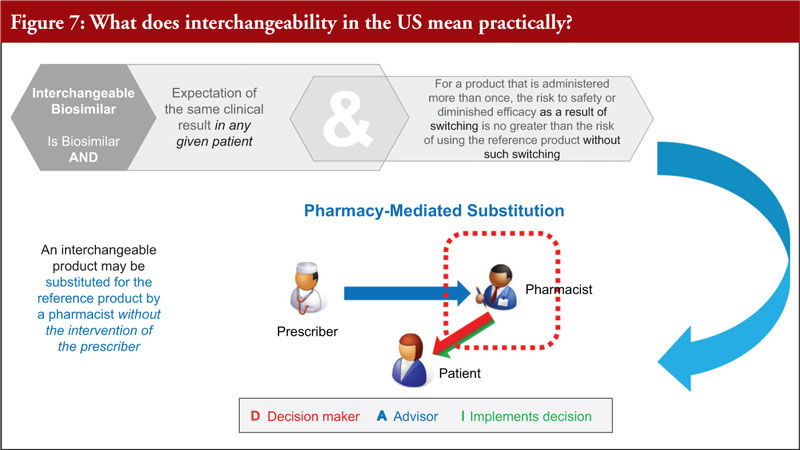
Interchangeable: In the US this is defined by statute to mean that a biosimilar product is expected to behave the same in ‘any given patient’ and that there is no negative impact resulting from alternating or switching between the biosimilar product and the reference product [26]. US state pharmacy laws permit substitution at the pharmacy of only biosimilars that FDA has deemed ‘interchangeable’ [7].
The Biologics Price Competition and Innovation Act (BPCI Act) of 2009 created an abbreviated licensure pathway for biological products that are demonstrated to be biosimilar to (and in some cases also interchangeable with) an already FDA-approved biological product. FDA uses this definition of inter changeability when reviewing a request for an interchangeability designation.
Referring to the US interchangeability designation – an interchangeable biosimilar is a biosimilar that is expected to give the same clinical result in any given patient, and for a product that is administered more than once, the risk to safety or diminished efficacy as a result of switching is no greater than the risk of using the reference product without such switching.
It was also highlighted that a designation as an interchangeable biosimilar:
- Requires additional data and information to scientifically support the statutory definition and pharmacy-level substitution.
- Does not imply anything about the quality of the product:
º Non-interchangeable biosimilars are held to the same quality standards as interchangeable biosimilars. - Is not required for physicians to prescribe a biosimilar product in place of the reference product to treatment-naïve patients or patients currently in treatment:
º Physicians should practice evidence-based medicine and consider the risks/benefits of switching patients between a reference product and its biosimilar.
Evidence to support interchangeability
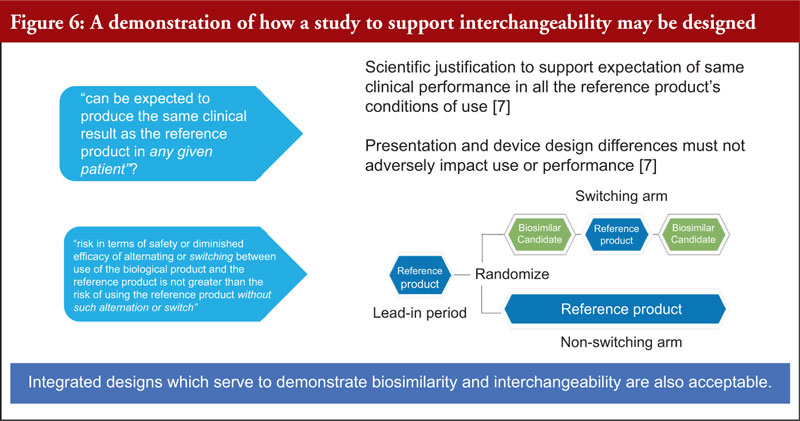
Dr Christl explained the evidence needed to support an interchangeable designation. Here, scientific justification to support the expectation of same clinical performance in all the reference product’s conditions of use is required. In addition, the presentation and device design differences must not adversely impact use or performance [7].
Figure 6 demonstrates how a study to support interchangeability may be designed. Integrated designs which serve to demonstrate biosimilarity and interchangeability are also acceptable.
Dr Christl shared the evidence required to support the interchangeability designation for Semglee® (insulin glargine-yfgn), see Table 3, and Cyltezo® (adalimumab-adbm), see Table 4.
Per the FDA Draft guidance for industry, Clinical Immunogenicity Considerations for Biosimilar and Interchangeable Insulin Products [31], insulin biosimilars generally do not need to have a switching study to support a demonstration of interchangeability. This is because ‘a comprehensive and robust comparative analytical assessment between a proposed interchangeable insulin product and the reference product demonstrating that the proposed interchangeable product is ‘highly similar’ to the reference product with very low residual uncertainty about immunogenicity generally would mean that an applicant would not need to conduct a comparative clinical immunogenicity study, e.g. a switching study, to support licensure under section 351(k)(4) of the PHS Act so long as the statutory criteria for licensure as an interchangeable are otherwise met’ [31].
Exclusivity for interchangeable biologicals
The first interchangeable biosimilar approved is entitled to a period of exclusivity [32]:
- Exclusivity is a period during which FDA must wait before granting a subsequent biosimilar to the same reference product an interchangeability designation
- Multiple exclusivity triggers and considerations
- The applicable trigger that would result in the shortest of any of the exclusivity periods is what would apply in a given situation.
FDA draft guidance on exclusivity for the first interchangeable biosimilar biological product is expected in 2022 [33] and that proposed legislation under discussion may impact first interchangeable exclusivity provisions.
The Purple Book
In the US, the ‘Purple Book’ is a searchable, online database of biological products, including any biosimilar and interchangeable biological products, licensed by FDA under the Public Health Service Act (the PHS Act) [34].
The Purple Book searchable database includes:
- The biological product licensing date
- If the biological product has been determined to be biosimilar to or interchangeable with a reference biological product
- The date of expiration of applicable regulatory exclusivity for a biological product if FDA has made an exclusivity eligibility determination.
- Patent information for certain licensed biological products.
There are other categories as well that can be selected through the advanced search feature. This is not an exhaustive list of the categories of information Purple Book provides.
Pharmacy substitution of interchangeable biosimilars
In practice, the interchangeability designation means that the interchangeable product may be substituted for the reference product by a pharmacist without the intervention of the prescriber, see Figure 7.
However, although FDA designates a biosimilar as interchangeable, US state pharmacy laws control substitution in the US [35]. Once an interchangeability designation is granted, a biosimilar may be substituted for the reference product by a pharmacist without the intervention of the prescriber in states that have approved legislation or regulations establishing state standards for biosimilar substitution [7, 35]. Dr Christl concluded by noting that as of June 2022, all 50 states, plus DC and Puerto Rico have enacted legislation establishing substitution laws for interchangeable biosimilars [35–37].
Patients’ perspective and expectations of non-medical switching of biologicals/biosimilars
Dr Durhane Wong-Rieger, President and CEO of the Canadian Organization for Rare Disorders explained that critics of non-medical switching and forced substitution policies of biologicals/biosimilars are often, and inaccurately described as being ‘against’ biosimilars. Regarding patients, she notes that they are not anti-biosimilar, but pro-biosimilar. In addition, patients share the same goals as government payers who are implementing non-medical and forced switching, these are:
- increased access to biological therapies
- lower costs (that free funding to spend on other health benefits)
- more treatment choices.
Despite having shared goals, there are disagreements with the method by which governments are pursuing these goals, they are:
- limiting patients’ role in their treatment decision-making
- restricting choice of patient/physician
- ignoring variability in individual patient response
- dismissing concerns of patient and physician organizations regarding forced switching
- collecting insufficient data tracking Real-World Evidence about patient response that would address concerns about switching.
Dr Wong-Rieger highlighted that patients support biosimilars but have concerns over unnecessary switching. Patients on biological therapies generally have chronic diseases, thus, changes in treatment can have a significant impact on them. Many patients take years to find a medicine that works for them to help control disease and if a biological that is working for a patient, decisions related to switching therapies should be carefully considered. Changes in therapy could lead to an immune response and/or a loss of response to the new and old therapy, exposing patients to a scenario with no, or fewer, treatment options.
Biosimilar switching: an oncology scenario
Dr Wong-Rieger gave the example of oncology patients leaders’ attitudes about biosimilar switching. She noted that cancer patients have only one chance to get the right drug and you cannot tell right away whether treatment is working or if there are side effects (may appear years later). Here, patients would prefer treatment with more safety and efficacy data rather than less. Regarding biologicals, these are highly specific to individual patients and you cannot extrapolate from one tumour type to another or from one stage to another. It is generally advised that, for cancer patients receiving adjuvant therapy, if patient is stable, they should not switch as there are unknowns related to individual impact. Oncology clinics lack long-term follow-up to assure pharmacovigilance needed for safe use of biosimilars. In this scenario, making healthcare savings is supported but prescribing should be based on ‘best for patient’ not ‘best for system’.
Biosimilar switching: an ophthalmology scenario
Biosimilars are now available in the field of ophthalmology. They offer new, cost-saving alternatives to current treatments for retinal diseases which can cause blindness. However, it is important to guarantee that the biosimilar substitution policies for vision care are patient-centred: there is no coming back from a switch that was inappropriate for a particular patient – patients and physicians must determine this based on a patient’s needs, not third parties.
Advocare Network Patient Survey
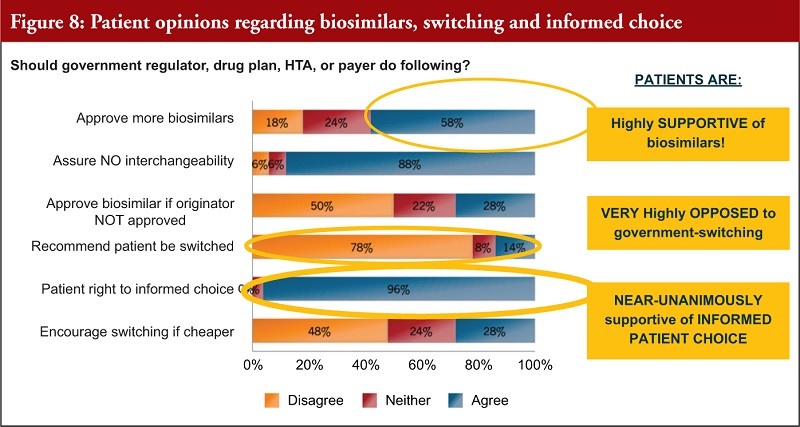
In mid-2016, a Consumer Advocare Network Patient Survey [38] of 200 patients with varied conditions from diabetes, inflammatory, blood disorders, immune-related, cancers, multisystemic, lysosomal storage, cardiovascular disease and cancer was undertaken.
Figure 8 shows that patients are highly supportive of biosimilars, very opposed to government switching and almost unanimously supportive of informed patient choice.
The Institute for Optimizing Health Outcomes – Europe
The Institute for Optimizing Health Outcomes has a scorecard that rates the biosimilar policy frameworks of European countries across success factors for biosimilar sustainability:
- Improving patient access and physician prescription choice of safe, high-quality biological medicines
- A framework that considers the needs of all stakeholders
- Providing a means to manage existing healthcare budgets
- Safeguarding a healthy level of competition and supply.
Dr Wong-Rieger highlighted that Canada’s public drug plans would have scored at the bottom. By taking away physician pre scription choice and mandating non-medical biosimilar switching, the government has set up a non-sustainable situation. This has led to patient backlash over the disregard for their biologicals management challenges and anxieties about switching.
Patient/physician choice does not need to be sacrificed to achieve biosimilar savings. The experience of European countries has shown that it is possible to build strong, successful and sustainable biosimilar markets that achieve a high number of approvals, high uptake rates, and high savings. These can come without:
- Automatic substitution of biologicals and biosimilars
- Switching current/stable patients
- Limiting which approved products can be prescribed/will be reimbursed.
It is important that Canadian patients should have the same options as their counterparts in Europe.
In conclusion, Dr Wong-Rieger noted that patients understand and share the goals of regulators who want to encourage use of biosimilars. However, they disagree with regulators’ methods, i.e. going around patients and physicians to accomplish this without their involvement. She stressed that these goals can be achieved without sacrificing patient/physician control of treatment decisions. Biosimilar policies need to factor in the concerns of patients and physicians and that these policies need to work for the patient community. In addition, patients, via patient organizations, should be included as partners in every step of the biosimilar policymaking process. Patients themselves must remain partners in their own treatment decision-making process. It is key that the government also works to address, not ignore, patient and physician concerns, e.g. track outcomes of biological switching and collect and analyse real-world evidence.
Canadian non-medical switching policies on biologicals/biosimilars – implications for physicians and patients
This presentation was given by Professor Philip J Schneider (Pharmacist, Ohio State University).
Professor Philip J Schneider, pharmacist, and Professor of Pharmacy at the Ohio State University, outlined that there are several issues to consider when assessing Canada’s non-medical and forced switching policies:
- Does it effectively achieve savings?
- Does it prioritize the patient?
- Does it adequately consider physician concerns/objections?
- Does it reflect the principles upon which it was enacted, i.e. as a means of ‘catching up to Europe’?
- Does it promote sustainable biosimilar market?
- Does it build confidence in biosimilars?
In Canada’s forced switching policies, there is relatively little transparency relative to those of Denmark and Norway, the only European countries to use a national tender system, Of these, only Denmark, following a transparent bidding process process, will solely reimburse the winning product except in rare substantiated circumstances. Many of the savings biosimilars bring come from innovator products and additional biosimilars cutting prices to compete. As such, forced-switching artificially achieves high market share but loses any savings which would occur from competition. Professor Schneider asks, ‘Are we sub stituting one monopoly for another?’.
In addition, patient objections to forced switching policies have been well-documented for many years. While creating numerous exceptions to these policies can mitigate some of their worst effects, this highlights the problem that the policy itself is wrong at its core because it makes the patient–physician relationship secondary or tertiary, rather than central to patient care.
Regarding physicians, it was noted that a key role for them is to function as learned intermediaries that can balance patient-specific factors against policies made by governments (and other payers) based on population-derived factors. However, we have seen that Canadian non-medical switching policies are typically enacted despite a lack of acceptance (and often strong objections) among physicians. It was also noted that in 2020, the INESSS report stated ‘Non-medical switching in patients being treated with a reference biologic is generally not accepted by learned societies and the consulted clinicians’ [10].
Canada’s policies have little similarity to the successful switching policies in Europe. However, the proponents of the forced-switching policy in Canada frequently cite European biosimilar policies as a basis for policy in Canada. These European governments achieved their success by:
- avoiding automatic substitution
- preserving and expanding rather than restricting patient/physician choice
- achieving savings through competition between many reimbursed products.
This is the direct opposite of what is happening in forced-switching provinces of Canada.
In addition, several of the questions highlighted above are considered in the white paper: ‘A critical review of substitution policy for biosimilars in Canada’ [9]. This white paper also identified three ‘must-haves’ for any biosimilars policy:
- Physicians should have the freedom to choose between off-patent originator biologicals and available biosimilars and to act in the best interest of their patients based on scientific evidence and clinical experience.
- Tenders should be designed to include multiple value-based criteria beyond price, e.g. education, services, available dose strengths, and provide a sufficient broad choice (multi-winner tenders versus single-winner tenders) to ensure continuity of supply and healthy competition.
- A level playing field between all participating manufacturers is the best way to foster competition; mandatory discounts which place artificial downward pressure on manufacturers do not engender a sustainable market environment.
Professor Schneider added that physician confidence in biosimilars is high, in Canada and elsewhere. However, they want more data showing safety of switching and they still want to be involved in substitution decisions. Data show these attitudes are consistent among physicians worldwide. He also noted that ignoring and dismissing these concerns does not build confidence. For example, and in contrast to Canada, the US is achieving uptake rates comparable to those of Europe (40%–80%) by addressing these concerns, e.g. interchangeability, state substitution laws.
The key reasons contributing to the failure of forced-switching policies include that:
- They ignore physician concerns and objections, fail to let confidence grow through data, prioritize short-term savings over long-term savings that result from competition, and undermine growth and sustainability of biosimilar markets.
- In effect, they fail by seeking a shortcut to uptake/savings rather than creating conditions for the desired behaviours to occur naturally, e.g. European and US markets.
Professor Schneider concluded that Health policies should always begin and end with the patient and the patient/physician relationship is central to making treatment decisions – a policy which removes this from the equation must meet a very high burden of proof.
Summary of panel discussions/Q&A
Following the speaker’s presentations, there was a panel discussion moderated by Dr Steven Stranne and five questions were discussed.
Question 1: Could you tell us more about the costs for the patient associated with non-medical switching?
Ms Maaghop from Canada’s Gastrointestinal Society noted that, in the context of Canada, the patient support programmes, which administer the originator biologicals and biosimilars are funded by pharmaceutical companies and the government does not have to bear the cost burden of these. Despite these programmes existing, a lot of coordination is still required. Throughout the forced switching processes there were delays in communication between the patients and the healthcare providers, which caused issues as outlined by Ms Attara.
Ms Maaghop gave the example of a study carried out by gastroenterologists in Alberta where they calculated that for 2,000 patients, it was estimated that a minimum of 650 clinic hours [39] were required for the administration of a non-medical switch for patients in addition to their usual, very-heavy work schedules. These increased hours were not even possible when the assessment was done during 2019 and the pandemic confounded the situation, making it very difficult for gastroenterologists to meet the demands. The doctors also calculated that this would lead to more than 60 avoidable surgeries in Alberta.
Ms Rathod of the International Federation of Ageing noted that there is a risk of loss of stability for the patients that are switched, i.e. increased risk of disease worsening [39]. This is significant as it often takes several years for a patient to find a drug that works well for them. There are also geographical and logistical issues that come with accessing biosimilars, especially when clinics for biologicals are separated from those where the biosimilars are located.
Professor Schneider noted that usually, switching therapies is done for cost-based reasons and so switching is likely to improve affordability for patients. However, this may also undermine confidence due to the perception that they are receiving a less expensive and therefore inferior drug. This creates a nocebo effect. Therefore, for patients to have confidence in a biosimilar, clinicians need to spend time with the patient, explaining the benefits of biosimilars in terms of cost, safety and efficacy and such conversations can be time consuming.
Dr McKibbin noted that, from a physician’s perspective and as lead author on a white paper of the Digestive Disease National Coalition [40] that looked at non-medical switching and authorization tactics. This showed that every type of pharmacy manipulation and control has a hidden cost to the system, such as lack of adherence. The idea that you are just switching one bottle of medication for another, cheaper one, in the pharmacy is not entirely straightforward and the hidden costs need to be considered. Patients have complex chronic diseases and need to be looked at individually. The one-dimensional perspective can be given, but each case is far more multifaceted.
Question 2: How can a patient organization keep track of developments like the interchangeable status?
Dr Christl noted that the best way to keep up to date on the status of biological products is with FDA’s Purple Book [34]. The Purple Book provides information on all FDA-approved biological products, including information regarding biosimilars and interchangeable biosimilars. The database is also set up so that if a brand name or active ingredient is searched for, then all available related products will come up. Here, prescribers and patients can see what products are approved, although not all those approved are available. FDA also shares information when they approve new products.
Ms Rathod added that this reiterates the importance of having information publicly available and for this to be regularly updated. This helps patient organizations ensure that the correct information is passed on to patients in a timely manner for decisions on treatment options to be made.
Dr Christl added that there is no all-encompassing resource for products that are undergoing clinical trials. There is also the registry site for clinical trials at www.clinicaltrials.gov. Getting information on this comes from, e.g. individual companies, trade press, market analyst positions. However, this type of information is not always publicly available. Patient organizations can potentially keep track of this and provide it as a resource, but this requires manual data mining.
Ms Maaghop noted that in Canada, the federal regulator, Health Canada, has deferred the authority over interchangeability status to provinces and territories. There is, therefore, variability in what the policies look like across the country. Patient organizations have a role to play here.
Question 3: Can we compare transparency in Canada to some of the experiences in Norway, Denmark, and the EU?
Professor Schneider noted that, in Europe, and specifically in Norway and Denmark, where they have adopted single tendering systems, patients and physicians are involved in decisions at the front end. This is not just a regulatory decision, but prescriber input is valued. In addition, alternative treatment options are available for selection by physicians. Norway moved from a single tendering system to a multi-tender system due to supply chain issues and to ensure reliability of supply. In addition, in these nations, culturally, it has been agreed by physicians and regulators that the issue of rising cost of healthcare needs to be addressed. However, this needs to be achieved in a way that addresses individual needs of patients, effectiveness and safety of therapy is paramount. These countries have adopted transparent policies, which are lacking in Canada.
Mr Reilly noted that in a meeting he attended with the British Columbia regulator, a representative from Norway’s health system presented but cautioned that their system would likely not work in Canada. He also noted that when he attends European biosimilars meetings, including periodic meetings at the European Commission, he does not see Canadian representatives. He remarked that this absence is unusual for a country supposedly modelling their biosimilar practices on those of Europe. The meetings are reflective and forward looking and a recent meeting highlighted that single tender systems are ineffectual. In Canada, it was suggested that adverse event tracking would be done through monitoring increased hospitalizations, yet this was not a realistic measure. He also highlighted the success of the US model and voiced the opinion that the Canadians are not adopting the correct approach which is not interactive and is not a long-term way to buy and build.
Question 4: What is the most important thing that policymakers should consider when contemplating a long-term sustainable biosimilars programme?
Dr Christl noted that, from a regulatory standpoint, global regulatory authorities like FDA and the European Medicines Agency (EMA) look at scientific alignment and leveraging global development programmes, recognizing that biosimilars are being developed globally. From a development standpoint, work needs to be done to ensure manufacturers are not conducting studies that are not scientifically warranted or recommended by regulatory authorities where authorization is sought. There are also aspects related to ensuring that data from clinical studies is not duplicated and here bridging data can be used to support the use of what is known as a non-regionally approved comparator data. It is key to look at ways to streamline development and make sure it is efficient. In addition, processes and approvals need to be done in a timely fashion. Maintaining robust, scientifically appropriate regulatory standards is essential to ensuring stakeholder confidence in biosimilars globally. There is confidence in what the stringent regulatory authorities are doing to approve the products and prescribers know that when a biosimilar is approved to be as safe and efficacious as a reference product, they can trust this. It is important to maintain the regulatory standards and confidence in them. This will ensure that the acceptance and uptake of biosimilars globally is maintained. Regulators are focused on this but are also looking at efficiencies in terms of global development to bring biosimilars to the market in a timely fashion.
Mr Reilly noted that streamlining of regulatory standards is important in terms of delivering a high standard in an efficient way. There are conversations going on about reducing regulatory standards and making them less stringent, particularly in countries that find it harder to meet the standards. He stressed that the ASBM has been involved in these conversations with European regulators and has discussed the morality of reducing the stringency of regulations in some regions. At the ASBM, it is believed that the confidence we have in biosimilars is due to the position of the standards and these should not be lowered, no matter where you are. The surveys that have been carried out in recent years across Latin America [41], Canada [22], and the EU [6], have all shown really high levels of physician confidence in biosimilars due to where the standards are.
Mr Reilly said that the Canadian regulators say that their policy is based on catching up with Europe, however, it is not possible to do this unless the foundational understanding of the European approach is there. The US is in a good position today in terms of competition and pricing of biosimilars. The Canadian approach is not good for patients or physicians and does not increase confidence. It is important to maintain high standards and foster competition in the marketplace and build confidence with the patient and physician community.
Dr McKibbin noted that from the advocacy patient-physician relationship, transparency is key. Adding that, if you look at drug studies and approvals, the definitions of endpoints change with time and what used to be considered a success is now much more stringent. Transparency in data that we continue to collect to monitor safety, immunogenicity and effectiveness of biosimilars, is the key to improving the future. The next step to improving the whole system is interchangeability. The 360 concept of continuous improvement gives us the confidence in the system to move forward. Interchangeability will cut costs and we will be able to substitute at the pharmacy, there will be less hassle and cost at the physician’s office. Transparency and discussion are the key to moving forward.
Question 5: As policymakers and other stakeholders, how fast can we forestall shortages of biological products (originators and/or biosimilars)?
Professor Schneider advised that as a pharmacist, drug shortages are constantly managed. These have been a problem for a long time, but they have been made much worse due to COVID and global supply chain issues. The recent infant formula shortage in the US is a sharp example; this happened because there was a concentrated source of formula, with 70% coming from one manufacturer and they had problems with the manufacturing facility. The US then had to import formula. Squeezing the market so that there are fewer sources of product is available and makes us vulnerable to supply chain issues.
Mr Reilly added that, in 2004 there was an influenza vaccine shortage, 1.5 million doses of flu vaccine or half of the US supply, was missing. This was due to a regulatory issue in the EU that prevented doses from a supplier reaching the US. This strengthens Professor Schneider’s point about having multiple suppliers. In the EU they discuss tenders and multi-winners as in single tender systems you can end up with shortages. It is important to have multiple competitors in the arena.
Concurrent online Q&A
Due to the nature of the webinar, the audience had the opportunity to ask questions throughout the meeting, by submitting them online during the presentations and Q&A session.
Question 1: Would you be able to provide some context to Health Canada’s website that states, ‘No differences are expected in efficacy and safety following a change in routine use between a biosimilar and its reference biologic drug in an authorized indication’. Is this statement misleading?
Ms Jaymee Maaghop of the Gastrointestinal Society, Canada replied that, ‘We asked for clarification on this sentence during a recent consultation with Health Canada, especially since they had a different definition in 2016: ‘Health Canada recommends that a decision to switch a patient being treated with a reference biologic drug to a biosimilar should be made by the treating physician in consultation with the patient and taking into account available clinical evidence and any policies of the relevant jurisdiction.’ Unfortunately, to date this is still not clear.
Question 2: How can patient organizations track other biologicals in line for gaining interchangeable status, like Cyltezo/Humira? The work we do to help patients through the transition will largely rely on access to which are approved as interchangeable, and which are not.
Dr Leah Christl advised that FDA’s Purple Book can be a good resource.
Conclusions
This webinar provided the opportunity to gain insight on ‘switching and substitution practices’ in Canada and Europe and the interchangeable designation in the US. The speakers shared their experience and knowledge on switching and substitution practices globally and principles on interchangeability of biosimilars in the US. There was specific emphasis on the forced-switching policies adopted in Canada and their shortcomings. The speakers also highlighted that improved information and protecting the physician–patient relationship in medical switching of biologicals/biosimilars will encourage smooth uptake of biosimilars. The regulatory and scientific standards for interchangeable biosimilars in the US were generally supported. In addition, the importance of a multi-stakeholder approach protecting the physician–patient relationship in the practice of switching of biologicals, including biosimilars, was emphasized. Overall, it was highlighted that maintaining trust in biosimilars is key to safeguarding the physician–patient relationship. This can be achieved through robust policy and regulation and upholding transparent practices.
Acknowledgement
The Generics and Biosimilars Initiative (GaBI) wishes to thank all speakers and moderator in delivering the presentations, implementing the panel discussion and clarifying information when finalizing the meeting report, as well as Mr Michael S Reilly for his strong support through the offering of advice and information during the preparation of the webinar.
The authors would like to acknowledge the help of the webinar speaker faculty and all participants, each of whom contributed to the success of the webinar and the content of this report, as well as the support of the moderator in facilitating meaningful discussion during the panel discussions, and contributing to the finalization of this meeting report.
Lastly, the authors wish to thank Ms Alice Rolandini Jensen, GaBI Journal Editor, in preparing and finalizing this meeting report manuscript.
Speaker Faculty, Panelists and Moderator
Speakers
Gail Attara
Leah Christl, PhD
Ralph D McKibbin, MD, FACP, FACG, AGAF
Michael S Reilly, Esq
Professor Philip J Schneider, MS, FASHP, FASPEN, FFIP
Durhane Wong-Rieger, PhD
Panelists
Jaymee Maaghop
Urvashi Rathod
Moderator
Steven Stranne, MD, JD
Editor’s comment
Speakers and moderator had provided feedback on the article content and panel discussion, read and commented the revised content of the manuscript, and approved the final report for publication.
Competing interests: The webinar was funded by ASBM.
Provenance and peer review: Not comm ission ed; externally peer reviewed.
Authors
Michael S Reilly, Esq
Gail Attara
Ralph D McKibbin, MD, FACP, FACG, AGAF
Professor Philip J Schneider, MS, FASHP, FASPEN, FFIP
References
1. GaBI Online – Generics and Biosimilars Initiative. Biosimilars approved in Europe [www.gabionline.net]. Mol, Belgium: Pro Pharma Communications International; [cited 2023 Jan 20]. Available from: www.gabionline.net/biosimilars/general/biosimilars-approved-in-europe
2. GaBI Online – Generics and Biosimilars Initiative. Biosimilars approved in the US [www.gabionline.net]. Mol, Belgium: Pro Pharma Communications International; [cited 2023 Jan 20]. Available from: www.gabionline.net/biosimilars/general/biosimilars-approved-in-the-us
3. Generics and Biosimilars Initiative. Non-medical switching of biologicals/biosimilars: Canada, Europe and the US; 20 July 2022. Available from: www.gabiworkshop.wixsite.com/asbm2
4. Generics and Biosimilars Initiative. Key factors for successful uptake of biosimilars: Europe and the US; 29 June 2022. Available from: www.gabiworkshop.wixsite.com/asbm1-1
5. Schneider PJ, Reilly MS. Policy recommendations for a sustainable biosimilars market: lessons from Europe. Generics and Biosimilars Initiative Journal (GaBI Journal). 2020;9(2):76-83. doi:10.5639/gabij.2020.0902.013
6. Feldman MA, Reilly MS. European prescribers’ attitudes and beliefs on biologicals prescribing and automatic substitution. Generics and Biosimilars Initiative Journal (GaBI Journal). 2020;9(3):116-24. doi:10.5639/gabij.2020.0903.020
7. U.S. Food and Drug Administration. Considerations in demonstrating interchangeability with a reference product. Guidance for Industry. May 2019 [homepage on the Internet]. [cited 2023 Jan 20]. Available from: https://www.fda.gov/media/124907/download
8. Feldman M, Reilly MS. A white paper: US biosimilars market on pace with Europe. Generics and Biosimilars Initiative Journal. (GaBI Journal). 2020;9(4):150-4. doi:10.5639/gabij.2020.0904.025
9. Reilly MS, Schneider PJ. A critical review of substitution policy for biosimilars in Canada. Generics and Biosimilars Initiative Journal (GaBI Journal). 2021;10(3):123-9. doi:10.5639/gabij.2021.1003.016
10. Institut national d’excellence en santé et en services sociaux. Safety of switching biologics and their interchangeability [homepage on the Internet]. [cited 2023 Jan 20]. Available from: https://www.inesss.qc.ca/en/publications/publications/publication/safety-of-switching-biologics-and-their-interchangeability.html
11. Gastrointestinal Society. Everything you need to know about biosimilars. Available from: https://badgut.org/biosimilars
12. Attara G, Bailey R, Bressler B, Marshall J, Panaccione R, Aumais G. Canadian patient and caregiver perspectives on subsequent entry biologics for inflammatory bowel disease. Available from: https://badgut.org/wp-content/uploads/SEB-Poster-Landscape-2016-02-04.pdf
13. Gastrointestinal Society. Biosimilar Focus Group (2017). Available from: https://badgut.org/biosimilars
14. Health Canada. Biosimilar biologic drugs in Canada: fact sheet. 2019. Available from: https://www.canada.ca/en/health-canada/services/drugs-health-products/biologics-radiopharmaceuticals-genetictherapies/applications-submissions/guidance-documents/fact-sheet-biosimilars.html
15. Canada’s Drug and Health Technology Agency. Biosimilar drugs. Available from: https://www.cadth.ca/biosimilar-drugs
16. Inesss. Etat des connaissances. Innocuité de la substitution et de l’interchangeabilité des médicaments biologiques. Mai 2020 [homepage on the Internet]. [cited 2023 Jan 20]. Available from: https://www.inesss.qc.ca/fileadmin/doc/INESSS/Rapports/Medicaments/INESSS_Biosimilaires_EC.pdf
17. Moayyedi P, Benchimol E, Armstrong D, Yuan C, Fernandes A, Leontiadis G. Joint Canadian Association of Gastroenterology and Crohn’s Colitis Canada position statement on biosimilars for the treatment of inflammatory bowel disease. J Canadian Assoc Gastroenterol. 2020;3(1):e1-e9.
18. Gastrointestinal Society. Forced to switch: Canadian biosimilar experience. Survey report. December 2020 [homepage on the Internet]. [cited 2023 Jan 20]. https://badgut.org/wp-content/uploads/Biosimilar-Survey-Report.pdf
19. Gastrointestinal Society. Biosimilars in Alberta and BC survey results [homepage on the Internet]. [cited 2023 Jan 20]. Available from: https://badgut.org/wp-content/uploads/Biosimilars-in-AB-BC-Survey-Results.pdf
20. McKibbin RD, Reilly MS. US prescribers’ attitudes and perceptions about biosimilars. Generics and Biosimilars Initiative Journal (GaBI Journal). 2022;11(3):96-103. doi:10.5639/gabij.2022.1103.016
21. Murby SP, Reilly MS. A survey of Australian prescribers’ view on the naming and substitution of biologics. Generics and Biosimilars Initiative Journal (GaBI Journal). 2017;6(3):107-13. doi:10.5639/gabij.2017.0603.022
22. Safe Biologics. Canada survey 2017 [homepage on the Internet]. [cited 2023 Jan 20]. Available from: https://safebiologics.org/surveys/Canada2017/
23. U.S. Food and Drug Administration. Prescribing Interchangeable Products [homepage on the Internet]. [cited 2023 Jan 20]. Available from: https://www.fda.gov/files/drugs/published/Prescribing-Interchangeable-Products.pdf
24. Canada’s Drug and Health Technology Agency. International Policies on the Appropriate Use of Biosimilar Drugs. 2018 [homepage on the Internet]. [cited 2023 Jan 20]. Available from: https://www.cda-amc.ca/sites/default/files/pdf/es0333_international-policies-on-use-of-biosimilar-drugs.pdf
25. European Commission. What you need to know about biosimilar medicinal products. 2013 [homepage on the Internet]. [cited 2023 Jan 20]. Available from: https://www.medicinesforeurope.com/wp-content/uploads/2016/03/biosimilars_report_en.pdf
26. Afzali A, Furtner D, Melsheimer D, Melsheimer R. The automatic substitution of biosimilars: definitions of interchangeability are not interchangeable. Adv Ther. 2021;38(5):2077-93.
27. Blevins TC, Barve A, Raiter Y, Aubonnet P, Athalye S, Sun B, et al. Efficacy and safety of MYL-1501D versus insulin glargine in people with type 1 diabetes mellitus: results of the INSTRIDE 3 phase 3 switch study. Diabetes Obes Metab. 2020;22(3):365-72.
28. U.S. Food and Drug Administration. BLA 761201. BLA Approval [homepage on the Internet]. [cited 2023 Jan 20]. Available from: https://www.accessdata.fda.gov/drugsatfda_docs/appletter/2021/761201Orig1s000ltr.pdf
29. Boehringer-Ingelheim. VOLTAIRE-X phase III data in patients with moderate-to-severe chronic plaque psoriasis support interchangeability application [home page on the Internet]. [cited 2023 Jan 20]. Available from: https://www.biospace.com/article/releases/voltaire-x-phase-iii-data-in-patients-with-moderate-to-severe-chronic-plaque-psoriasis-support-interchangeability-application/
30. U.S. Food and Drug Administration. BLA761058/S008. Supplemental approval fulfillment of post marketing requirement [homepage on the Internet]. [cited 2023 Jan 20]. Available here: https://www.accessdata.fda.gov/drugsatfda_docs/appletter/2021/761058Orig1s008ltr.pdf
31. U.S. Food and Drug Administration. Clinical immunogenicity considerations for biosimilar and interchangeable insulin products. Guidance For Industry. Draft Guidance. November 2019 [homepage on the Internet]. [cited 2023 Jan 20]. Available from: https://www.fda.gov/media/133014/download
32. U.S. Food and Drug Administration. Biologics Price Competition and Innovation (BPCI) Act [homepage on the Internet]. [cited 2023 Jan 20]. Available from: https://www.fda.gov/media/78946/download
33. U.S. Food and Drug Administration. CDER guidance agenda new & revised draft guidance documents planned for publication in calendar year 2022 (July 2022) [homepage on the Internet]. [cited 2023 Jan 20]. Available from: https://www.fda.gov/media/134778/download
34. U.S. Food and Drug Administration. Purple book database of licensed biological products [home page on the Internet]. [cited 2023 Jan 20]. Available from: https://purplebooksearch.fda.gov/about
35. National Conference of State Legislatures. State laws and legislation related to biologic medications and substitution of biosimilars. 22 October 2018 [homepage on the Internet]. [cited 2023 Jan 20]. Available from: www.ncsl.org/research/health/state-laws-and-legislation-related-to-biologic-medications-and-substitution-of-biosimilars.aspx
36. Safe Biologics. Oklahoma becomes final state to permit biosimilar substitution. 2021 [homepage on the Internet]. [cited 2023 Jan 20]. Available from: https://safebiologics.org/2021/05/oklahoma-becomes-final-state-to-permit-biosimilar-substitution/
37. Council of the District of Columbia. D.C. Law 23-133. Access to Biosimilars Amendment Act of 2020 [homepage on the Internet]. [cited 2023 Jan 20]. Available from: https://code.dccouncil.us/us/dc/council/laws/23-133
38. Consumer Advocare Network. Patient survey [homepage on the Internet]. [cited 2023 Jan 20]. Available from: https://safebiologics.org/wp-con tent/uploads/2022/12/Advocare-Biosimilars-Pati ent-Perspective-Update-Nov-2018.pdf
39. Kaplan GG, Ma C, Seow CH, Kroeker KI, Panaccione R. The argument against a biosimilar switch policy for infliximab in patients with inflammatory bowel disease living in Alberta. J Canadian Assoc Gastroenterol. 2020;3(5):234-42.
40. McKibbin RD. Patient access to care and treatments in the cost-shifting era: preserving the patient-provider decision-making relationship. Available from: https://www.ostomy.org/wp-content/uploads/2020/11/HMC_2_DDNC_Whitepaper_2020.pdf
41. Reilly MS, Gewanter HL. Prescribing practices for biosimilars: questionnaire survey findings from physicians in Argentina, Brazil, Colombia and Mexico. Generics and Biosimilars Initiative Journal (GaBI Journal). 2015;4(4):161-6. doi:10.5639/gabij.2015.0404.036
|
Author for correspondence: Michael S Reilly, Esq, Executive Director, Alliance for Safe Biologic Medicines, PO Box 3691, Arlington, VA 22203, USA |
Disclosure of Conflict of Interest Statement is available upon request.
Copyright © 2023 Pro Pharma Communications International
Permission granted to reproduce for personal and non-commercial use only. All other reproduction, copy or reprinting of all or part of any ‘Content’ found on this website is strictly prohibited without the prior consent of the publisher. Contact the publisher to obtain permission before redistributing.