Summative usability evaluation of the YLB113 etanercept biosimilar autoinjector via simulation
Published on 2020/11/30
Generics and Biosimilars Initiative Journal (GaBI Journal). 2021;10(2):61-7.
Author byline as per print journal: Kelly Canham1, BSc Hons; Claire Newcomb2, MSc
|
Introduction/Study Objectives: Etanercept is a tumour necrosis factor inhibitor indicated for the treatment of several inflammatory disorders. Patients with these diseases may experience manual dexterity challenges. Autoinjectors may improve dose accuracy, treatment adherence and quality of life; and reduce injection-site reactions. Studies have indicated patients prefer autoinjectors to other injection methods, however, patients must be able to demonstrate safe and effective use of an autoinjector for it to be a viable option. The YLB113 etanercept autoinjector may be a substitutable biosimilar to reference etanercept (Pfizer Manufacturing, Puurs, Belgium). This study sought to confirm intended users of the YLB113 etanercept autoinjector could demonstrate safe and effective use. |
Submitted: 12 October 2020; Revised: 11 February 2021;Accepted: 15 February 2021; Published online first: 1 March 2021
Introduction/Study Objectives
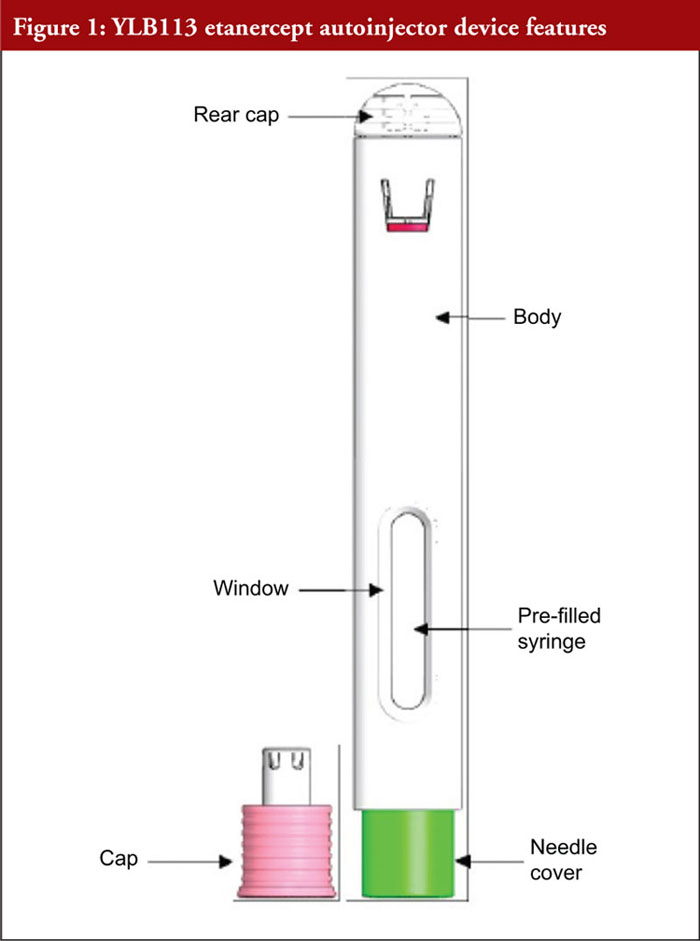
Etanercept is a tumour necrosis factor inhibitor indicated for the treatment of rheumatoid arthritis, juvenile idiopathic arthritis, psoriatic arthritis, ankylosing spondylitis, non-radiographic axial spondyloarthritis, and adult and paediatric psoriasis [1]. Patients suffering from the diseases for which etanercept is indicated may experience difficulties with manual dexterity that limit their ability to self-administer injections [2]. Autoinjectors (AIs) have been shown to offer several benefits to patients, including improved dose accuracy, ease of use, improved treatment adherence, and a reduced number of injection-site reactions [2]. In addition, studies suggest patients prefer AIs to other injection methods [3, 4]. However, for an AI to be a viable option, patients must be able to demonstrate safe and effective use [2]. Mylan, in collaboration with Lupin, is developing an etanercept AI, referred to here as the YLB113 etanercept AI, as a substitutable biosimilar product to Enbrel® (Pfizer Manufacturing, Puurs, Belgium) in the SureClick® AI. The etanercept solution is supplied in a single-use, disposable AI containing 50 mg/mL, as shown in Figure 1. Figure 2 shows a complete chronological task list for the injection process. In general, to administer a dose, the user must remove the needle cap and press the AI against the injection site. When the button is pressed, the AI clicks to indicate the start of the injection. A second click indicates the end of the injection. Following the second click, the AI must be held in place for an additional 15 seconds to ensure a full dose is delivered. After 15 seconds, the user can remove the AI from the skin and discard it in a sharps container.
The objective of this study was to confirm the intended users of the YLB113 etanercept AI could demonstrate safe and effective use through usability criteria predefined through risk assessment of the tasks. This is consistent with guidance from the European Union and United States regulatory authorities that, when needed, a human factors/usability study with the final version of the device should be completed to validate users’ performance with representative users in the environment of use [5-7].
Methods
The study was conducted in accordance with the principles of Good Clinical Practice and the provisions of the Declaration of Helsinki of 1964 and its later amendments. Participants eligible for study recruitment had the nature, purpose and risks of the study explained to them by the moderator. Informed consent was given by all participants and those aged younger than 18 years completed assent forms and were accompanied by a parent or guardian. Participants were provided a copy of the informed consent form for the study and were allowed time to consider whether they wanted to participate. Participant names were not included on the video, and all data were stored under protected computer systems that were only available to the usability vendor project team. The study protocol and supporting materials were reviewed and approved on 21 June 2019, by Core Human Factors, an independent Institutional Review Board located in Philadelphia, PA, USA, prior to collecting data from participants.
This summative usability evaluation study was performed among participants representative of intended YLB113 etanercept AI users that included patients, caregivers and healthcare providers (HCPs). Patients were included if they were diagnosed with a condition as described in the therapeutic indications section of the Enbrel summary of product characteristics [1] or, as few as possible children who had used any AI, e.g. epinephrine. Patients were divided into age groups of 12 to 17, 18 to 64, and 65 years and older. Caregivers (non-professionals) were included if they routinely helped someone ad-minister their medications. HCPs were included if they were a licensed nurse practitioner or registered nurse who administers or trains patients to administer medications using an AI, regardless of the patient diagnosis.
Intended YLB113 etanercept AI users may or may not have experience with AIs, therefore, half of the patient and caregiver groups were naïve to AI use and half had prior experience with AIs. Testing was conducted at multiple independent research facilities in the UK by a usability vendor. The study was conducted in a simulated home-use environment in which a moderator, following a script, interacted 1-on-1 (moderator to participant) or 1-on-2 (moderator to adolescent and their parent or guardian) to observe participants interacting with the user interface to simulate dose administration in accordance with a predefined task. The environmental conditions included standard room lighting, minimal noise distraction, and low-level background noise. The YLB113 etanercept AIs were presented in a carton representative of the shape and design of the commercial packaging. The labelling used in this study represented the Australian product as European labelling was still in development, see Figure 3 and Figure 4. The samples used in the study were not suitable for human injection and were intended only for simulated injection into a foam pad on the table. The foam pad had a hard plastic base to minimize the risk for accidental needle-stick injury. Supplies were presented to the participants or were available in the testing room for each session, and included a carton with four AIs, instructions for use, foam injection pad, hand sanitizer and paper towels, alcohol swabs, adhesive bandages, sharps bin, first aid kit, portable eye wash kit, and telephone. In addition, the testing room was equipped with cameras to enable a wide view of the moderator and participant, as well as a close-up view of the participant completing the task, to allow the project team to re-watch the session to aid in any root cause analysis.
As intended for all YLB113 etanercept AI users, participants received training on the usage of the commercially equivalent, single-use disposable AI according to a training guide one day before usability testing. A minimum of 18 hours with only one overnight between training and testing sessions was required to represent a reasonable time for knowledge decay from the time of receiving a prescription to administration of the first dose. During the testing session, participants were asked to simulate delivering a dose of medication into a foam pad twice in a row. Participants were given an opportunity to use the system independently and in as realistic a manner as possible, without guidance, coaching, or critique. Additional tasks and knowledge-based questions were used for further demonstration of safe and effective use. At the end of the task, a post-task interview was conducted to investigate the root cause of any use errors or close calls through open-ended questions and to record the participant’s perspective of their interaction with the user interface. In addition, a dynamometer was used to assess grip strength. The highest of three measurements from the dynamometer was taken from each participant and was averaged to calculate the mean maximum grip strength for each user group. A pinch gauge was also used to assess pinch strength. The highest of three measurements from the pinch gauge was taken from each participant and was averaged to calculate the mean maximum pinch strength for each user group.
Results
A total of 79 participants were included in this summative usability evaluation study. All the HCP participants in this study were female (n = 15). Across all other user groups, 63% were female. Demographic information, including gender, dominant hand, mean maximum grip strength, and mean maximum pinch strength are shown in Table 1.
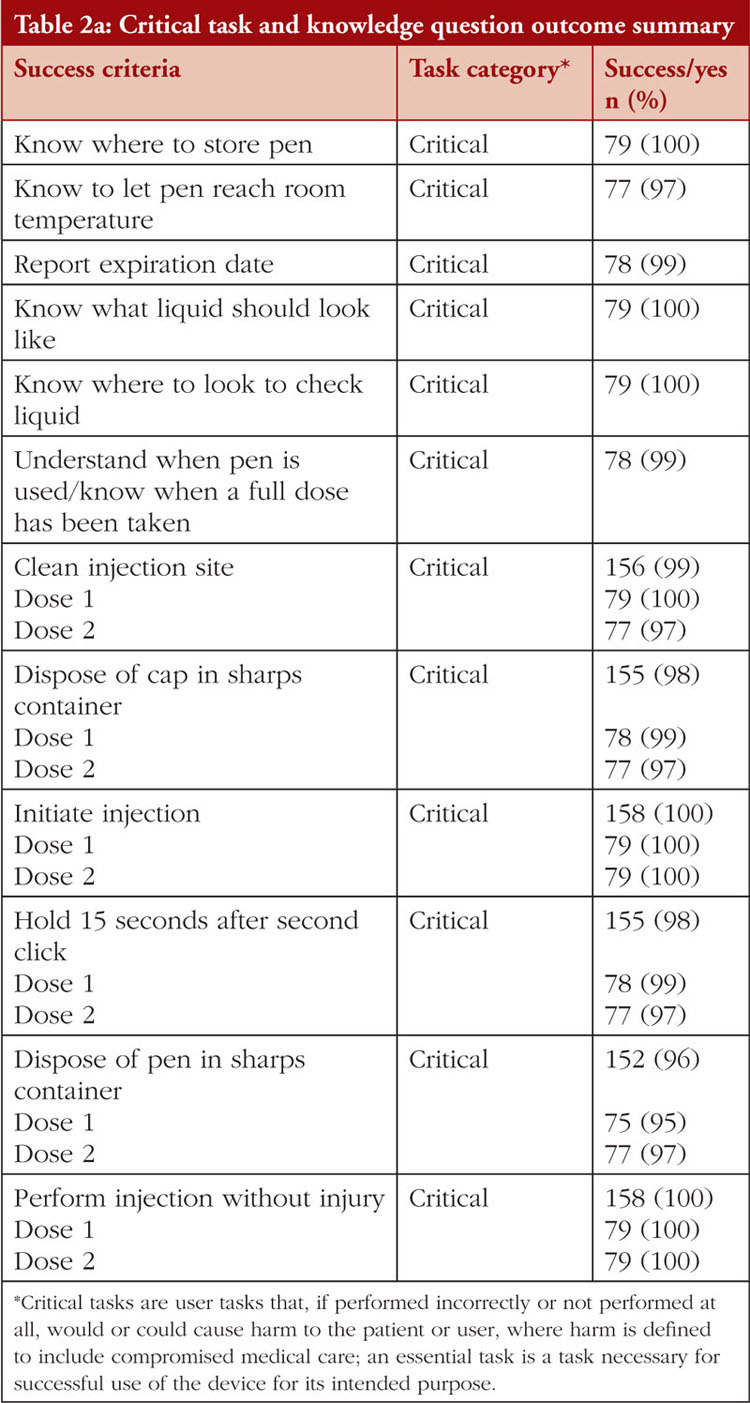
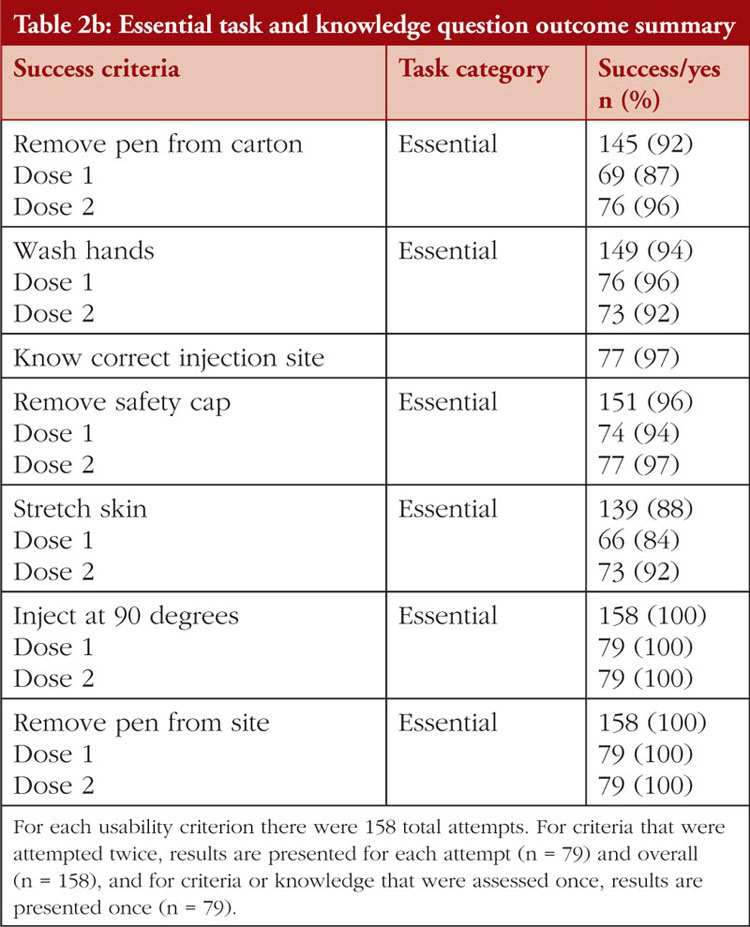
The task and knowledge question outcome summary across all groups is shown in Table 2a and Table 2b. All (100%) participants successfully delivered both simulated doses from the YLB113 etanercept AI into the foam pad and answered knowledge questions on where to store the pens, what the liquid should look like, where to look to see the liquid, injecting at 90°, initiating the injection, removing the pen from the injection site, and performing the injection without injury. Some participants experienced use errors, use difficulties, or close calls while performing tasks or answering knowledge questions. Success was recorded when no use errors, usability issues, close calls, or issues necessitating assistance were observed, see Table 3. Success rates for the tasks and knowledge questions were: removing the pen from carton (92%), knowing to let the pen reach room temperature (97%), reporting the expiry date (99%), knowing when a full dose has been taken (99%), washing their hands (94%), knowing the correct injection site (97%), cleaning the injection site (99%), removing the safety cap (96%), disposing of the cap in a sharps container (98%), stretching the skin (88%), holding the AI down for 15 seconds after the second click (98%), and disposing of the pen in the sharps container (96%). A total of 11 use errors occurred on critical tasks over 158 simulated injections. The root cause analyses of these use errors are shown in Table 4. Use difficulties only occurred when removing the pen from the carton (n = 11) and stretching the skin at the injection site (n = 1)
Discussion
In this summative usability evaluation study conducted at multiple independent research facilities in the United Kingdom, patients across all age groups, caregivers, and HCPs demonstrated a high rate of injection success using the YLB113 etanercept AI. All participants were able to successfully deliver two simulated injections into a foam pad, including patients and caregivers with or without prior AI experience. In addition, all participants indicated they knew where to store the pens, what the liquid should look like, and where to look to see the liquid. All participants also demonstrated success with injecting at 90°, initiating the injection, removing the pen from the site, and performing the injection without injury. Although the vast majority of participants demonstrated success across all other success criteria, some participants experienced use errors, use difficulties, or close calls while performing tasks or answering knowledge questions, see Table 4.
Limitations
A limitation of this study is that injections were simulated into a foam pad at a research facility as opposed to using the AI in a clinical or home-use setting. However, it is important to note that simulated-use testing, as employed in this study, is sufficient to assess the adequacy of the user interface for most combination products, according to health authority guidance [5-7]. In addition, it is anticipated that patients being prescribed the YLB113 etanercept AI will receive training, however, the training of all participants prior to simulating an injection limits the generalizability of these findings to those who have not received such training.
Conclusions
Patients across all age ranges, grips strengths, and pinch strengths; caregivers and HCPs were able to successfully deliver two doses of etanercept into a foam pad to demonstrate safe and effective use of the AI. The results from this study support the demonstration of safe and effective use of the YLB113 etanercept AI as a substitutable biosimilar product to Enbrel.
For patients
Antitumour necrosis factor inhibitors, such as etanercept, are indicated for use in patients suffering from inflammatory conditions in the form of rheumatoid arthritis, juvenile idiopathic arthritis, psoriatic arthritis, axial spondyloarthritis, and adult and paediatric psoriasis. Patients suffering from these conditions may receive benefits from the use of self-administered injectable medications, such as improved compliance, ease of use, dosing accuracy, and fewer injection-site reactions. Research indicates patients prefer autoinjectors (AI) to traditional injections, however, inflammatory diseases commonly present with clinical manifestations that limit dexterity and ability to manoeuvre AI devices. The YLB113 etanercept AI may be a substitutable biosimilar product to Enbrel®, and this manuscript reviews the summative usability studies of YLB113 etanercept AI among intended users. The findings confirm that intended users across all age ranges, and grip and pinch strengths, including patients, caregivers and healthcare professionals, were able to demonstrate safe and effective use of the device. The results from this study support the demonstration of safe and effective use of the YLB113 etanercept AI as a biosimilar substitute product to Enbrel®.
Declarations
Compliance with ethics guidelines
The study was conducted in accordance with the principles of Good Clinical Practice and the provisions of the Declaration of Helsinki of 1964 and its later amendments. Participants eligible for study recruitment had the nature, purpose, and risks of the study explained to them by the moderator. Informed consent was given by all participants and those aged younger than 18 years completed assent forms and were accompanied by a parent or guardian. Participants were provided a copy of the informed consent form for the study and were allowed time to consider whether they wanted to participate. Participant names were not included on the video, and all data were stored under protected computer systems that were only available to the usability vendor project team. The study protocol and supporting materials were reviewed and approved on 21 June 2019 by Core Human Factors, an independent Institutional Review Board located in Philadelphia, PA (USA), prior to collecting data from participants.
Author contributions
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this manuscript, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published. The authors made all content and editorial decisions and received no financial support or other form of compensation related to the development of this manuscript. All authors had final approval of the manuscript and are accountable for all aspects of the work in ensuring the accuracy and integrity of this manuscript. Authors have full control of all primary data and agree to allow the journal to review the data if requested.
Competing interests: KC and CN are employee and shareholder of Mylan.
Provenance and peer review: Not commissioned; externally peer reviewed.
Authors
Kelly Canham1, BSc Hons
Senior Device Development Manager and Human Factors Lead
Claire Newcomb2, MSc
1Viatris/Mylan Pharma UK Ltd (formerly Mylan Pharma UK Ltd), Suite 13 Science Village, Chesterford Research Park, Cambridge CB10 1XL, UK
2Viatris/Mylan Pharma UK Ltd (formerly Mylan Pharma UK Ltd), Floor 3, Discovery Park House, Discovery Park, Ramsgate Road, Sandwich, Kent CT13 9ND, UK
References
1. Enbrel® (INN-etanercept) [summary of product characteristics]. Puurs, Belgium: Pfizer Manufacturing; 2019.
2. Weinhold T, Del Zotto M, Rochat J, Schiro J, Pelayo S, Marcilly R. Improving the safety of disposable auto-injection devices: a systematic review of use errors. AAPS Open. 2018;4:7.
3. Vermeire S, D’heygere F, Nakad A, Franchimont D, Fontaine F, Louis E, et al. Preference for a prefilled syringe or an auto-injection device for delivering golimumab in patients with moderate-to-severe ulcerative colitis: a randomized crossover study. Patient Prefer Adherence. 2018;12:1193-202.
4. Gandell DL, Bienen EJ, Gudeman J. Mode of injection and treatment adherence: results of a survey characterizing the perspectives of health care providers and US women 18-45 years old. Patient Prefer Adherence. 2019;13:351-61.
5. EN 62366:2015 Medical devices – Part 1: Application of usability engineering to medical devices.
6. Medical & Healthcare Products Regulatory Agency. Human factors and usability engineering – Guidance for medical devices including drug-device combination products. 2021 [homepage on the Internet]. [cited 2021 Feb 11]. Available from: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/645862/HumanFactors_Medical-Devices_v1.0.pdf
7. U.S. Food and Drug Administration. Human factors studies and related clinical study considerations in combination product design and development. Draft guidance for industry and FDA staff. 2016. [homepage on the Internet]. [cited 2021 Feb 11]. Available from: https://www.fda.gov/media/96018/download
|
Author for correspondence: Kelly Canham, BSc Hons, Senior Device Development Manager and Human Factors Lead, Viatris/Mylan Pharma UK Ltd (formerly Mylan Pharma UK Ltd), Suite 13 Science Village, Chesterford Research Park, Cambridge CB10 1XL, UK |
Disclosure of Conflict of Interest Statement is available upon request.
Copyright © 2021 Pro Pharma Communications International
Permission granted to reproduce for personal and non-commercial use only. All other reproduction, copy or reprinting of all or part of any ‘Content’ found on this website is strictly prohibited without the prior consent of the publisher. Contact the publisher to obtain permission before redistributing.