Systematic analysis of injection-site pain and reactions caused by subcutaneous administration of the adalimumab biosimilar FKB327 versus the adalimumab reference product via different delivery methods
Published on 2020/08/03
Generics and Biosimilars Initiative Journal (GaBI Journal). 2020;9(3):108-15, 96.
Author byline as per print journal: Rieke Alten1, MD, PhD; Herbert Kellner2, MD; Malcolm Boyce3, MD; Takuma Yonemura4, MD; Takahiro Ito5, MSc; Mark C Genovese6, MD
|
Introduction/Study objectives: FKB327 is a biosimilar of the adalimumab reference product. Studies in healthy subjects and patients with rheumatoid arthritis demonstrated biosimilarity between FKB327 and the reference product in safety profile, efficacy and immunogenicity. FKB327 formulation excipients differ from the citrate-containing formulation of the reference product, and injection-site pain differences have been reported. The current analysis examines pooled data to assess the amount of injection-site pain resulting from injecting FKB327 using a pre-filled syringe, autoinjector, or vial/syringe versus the reference product. |
Submitted: 1 July 2020; Revised: 31 July 2020; Accepted: 31 July 2020; Published online first: 14 August 2020
Introduction/Study objectives
Adalimumab, a recombinant human monoclonal antibody against tumour necrosis factor-alpha, is indicated for the treatment of patients with rheumatoid arthritis; juvenile idiopathic arthritis; psoriatic arthritis; ankylosing spondylitis; hidradenitis suppurativa; plaque psoriasis; adult and paediatric Crohn’s disease; ulcerative colitis in adults; and non-infectious intermediate, posterior and panuveitis in adult patients [1, 2]. Injection-site reactions (ISRs) are commonly reported with biological therapies [3, 4]. Local ISRs are estimated to affect 12% to 37% of patients treated with adalimumab [5, 6]. Local ISRs include erythema, pruritus, pain, inflammation, rash, induration, itching and oedema [5], potentially causing patients stress, nervousness, reduced quality of life, and a negative impact on willingness to self-administer and adhere to medication [7]. ISRs typically occur during the first month of treatment, last for 3 to 5 days, and commonly resolve without additional therapy [5]. Most ISRs caused by adalimumab are mild to moderate and generally do not require drug discontinuation; but may lead to patient discomfort [2]. Risk factors for having hypersensitivity reactions depend on both the drug and the individual patient characteristics, i.e. disease for which the treatment is indicated, the patient’s immune status, and the concomitant treatments received [4, 8].
Among ISRs, injection-site pain (ISP) is an important element that has been reported with some biologicals due to several factors, including formulation, needle size, anatomic region of injection, and buffer [9]. Patient injection experience may be a significant factor in the selection of biological treatment and in discontinuing therapy to avoid pain and discomfort [10]. Among patients with rheumatoid arthritis, ISP can have an impact on treatment adherence, which is often suboptimal in routine clinical practice [11, 12].
The stability of the formulation of biologicals is improved by using additives such as buffers, amino acids and sugars. An association between ISP and the use of citrate as a buffer has been previously described [9]. Excipients are designated by the regulatory authorities to be inactive and not considered the primary, biologically active ingredient [13]. Excipients in monoclonal antibody therapies are intended to stabilize the active protein from manufacturing until its use by the patient [13]. It has been hypothesized that ISRs associated with products containing excipients are a result of the complement activation-derived inflammatory mediators; degradation of the excipient may be the causative factor [13].
Subsequently, it has been reported that the citrate-containing formulation (CCF) of the adalimumab reference product (RP), which was initially approved in the European Union, the US, and other countries, may be associated with ISP. The formulation excipients of the biosimilar product, FKB327, differ from those of the CCF-RP in that it does not contain citrate, and differing ISP with subcutaneous injection has been reported. FKB327 and the RP are formulated such that the pH of both products is approximately 5.2 [2, 14]. The primary objective of this analysis was to comprehensively characterize ISP and ISRs among patients with moderate-to-severe rheumatoid arthritis and healthy subjects treated with FKB327 compared with CCF-RP. The current analysis also examines pooled data from these studies comparing the amount of ISP resulting from injecting FKB327 using a pre-filled syringe (PFS), an autoinjector (AI), or a vial with regular syringe (RS) versus the CCF-RP.
Methods
Study design
Data were derived from four randomized studies (FKB327-001, -002, -003, and -004) evaluating the efficacy, safety profile and pharmacokinetics (PK) of FKB327 in patients with rheumatoid arthritis and healthy subjects. The studies were conducted from April 2013 to January 2018. The designs of these studies have been previously published [15, 16] and the designs are available at clinicaltrials.gov (FKB327-002 [NCT02260791; EudraCT No.: 2014-000109-11] and -003 [NCT02405780; EudraCT No.: 20140000110-61] studies [dx.doi.org/10.17504/protocols.io.3r2gm8e]).
The studies were conducted in accordance with the Declaration of Helsinki and International Conference on Harmonization Guidelines for Good Clinical Practice. Study protocols were reviewed and approved by an independent ethics committee or institutional review board for each study centre. Written informed consent was obtained from all subjects and patients before study entry.
Studies 001 and 004
Study 001 was a randomized, double-blind, parallel-group phase I study comparing the safety profile and PK in healthy adult subjects after a single-dose of the study drug. Subjects were randomized to receive 40 mg of FKB327, the EU CCF-RP, or the US CCF-RP in a 1:1:1 ratio. The study drug was administered subcutaneously in the abdomen using identical syringes with 25-G 5/8 needles that had been pre-filled by pharmacy staff and labelled such that it was not possible to distinguish between FKB327 and either of the CCF-RPs.
FKB327-004 was a randomized, active-controlled, single-blind, parallel-group phase I clinical pharmacology study evaluating the safety profile and PK in Japanese healthy male subjects after a single-dose of the study drug. Subjects were randomized to receive 40 mg of either FKB327 or the CCF-RP in a 1:1 ratio. FKB327 PFS with a 29-G needle and CCF-RP with 27-G needle were administered subcutaneously in the abdomen. In both studies, PK was assessed by evaluation of serum concentrations of adalimumab. The safety profile was assessed by evaluation of safety laboratory tests, physical examination, vital signs, and electrocardiograms. In both studies, ISP and ISRs were assessed within 30 minutes of subcutaneous dosing and were monitored for 24 hours and 8 days or 4 days after single-dosing, respectively.
Studies 002 and 003
Study 002 was a multicentre, randomized, double-blind, parallel-arm, active-comparator phase III equivalence study to evaluate efficacy and safety profile similarity in patients with active rheumatoid arthritis. Patients were randomized in a 1:1 ratio to receive a 40-mg injection subcutaneously in the abdomen or thigh of either FKB327 (using a vial with RS and 30-G needle) or the CCF-RP (using a PFS and 27-G needle) every other week for 24 weeks.
This was followed by Study 003, a phase III open-label extension study, which consisted of two parts (Period I and Period II). In Period I, patients were rerandomized to receive either FKB327 (administered via PFS with 29-G needle) or CCF-RP (administered via PFS with 27-G needle) for 30 weeks, so that two-thirds of the patients continued the same treatment they had received in the preceding study and one-third received the alternate treatment. In Period II, patients were treated with FKB327 (single arm) for an additional 46 weeks. All patients outside the US were introduced to the FKB327 AI (using a 29-G needle) during Period II of this study.
Assessment of injection-site pain
Pain at the injection site was assessed using a 100-mm visual analogue scale (VAS) score. In all four studies, subjects were asked to determine the extent of their pain by placing a small vertical mark on a horizontal line, with the left endpoint signifying ‘no pain’ and the right endpoint signifying ‘intolerable pain’. Subjects were able to see their previous responses to reduce variability. In the single-dose studies (Studies 001 and 004), assessments of ISP were performed immediately after dosing and at 0.5, 1, 12, and 24 hours post-dose. In the multiple-dose studies (Studies 002 and 003), assessments of ISP were performed within 30 minutes of administration of the first dose.
Assessment of injection-site reactions
For all the studies, study staff applied light pressure at the injection site and recorded any tenderness, erythema and induration. The size of ISRs was measured along the longest axis.
Local reactions were assessed within 30 minutes of dosing according to the US Food and Drug Administration ‘Guidance for industry on skin irritation and sensitization testing of generic transdermal drug products’ as follows [17]:
- 0 = no evidence of irritation
- 1 = minimal erythema, barely perceptible
- 2 = definite erythema, readily visible; minimal oedema or minimal papular response
- 3 = erythema and papules
- 4 = definite oedema
- 5 = erythema, oedema and papules/li>
- 6 = vesicular eruption
- 7 = strong reaction spreading beyond test site.
In the single-dose studies (001 and 004), assessment of ISRs was performed immediately after dosing and at 12, 24, 48, 72 and 96 hours after injection; in addition, assessment of ISRs was performed at 192 hours in Study 001. In the multiple-dose studies (002 and 003), assessments were performed within 30 minutes of administration of the first dose.
Statistical analysis
The analysis set included all the healthy subjects in the single-dose studies (assessment immediately after dosing) and all the patients in the multiple-dose studies (assessment within 30 minutes of dosing) who received the study drug and had ≥ 1 assessments of ISP and/or ISRs. Data from the four studies were pooled to compare FKB327 with the CCF-RP, the FKB327 methods of administration, i.e. PFS, AI, or vial with RS; and injection sites, i.e. abdomen or thigh. The comparison of the ISP VAS scores of FKB327 versus the CCF-RP was performed by using the linear mixed model with 8 fixed effects, consisting of subject population (healthy subject or patient with rheumatoid arthritis), age (< 50 years or ≥ 50 years), gender, race, ethnicity, body weight (< 70 kg or ≥ 70 kg), device (PFS, AI, or vial with RS), and treatment (FKB327 or CCF-RP), as well as two random effects: country and subject.
This analysis also evaluated the impact on ISP of differences in needle gauge (25 G, 27 G, 29 G, and 30 G) used in the various injections. The comparisons between FKB327 presentations and injection sites were performed using the same model. The thresholds of age and body weight were determined based on the median of actual data.
The results of the linear mixed model for the ISP VAS scores were reported with least squares means, 95% confidence intervals (CIs), and p values, where appropriate. The significance level of 5%, i.e. p < 0.05, indicated statistical significance. For Studies 002 and 003 in patients with rheumatoid arthritis, adverse events (AEs) related to ISRs were analysed by exposure-adjusted incident rate (per 100 patient-years) due to different treatment exposures between FKB327 and the CCF-RP, or among FKB327 methods of administration across the studies. All the analyses were performed by using SAS® version 9.1 (Cary, NC, US) or higher.
Results
Patient demographics
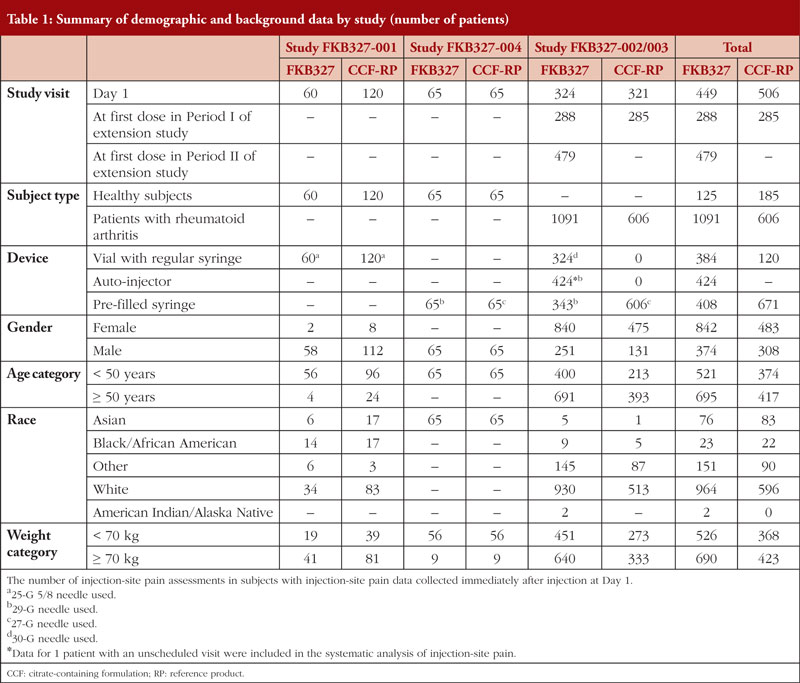
Data analysed included a total of 2,007 assessments in 1,001 subjects and patients. The demographic and background data from the randomized subjects and patients with ISP data are shown by study in Table 1.
Injection-site pain
Injection-site pain in individual studies
In Study 001, immediately after dosing, subjects who received the EU- and US-sourced CCF-RP reported more ISP than subjects who received FKB327. Mean VAS scores were 5.5, 12.9, and 18.4 for the FKB327, EU CCF-RP, and US CCF-RP treatment groups, respectively. At all other time points, the VAS pain scores were similar across the treatment groups.
In Study 004, immediately after dosing, the VAS pain score was also lower in the FKB327 treatment group than in the CCF-RP treatment group. Mean VAS pain scores were 5.2 and 29.8 in the FKB327 and CCF-RP treatment groups, respectively. Thereafter, no substantial differences were observed in the VAS pain scores between the treatment groups.
In Study 002, at Day 1, patients in the FKB327 treatment group reported less pain than patients in the CCF-RP treatment group. Mean VAS scores were 9.3 and 20.2 in the FKB327 and CCF-RP treatment groups, respectively.
In Study 003, at Week 0, patients in the FKB327 treatment group reported less pain than patients in the CCF-RP treatment group. At Week 0 of Study 003, the mean VAS ISP score was slightly higher among patients who received the CCF-RP compared with patients who received FKB327 (12.9 vs 6.2). The mean VAS ISP score was slightly lower in patients who switched from the CCF-RP to FKB327 compared with those who received continuous treatment with the CCF-RP in Studies 002 and 003 (4.9 vs 11.1; see Table 2). Inversely, the mean VAS ISP score was slightly higher in patients who switched from FKB327 to the CCF-RP compared with those who received continuous FKB327 treatment (16.3 vs 6.8). At Week 30 of Study 003, the total mean VAS ISP score was 5.2, without any difference among treatment groups, because all patients were dosed with FKB327 via AI or PFS.
Of the 507 patients who switched to the AI device in Period II of Study 003, 423 patients had an ISP VAS score recorded at the time of switch. The mean VAS score did not change in patients who switched from the PFS device to AI device (F-F-F, 6.7-5.2; RP-F-F, 4.4-5.7), although it decreased in patients who switched from the CCF-RP to FKB327-AI (F-RP-F, 15.4-4.1; RP-RP-F, 7.4-4.5; see Table 3).
Pooled analysis of injection-site pain visual analogue scale score
Because we observed numerical differences in ISP VAS score between patients treated with FKB327 and CCF-RP, we pooled the data from all four studies to increase the number of subjects and assess for statistical significance. A linear mixed model of the ISP VAS score for FKB327 versus the CCF-RP across all four studies showed a 12.6-point improvement (95% CI, –14.3 to –10.8; p < 0.001; see Table 4). The AI showed a 4.4-point lower VAS pain score compared with the RS (95% CI, –5.9 to –2.8; p < 0.001). The AI showed a 1.7-point lower VAS pain score compared with the PFS (95% CI, –3.3 to –0.1; p = 0.035). No statistically significant differences in ISP were identified for gender, age, body weight, population (healthy subject or patient), and injection site (thigh or abdomen). Although the difference in ISP VAS score was significant among races, the majority of subjects and patients were white.
A forest plot of the mean treatment difference of VAS scores showed favourability toward FKB327 over the CCF-RP in most subpopulations except for some countries and races, due to wide variability of 95% CI in a small number of subjects in the subpopulation, see figure 1.
Because of the differences in needle gauge used among the studies, we included needle gauge differences (25 G to 30 G) in the analysis. No statistically significant impact of needle gauge on pain VAS was observed (p = 0.786). Significantly lower pain VAS scores among those treated with FKB327 persisted even when needle gauge was included in the analytical model (–13.1; p < 0.001).
Injection-site reactions
Single-dose studies
In Study 001, the majority of subjects (94.4%) did not experience irritation at the injection site. Of the 10 subjects who experienced minimal erythema immediately post-dose, more subjects in the EU CCF-RP and US CCF-RP treatment groups (5 subjects [8.3%] and 4 subjects [6.7%]) experienced irritation than in the FKB327 treatment group (1 subject [1.7%]). Subjects receiving EU CCF-RP and US CCF-RP experienced more irritation and ISP immediately post-dose than those receiving FKB327. No subjects, including those experiencing minimal erythema immediately post-dose, had any evidence of irritation at subsequent time points (from 12 hours post-dose). In Study 004, the majority of subjects did not show any evidence of local site reactions. Local site reactions were reported slightly more frequently in the CCF-RP treatment group (9 subjects [13.8%]) than in the FKB327 treatment group (3 subjects [4.6%]) throughout the assessment period. In Study FKB327-004, ISR was reported in 12 (9.2%) subjects.
Multiple-dose studies
The number of patients with ISRs and the nature of the reactions are presented in Table 5. A small number of patients with ‘minimal erythema, barely visible reaction’ was reported in the multiple-dose studies (3.1% at Week 0 of Study 002, 4.7% at Week 0 of Period I, and 2.7% at Week 30 of Period II of Study 003). Reports of ‘definitive erythema, readily visible reaction’ were low (0.8% at Week 0 of Study 002, 0.3% at Week 0 of Period I, and 0.7% at Week 30 of Period II of Study 003). No events were judged as ‘definite oedema’, ‘erythema, oedema and papules’, ‘vesicular eruption’, or ‘strong reaction spreading beyond test site’, nor were there any severe ISRs in the FKB327 or CCF-RP treatment groups. Overall, the number of patients with an ISR was very low. No important differences were observed in ISRs between the FKB327 and CCF-RP treatment groups or as a result of switching treatments.
Adverse events related to injection-site reactions
Single-dose studies
In Study 001, injection-site haematoma was one of the most common treatment-emergent AEs. Injection-site haematoma was reported for more subjects in the FKB327 treatment group (n = 4; 6.7%) than in the EU CCF-RP (n = 1; 1.7%) and US CCF-RP (n = 2; 3.3%) treatment groups. In Study 004, ISRs were reported in 3 (4.6%) subjects who received FKB327 and 9 (13.8%) subjects who received the CCF-RP.
Discussion
Advancements in the development of biologicals have provided improvements in local bioavailability and tolerability, as well as in maintaining drug stability against degradation or aggregation [18]. An association between ISP and the use of citrate as a buffer has been previously reported [9]. FKB327 was developed without citrate, which differs from the CCF-RP. In these studies, the adalimumab biosimilar FKB327 was associated with clinically significantly less ISP (12.6-point VAS score improvement) immediately after the first study drug dose than the CCF-RP. It has been reported that the minimum clinically significant difference in VAS pain score on a 100-mm scale is 9 mm to 10 mm, regardless of gender, age, or cause of pain [19, 20]. Therefore, the 12.6-point improvement in VAS pain score in subjects receiving FKB327 is a clinically meaningful change compared with those receiving the CCF-RP. In addition, advancements in the methods of delivery of adalimumab may also result in lower ISP than the standard vial with RS. In these studies, FKB327 delivered via AI showed a 4.4-point lower VAS pain score compared with the vial with RS and a 1.7-point lower VAS pain score compared with the PFS, although the small difference is not considered clinically meaningful [19, 20], while achieving similar PK and safety profiles. This suggests that the AI may be the preferred method of delivery for FKB327 when considering ISP.
In survey-based studies directed toward healthcare providers (HCPs) or consumers, 69% of all HCPs reported a preference for the use of an AI compared with a traditional syringe and needle [21]. HCPs identified benefits for administering injections using an AI, including ease of use, more consistent dosing, faster administration, and fewer needle-stick injuries. In addition, 90% of HCPs thought patients would be more adherent to therapy using an AI, which was supported by 86% of women who reported they would be more likely to adhere to therapy using an AI. A phase II study designed to compare the usability of an etanercept biosimilar via PFS and AI demonstrated that 81.1% of patients preferred the AI, 7.5% preferred the PFS, and 11.3% had no preference [22]. The AI was preferred in terms of all categories investigated, including convenience, ease of use, safety profile, time to administer injection, and decreased pain.
Additional benefits of using an AI include helping patients to overcome needle anxiety [23], allowing for use in patients with functional deficits [24], and improving treatment adherence and persistence [25]. AIs represent an alternative option to oral administration and the requirement for the use of vials and syringes. Benefits of AIs include a design wherein the needle is not visible, ease of use and a low level of associated pain. Together, these benefits of using an AI are thought to promote adherence and persistence of treatment.
A difference in administration between FKB327 and the CCF-RP was the needle gauge used. The FKB327 PFS and AI were manufactured with a 29-G needle, whereas the CCF-RP PFS contained a 27-G needle. Of importance, in the current study, the clinically meaningful differences in pain VAS scores between FKB327 and the CCF-RP persisted when we included needle gauge in the analytical model, demonstrating that needle gauge played no role in the differences in ISP observed between FKB327 and the CCF-RP.
A survey-based study investigating needle performance and patient preference for administration of glatiramer acetate for multiple sclerosis treatment revealed that significantly fewer patients reported problems after 30 days of use (including fewer injection-site experiences) and more patients preferred the 29-G needle overall compared with a 27-G needle [26]. In a study investigating 30-G needles versus 32-G needles for injection of botulinum toxin type A, the average injection pain scores were nominally but not significantly different in the arm or the face [27]. Furthermore, there were no clinically significant differences in pain associated with needle type following arm injections, with no significant differences in the character of clinically important pain. A Cochrane review demonstrated low-quality evidence that a wide needle (23 G) may slightly reduce pain associated with a vaccination procedure in 1,135 healthy infants compared with a narrow needle (25 G); however, these differences were thought to be too small to be of practical importance [28]. In a randomized crossover comparison of ISP with 40 mg/0.4 mL (29-G needle) or 40 mg/0.8 mL (27-G needle), significantly lower injection-related pain immediately after injection was reported for the 40 mg/0.4 mL formulation [29]. However, it is unclear whether the pain reduction was most attributable to differences in composition, volume and/or needle size.
In the current study, significantly decreased ISP was reported with FKB327 compared with CCF-RP, which may be due to the citrate buffer that is present in CCF-RP but not in FKB327, as an association between citrate buffer and ISP has been demonstrated previously [9]. Results have shown that a citrate-free formulation of the RP was associated with significantly decreased ISP [29, 30]. Therefore, the results of the current study support previous findings demonstrating an association between a decreased level of ISP and citrate-free formulations.
This study has a number of limitations. The systematic analysis presented requires combining data/events across a number of different studies, which may introduce bias. Moreover, each study used different patients, sites and administration devices such that the results of the systematic analysis may not be as valid as the individual results from each trial. Some of the studies were of short duration, and the concern over pain from a single injection may be different from ongoing pain associated with longer-term administration. The timing of ISP assessment may also limit the conclusions of this analysis based on variability of measurement immediately after injection (Studies 001 and 004) or within 30 minutes after injection (Studies 002 and 003), resulting in the potential for pain resolution within this time frame.
Conclusions
FKB327 (citrate-free formulation adalimumab biosimilar) showed clinically significantly less ISP compared with the CCF-RP.
No statistically significant differences were observed in ISRs or AEs related to ISRs between FKB327 and the CCF-RP or among FKB327 methods of administration. The lower ISP score observed with FKB327 delivered via an AI compared with a vial and RS or a PFS suggests FKB327, especially when delivered via AI, can result in less ISP than the CCF-RP. This is important, because ISRs and ISP can interfere with patients adhering to injectable medications as prescribed.
For patients
Biosimilars are drugs that act in a similar way compared with the reference product but may have small differences in ingredients. Drug ingredients and needle size may affect the amount of injection-site pain that is experienced when the drug is injected into the skin. This paper tested the difference in injection-site pain between the biosimilar drug, FKB327 and the reference product, and differences in pain with a pre-filled syringe, an autoinjector, and a vial/syringe. The study found that patients reported lower pain scores with FKB327 compared with the reference product, and lower pain scores were reported with the autoinjector. These findings mean that receiving FKB327 injections with an autoinjector leads to lower amounts of pain and side effects that may help to make it easier to continue treatment with this drug.
Clinical trials registration
FKB327-001 – EU Clinical Trials, EudraCT No.: 2012-005140-23, protocols.io, registered 12 July 2019, www.protocols.io/view/systemic-analysis-of-injection-site-pain-caused-by-3r2gm8e/abstract FKB327-002 – National Institutes of Health (NIH) US National Library of Medicine, NCT02260791, prospectively registered 29 July 2014, https://clinicaltrials.gov/ct2/show/NCT02260791 FKB327-003 – NIH US National Library of Medicine, NCT02405780, prospectively registered 17 March 2015, https://clinicaltrials.gov/ct2/show/NCT02405780 FKB327-004 – protocols.io, registered 12 July 2019, www.protocols.io/view/systemic-analysis-of-injection-site-pain-caused-by-3r2gm8e/abstract
Funding sources
Funding for the FKB327-001 (EudraCT No.:2012-005140-23), FKB327-002 (NCT02260791), FKB327-003 (NCT02405780), and FKB327-004 studies was sponsored by Fujifilm Kyowa Kirin Biologics Co Ltd. Technical, editorial and medical writing assistance was provided under the direction of the authors by The Lynx Group LLC. Funding for this support was provided by Mylan Inc.
Author contributions
RA, HK, MG: substantial contributions to the conception and design of the work; RA, HK, MB, TY, TI, MG: the acquisition, analysis and interpretation of data; RA, MB: drafted the work or substantively revised it; RA, HK, MB, TY, TI, MG: approved the submitted version of the manuscript. The authors made all content and editorial decisions and received no financial support or other form of compensation related to the development of this manuscript. All authors had final approval of the manuscript and are accountable for all aspects of the work in ensuring the accuracy and integrity of this manuscript. Authors have full control of all primary data and agree to allow the journal to review the data if requested.
Declarations
Ethics approval and consent to participate
The studies were conducted in accordance with the Declaration of Helsinki and International Conference on Harmonization Guidelines for Good Clinical Practice. Written informed consent was obtained from all subjects and patients before study entry. Study protocols were reviewed and approved by an independent ethics committee or institutional review board (IRB) for each study centre (protocol no. FKB327-001, EudraCT No.: 2012-005140-23; approved 12 March 2013, by Scotland A Research Ethics Committee; protocol no. FKB327-002, EudraCT No.: 2014-000109-11/NCT02260791, approved 25 November 2014, by United States of America Quorum IRB [central IRB for California sites in US], approval no. 29659; protocol no. FKB327-003, EudraCT No.: 2014-000110-61/NCT02405780, approved 25 November 2014, by United States of America Quorum IRB, approval no. 30276; approval was obtained for each of the study sites – 109 sites in -002 study and 92 sites in -003 study; protocol no. FKB327-004, approved 18 December 2015, by Hakata Clinic IRB).
Competing interests: RA has received research grants and consultant fees from Fujifilm Kyowa Kirin Biologics Co Ltd. RA has received consultant fees from Mylan Inc and has been a paid speaker for the Speakers Bureau of Mylan Inc. HK has nothing to disclose. MB has nothing to disclose. TY received a grant from Fujifilm Kyowa Kirin Biologics Co Ltd during the conduct of this study. TI is an employee of Fujifilm Kyowa Kirin Biologics and reports personal fees from Fujifilm Kyowa Kirin Biologics Co Ltd during the conduct of this study. MG has received consultant fees from Fujifilm Kyowa Kirin Biologics Co Ltd, including for the design of this trial.
Provenance and peer review: Not commissioned; externally peer reviewed.
Authors
Rieke Alten1, MD, PhD
Herbert Kellner2, MD
Malcolm Boyce3, MD
Takuma Yonemura4, MD
Takahiro Ito5, MSc
Mark C Genovese6, MD
1University Medicine Berlin, Berlin, Germany
2Specialist Practice in Rheumatology and Gastroenterology, Munich, Germany
3Hammersmith Medicines Research, London, UK
4Souseikai Sumida Hospital, Tokyo, Japan
5Fujifilm Kyowa Kirin Biologics, Tokyo, Japan
6Stanford University Medical Center, Palo Alto, CA, USA
References
1. European Medicines Agency. Humira (adalimumab). 2019 [homepage on the Internet]. [cited 2020 Jul 31]. Available from: ww.ema.europa.eu/en/medicines/human/EPAR/humira
2. Abbott Laboratories. HUMIRA (adalimumab) [prescribing information]. North Chicago, IL: AbbVie Inc; 2019.
3. Nakamizo S, Miyachi Y, Kabashima K. Addition of cyclosporine to adalimumab improved psoriasis and adalimumab-induced injection site reaction. Indian J Dermatol. 2014;59(5):522-3.
4. Corominas M, Gastaminza G, Lobera T. Hypersensitivity reactions to biological drugs. J Investig Allergol Clin Immunol. 2014;24(4):212-25.
5. Sator P. Safety and tolerability of adalimumab for the treatment of psoriasis: a review summarizing 15 years of real-life experience. Ther Adv Chronic Dis. 2018;9(8):147-58.
6. Mease PJ. Adalimumab in the treatment of arthritis. Ther Clin Risk Manag. 2007;3(1):133-48.
7. Matsui T, Umetsu R, Kato Y, Hane Y, Sasaoka S, Motooka Y, et al. Age-related trends in injection site reaction incidence induced by the tumor necrosis factor-α (TNF-α) inhibitors etanercept and adalimumab: the Food and Drug Administration adverse event reporting system, 2004-2015. Int J Med Sci. 2017;14(2):102-9.
8. Murdaca G, Spanò F, Puppo F. Selective TNF-α inhibitor-induced injection site reactions. Expert Opin Drug Saf. 2013;12(2):187-93.
9. Laursen T, Hansen B, Fisker S. Pain perception after subcutaneous injections of media containing different buffers. Basic Clin Pharmacol Toxicol. 2006;98(2):218-21.
10. Bolge S, Eldridge H, Doshi D, Ellis L, Roland B, Woelfel J. Patient satisfaction and experience with golimumab, adalimumab, and etanercept for the treatment of rheumatoid arthritis, psoriatic arthritis, and ankylosing spondylitis. 2012 ACR/ARHP Annual Meeting; 9-14 Nov 2012; Washington, D.C.
11. Betegnie AL, Gauchet A, Lehmann A, Grange L, Roustit M, Baudrant M, et al. Why do patients with chronic inflammatory rheumatic diseases discontinue their biologics? An assessment of patients’ adherence using a self-report questionnaire. J Rheumatol. 2016;43(4):724-30.
12. Gely C, Marin L, Gordillo J, Mañosa M, Bertoletti F, Cañete F, et al. N032 Impact of pain due to subcutaneous administration of a biological drug. J Crohns Colitis. 2018;12(1):S582-3.
13. Singh SK, Mahler HC, Hartman C, Stark CA. Are injection site reactions in monoclonal antibody therapies caused by polysorbate excipient degradants? J Pharm Sci. 2018;107(11):2735-41.
14. HULIO (adalimumab-fkjp) [prescribing information]. Morgantown, WV: Mylan Pharmaceuticals Inc; 2020.
15. Puri A, Niewiarowski A, Arai Y, Nomura H, Baird M, Dalrymple I, et al. Pharmacokinetics, safety, tolerability and immunogenicity of FKB327, a new biosimilar medicine of adalimumab/Humira, in healthy subjects. Br J Clin Pharmacol. 2017;83(7):1405-15.
16. Al-Salama ZT. FKB327: an adalimumab biosimilar. BioDrugs. 2019;33(1):113-6.
17. U.S. Food and Drug Administration. Guidance for industry on skin irritation and sensitization testing of generic transdermal drug products. 3 February 2000 [homepage on the Internet]. [cited 2020 Jul 31]. Available from: www.federalregister.gov/documents/2000/02/03/00-2299/guidance-for-industry-on-skin-irritation-and-sensitization-testing-of-generic-transdermal-drug
18. Elgundi Z, Reslan M, Cruz E, Sifniotis V, Kayser V. The state-of-play and future of antibody therapeutics. Adv Drug Deliv Rev. 2017;122:2-19.
19. Kelly AM. Does the clinically significant difference in visual analog scale pain scores vary with gender, age, or cause of pain? Acad Emerg Med. 1998;5(11):1086-90.
20. Powell CV, Kelly AM, Williams A. Determining the minimum clinically significant difference in visual analog pain score for children. Ann Emerg Med. 2001;37(1):28-31.
21. Gandell DL, Bienen EJ, Gudeman J. Mode of injection and treatment adherence: results of a survey characterizing the perspectives of health care providers and US women 18–45 years old. Patient Prefer Adherence. 2019;13:351-61.
22. Rho YH, Rychlewska-Hańczewska A,Śliwowska B, Kim TH. Usability of prefilled syringe and autoinjector for SB4 (an etanercept biosimilar) in patients with rheumatoid arthritis. Adv Ther. 2019;36(9):2287-95.
23. Phillips JT, Fox E, Grainger W, Tuccillo D, Liu S, Deykin A. An open-label, multicenter study to evaluate the safe and effective use of the single-use autoinjector with an Avonex® prefilled syringe in multiple sclerosis subjects. BMC Neurol. 2011;11:126.
24. Freundlich B, Kivitz A, Jaffe JS. Nearly pain-free self-administration of subcutaneous methotrexate with an autoinjector: results of a phase 2 clinical trial in patients with rheumatoid arthritis who have functional limitations. J Clin Rheumatol. 2014;20(5):256-60.
25. Xie L, Zhou S, Wei W, Gill J, Pan C, Baser O. Does pen help? A real-world outcomes study of switching from vial to disposable pen among insulin glargine-treated patients with type 2 diabetes mellitus. Diabetes Technol Ther. 2013;15(3):230-6.
26. Glenski S, Conner J. 29 gauge needles improve patient satisfaction over 27 gauge needles for daily glatiramer acetate injections. Drug Healthc Patient Saf. 2009;1:81-6.
27. Alam M, Geisler A, Sadhwani D, Goyal A, Poon E, Nodzenski M, et al. Effect of needle size on pain perception in patients treated with botulinum toxin type A injections: a randomized clinical trial. JAMA Dermatol. 2015;151(11):1194-9.
28. Beirne PV, Hennessy S, Cadogan SL, Shiely F, Fitzgerald T, MacLeod F. Needle size for vaccination procedures in children and adolescents. Cochrane Database Syst Rev. 2018;8(8):CD010720.
29. Nash P, Vanhoof J, Hall S, Arulmani U, Tarzynski-Potempa R, Unnebrink K, et al. Randomized crossover comparison of injection site pain with 40 mg/0.4 or 0.8 mL formulations of adalimumab in patients with rheumatoid arthritis. Rheumatol Ther. 2016;3(2):257-70.
30. Yoshida T, Otaki Y, Katsuyama N, Seki M, Kubota J. New adalimumab formulation associated with less injection site pain and improved motivation for treatment. Mod Rheumatol. 2019;29(6):949-53.
|
Author for correspondence: Rieke Alten, MD, PhD, Head of Department of Internal Medicine II, Professor of Medicine, Director of Rheumatology Research Center, Rheumatology, Clinical Immunology, Osteology, Physical Therapy and Sports Medicine, Schlosspark-Klinik Charité, University Medicine Berlin, 2 Heubnerweg, DE-14059 Berlin, Germany |
Disclosure of Conflict of Interest Statement is available upon request.
Copyright © 2020 Pro Pharma Communications International
Permission granted to reproduce for personal and non-commercial use only. All other reproduction, copy or reprinting of all or part of any ‘Content’ found on this website is strictly prohibited without the prior consent of the publisher. Contact the publisher to obtain permission before redistributing.