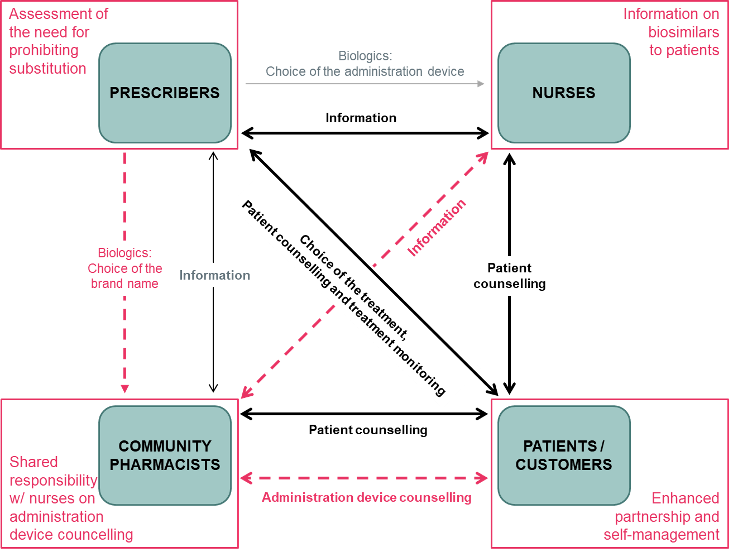
Effective and safe handling of pre-filled syringe (PFS) for administration of biosimilar candidate AVT06 (aflibercept) in patients with chorioretinal vascular diseases
Introduction: Chorioretinal vascular diseases are among the leading causes of irreversible blindness in industrialized countries. The prognosis of chorioretinal vascular diseases has been largely improved with the introduction of the vascular endothelial growth factor (VEGF) inhibitors, biological drugs which have become the first line therapy for patients with these conditions. The development of biosimilars may […]