Do pricing and usage-enhancing policies differ between biosimilars and generics? Findings from an international survey
Published on 2017/03/31
Generics and Biosimilars Initiative Journal (GaBI Journal). 2017;6(2):79-88.
|
Introduction/Study objective: This paper aims to survey the policies implemented by European countries for pricing and promoting the use of biosimilar medicines and to explore similarities and differences with policies for generic medicines. |
Submitted: 30 March 2017; Revised: 26 May 2017; Accepted: 26 May 2017; Published online first: 9 June 2017
Introduction/Study objective
Public payers are concerned with ensuring patient access to medicines, particularly in the light of increasing pressure on budgets and the market entry of new, high-priced medicines [1]. One opportunity to generate savings and thus free resources for further investments in health is increased uptake of lower-priced medicines, such as generics. The use of generic medicines has been recommended by the World Health Organization (WHO) [2] and policymakers employ a range of supply- and demand-side tools to increase their uptake. These include generics substitution, physicians prescribing by the International Nonproprietary Name (INN) rather than the brand name, a reference price system, i.e. fixed reimbursement within a cluster of identical and similar medicines, and awareness-raising campaigns [3–9]. The ability of generics-promoting policies to reduce medicine prices and generate savings for health care has been well documented [10, 11].
Since biological medicines also significantly contribute to the pharmaceutical bill, policymakers are awaiting the entry of biosimilar medicines [12, 13], which are expected to generate substantial savings [14]. Recent years have seen several examples of tendering for biosimilar medicines successfully reducing prices [15, 16].
Under the Platform on Access to Medicines in Europe of the Corporate Social Responsibility Process, a multi-stakeholder working group was dedicated to biosimilar medicines. The working group produced a European Commission Consensus Information Document agreed by all stakeholders represented, the document provided key information about biosimilar medicines in order to foster stakeholders’ understanding of biosimilars [17]. However, the working group did not investigate which pricing and usage-enhancing policies European Union Member States applied for biosimilar medicines.
While there is good evidence of the implementation of pricing and demand-side measures for generics in Europe [3–11], the policies that European countries have been implementing to deal with biosimilar medicines are comparatively less known. To the best of the authors’ know ledge, there are few studies in the literature that provide information about pricing and demand-side policies for biosimilar medicines and the only comparative exercise performed across a large number of countries was published by the European Biopharmaceutical Enterprises in 2015 [18]. While the authors recognize the importance of this study, it did not fully explore all aspects of biosimilar medicines policies. Further studies that investigate biosimilar medicines policies are limited to a few countries [19–21] and/or to a single policy [22]. Furthermore, the findings of these studies differ.
Against this backdrop, this manuscript aims to survey the pricing and usage-enhanced policies that different countries, in particular in the European region, have implemented for biosimilar medicines, and to explore whether these policies differ from generic medicines policies.
Methods
We conducted a survey with the members of the Pharmaceutical Pricing and Reimbursement Information (PPRI) network [23]. This is a network of competent authorities for pharmaceutical pricing and reimbursement in 46 countries, thereof 43 European countries. It should be noted that European countries are those as defined by WHO [24], and thus include countries such as Israel, Kazakhstan and Kyrgyzstan.
We prepared questions about the status of generic and biosimilar medicines policies. We explored the pricing policies for generics and biosimilars, in particular regarding possible regulation linking the generic and/or biosimilar price to the originator price. We surveyed whether INN prescribing and substitution by generic and biosimilar medicines was permitted, and whether it was mandatory. We also aimed to identify further specific pricing policies, e.g. tendering.
While the focus of this survey was on policies for biosimilar medicines, we also aimed to survey, or validate, information on gen eric medicines policies in order to explore possible differences between policies for the two medicine groups.
As far as possible, we pre-filled the questionnaire with information available to us, through previous research and literature review. This was predominantly only possible in the field of generics. Respondents from the competent authorities were invited to provide, or validate, information on biosimilar and generic medicines policies valid in the first quarter of 2016.
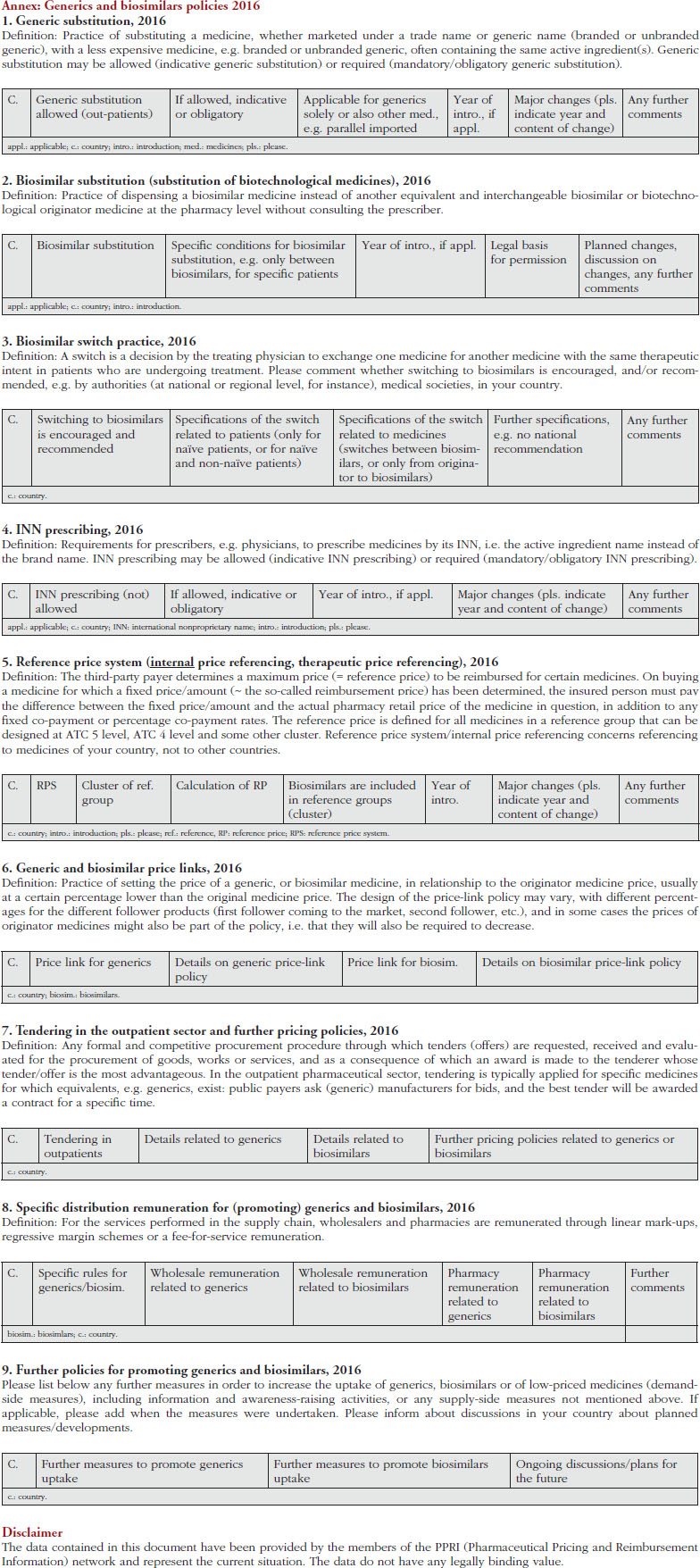
We sent the survey to the PPRI network members on 7 January 2016, requesting their responses by 19 January 2016. A friendly reminder was sent before the deadline, and a further personalized reminder that was focused on key questions was sent on 11 February 2016. Preliminary results were presented and discussed during a meeting with PPRI network members on 28 April 2016, where any misunderstandings could be clarified. In response to this discussion, we created a revised version of the questionnaire, which was circulated for validation on 30 May 2016. During the survey, respondents were encouraged to reply and clarification was sought in the case of answers that raised additional questions. On 1 August 2016, the survey was officially closed and the results were shared with participants. An uncompleted version of the revised questionnaire, i.e. without pre-filled answers, is available in the Annex.
While this survey with the PPRI network was the key survey tool, where considered appropriate we added relevant information from the literature (indicated by references).
Results
Response rate
We received responses from 36 of the 43 European PPRI members, as well as Canada and South Africa. Replies from the European region were provided by 25 of the 28 EU Member States (no data received from Ireland, Italy and Luxembourg) plus Albania, Belarus, Iceland, Israel, Kazakhstan, Kyrgyzstan, Norway, Russia, Serbia, Turkey and Ukraine. Data from the missing three EU Member States and Switzerland were added, wherever possible, from literature and previous PPRI network queries on related topics. As a result, this manuscript includes information from 40 European countries, Canada and South Africa.
Pricing policies
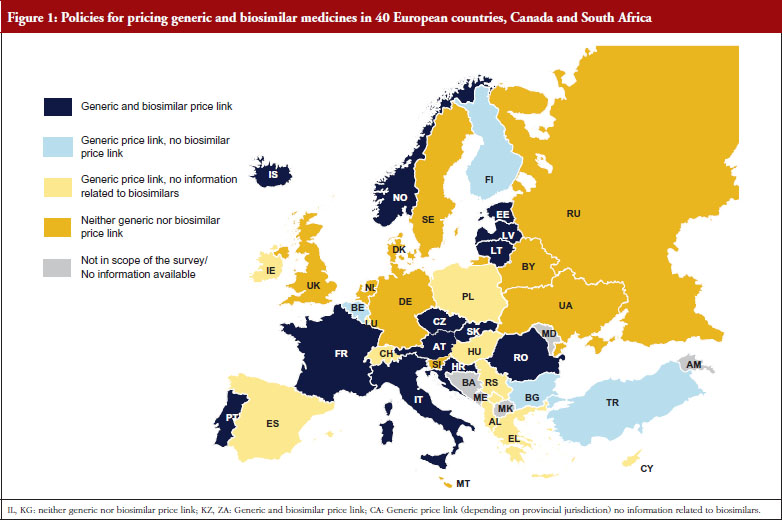
Several countries apply a pricing policy that sets the price of follower products in relation to the price of the originator medicine. For generics, this is called ‘generic price link’. It is a commonly used practice that is applied in 30 of the 42 surveyed countries. Fifteen countries reported that they also apply such a strategy for biosimilar medicines. These are Austria, the Baltic States, Croatia, the Czech Republic, France, Iceland, Italy, Kazakhstan, Norway, Portugal, Romania, Slovakia and South Africa. Four ‘generic price link’ countries (Belgium, Bulgaria, Finland and Turkey) informed us that they do not apply a price-link policy for biosimilars. A further 10 European countries and Canada apply a generic price link but did not report whether they also use this policy for pricing biosimilar medicines.
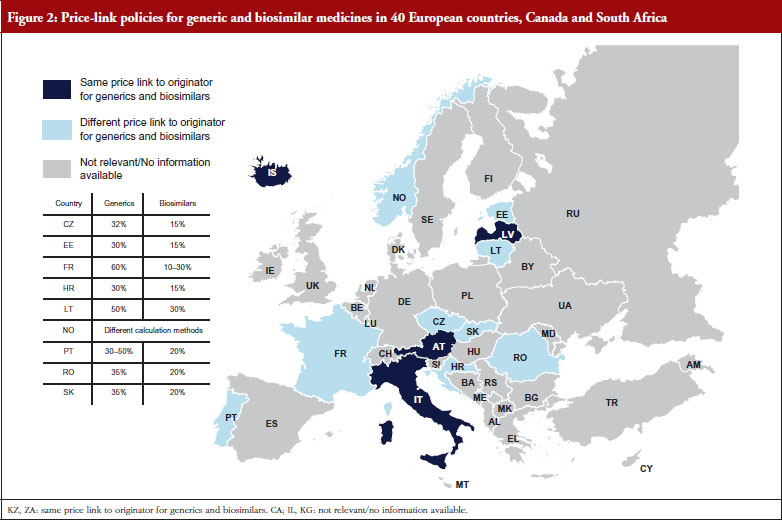
In most of the countries that apply a generic and biosimilar price link, the price difference between the originator medicine and the biosimilar is, sometimes considerably, lower than that between originator and generic medicine. This implies that biosimilar medicines tend to have higher prices. For instance, in the Czech Republic the first generic drug must be priced 32% below that of the originator, whereas the price of the first biosimilar must only be 15% lower than the originator. Six countries (Austria, Iceland, Italy, Kazakhstan, Latvia and South Africa) apply the same price link for generic and biosimilar medicines. However, the design of this link is heterogeneous. In Italy for example, a Decree was passed that treats generics and biosimilars in the same way in the procedure of reimbursement. Both can automatically be reimbursable and classify for the same reference group as their originator, if the price proposed by the respective marketing authorization holder is favourable to the Italian Health Service. Austria (at the time of the survey, for information on further developments see the Discussion paragraph) and Latvia, on the contrary, have defined a percentage threshold under which the first follower – either generic or biosimilar – must be priced (48% and 30%, respectively), and percentage rates of how much the prices of further ‘followers’ must be lower than of previous generics or biosimlars. In Iceland, the price link is calculated based on the maximum wholesale price allowed for generic and biosimilar medicines. Figures 1 and 2 describe price-link policies for biosimilars and generics, including price differences.
Some countries in the survey described the use of tendering to procure biosimilars. Iceland and the UK for instance have been tendering for medicines, including biosimilars, in the inpatient sector. In Denmark, all medicines (including biosimilars) for the inpatient sector are procured by a national procurement agency (AMGROS) which is owned by the five ‘health regions’ [25]. The Norwegian Drug Procurement Cooperation is responsible for purchasing medicines for public hospitals through annual tender processes. To ensure the acceptance of the awarded products, the results of the tender process and recommendations are presented by an expert group to affected stakeholders (industry, patient organizations, doctors) [26]. A similar approach is applied in Italy, however, in a decentralized manner; 20 Italian Regional Health Authorities (RHA) are responsible for planning healthcare services and allocating financial resources. All RHAs have established an organization for purchasing goods and services and two of them (Emilia-Romagna and Tuscany) additionally appointed a separate authority for procuring medicines. Various tenders for off-patent biologicals are conducted at regional levels [27]. In Spain, a pilot project of centralized procurement was reported to have taken place for the glycoprotein hormone and anaemia treatment erythropoietin (EPO).
Tendering in the outpatient sector is particularly used to procure for ‘public functions’, e.g. vaccines, centralized procurement in emergency situations such as pandemics [28]. Cyprus and Malta, both countries in which pharmaceutical services are provided separately by a public and a private sector, procure medicines (including biosimilars) for the public sector through tendering [29–31]. Some European countries have introduced tenders or tender-like systems in the outpatient sector; public payers launch tender calls for medicines that have generic (same active ingredient) or therapeutic alternatives. The lowest bidder will be either warded the contract to supply the whole market or will be granted a preferential position on the reimbursement list, e.g. through higher coverage. Such a policy is applied in The Netherlands, where through the ‘preferential pricing policy’ health insurers tender for the lowest-priced off-patent medicine [28, 32, 33]. However, biosimilar medicines were only recently included in the tenders, and only by a limited number of insurers [25]. A tender-like procedure is also applied in the Danish off-patent outpatient market, which also includes biosimilar medicines. In their system, pharmaceutical companies submit bi-monthly price bids and the lowest-priced medicines are selected for full reimbursement within a two-week period [25, 33–36].
Demand-side measures to encourage biosimilar uptake
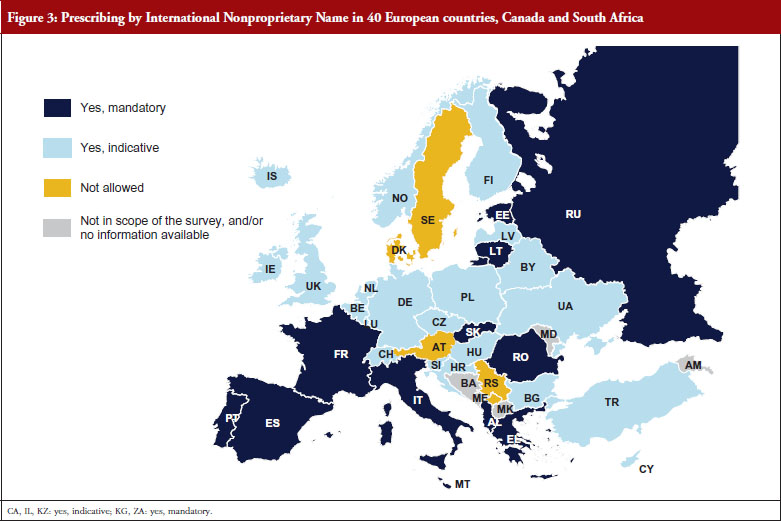
Prescribing by INN is a measure enforced by doctors that supports the uptake of generics as well as biosimilars. INN prescribing is in place in 35 European countries, Canada and South Africa, and is mandatory in 14 of the surveyed countries. It is only in Austria, Denmark, Serbia and Sweden that prescription by INN is not permitted, see Figure 3.
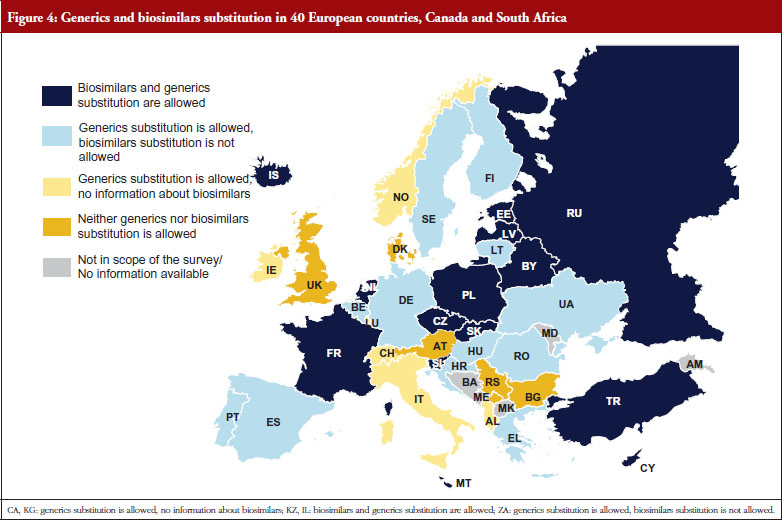
Another key demand-side measure to enhance the uptake of off-patent medicines is to allow community pharmacists to substitute the originator medicine with an off-patent medicine. Generics substitution is a commonly used practice. It is applied in 37 of the 42 countries (it is not permitted in Austria, Bulgaria, Denmark, Luxembourg and Serbia), and mandatory in 15 countries. In contrast, substitution of biosimilar medicines is only in place in some, mainly Central and Eastern European countries: Belarus, Cyprus, Czech Republic, Estonia, France, Iceland, Israel, Kazakhstan, Latvia, Malta, The Netherlands, Poland, Russia, Slovakia, Slovenia and Turkey. In some countries, e.g. Latvia, the substitution of an originator medicine by a biosimilar has not been explicitly implemented, but INN prescribing is obligatory and the medicine of the lowest price must be dispensed in the pharmacy, e.g. Latvia. There is usually no specific legal basis for biosimilar substitution; no law explicitly prohibited biosimilar substitution. In France, biosimilar substitution was introduced by the Social Insurance Law at the beginning of 2014 but is not yet in practice. Other countries reported opposition to biosimilar substitution by some stakeholders. In the Czech Republic for example, the Chamber of Pharmacists recommended against biosimilar substitution.
Discussion
Pricing and reimbursement policies for biosimilar medicines, as for generics, are embedded in the overall pricing and reimbursement framework. Policies for biosimilar medicines might be expected to be similar to those for generics, yet our survey showed that this is the case for only some policies and varies by country.
With regard to pricing of generic and biosimilar medicines, there are, in principle, two different approaches: to allow free pricing for off-patent medicines, and to allow for competition. The incentive for pharmaceutical manufacturers to offer low prices is to achieve higher market shares, since the lowest-priced medicines are likely to obtain more public funding and/or be recommended to pharmacists, doctors and patients. Reimbursement strategies, such as a reference price system or tendering for off-patent medicines, likely support competition.
An alternative policy is price regulation, typically in the form of a price link, whereby the price of a generic or biosimilar medicine is determined in relation to the originator price. This pricing policy appears to be commonly applied for generic medicines, even in combination with external price referencing (international price comparison) in several countries including Belgium, Hungary, Poland and Spain. In some countries, a price-link policy is applied for biosimilars as well. All countries that apply the price-link policy for biosimilar medicines do the same for generics. The survey showed, however, that several countries with a price-link policy for generics do not have one for biosimilar medicines. While four generic price link countries explicitly advised that they do not apply a price link for biosimilars, other countries with a generic price-link policy did not respond to the question about the use of price linkage for biosimilars. This suggests that legislation on this issue has not yet been decided, likely due to the novelty of the topic.
In 2016, a few countries, e.g. Austria, South Africa, did not apply specific pricing regulation for biosimilar medicines. They used the same procedures for all off-patent medicines, whether or not these medicines were generics or biosimilars (in Austria, for instance, legislation valid at the time of the survey referred only to ‘follower products’). Furthermore, the required price difference between the originator and follower medicine did not distinguish between biosimilars and generics in these countries. In most countries, however, the price difference was lower for the originator-biosimilar pair compared to the originator-generic pair. This indicates that competent authorities in these countries grant comparatively higher prices to biosimilar medicines.
Few large-scale price comparisons include biosimilar medicines, and therefore information about the impact of the two approaches for pricing biosimilars (price link versus competition) is not available. For generics, an illustrative study of selected active ingredients [37] showed that countries that base their pricing policy for generic medicines on competition tend to have a larger price difference between the originator and the generic medicine, and generics prices are often (but not consistently) lower [7, 38–41].
Overall, tendering appears to be an effective instrument to generate savings for public payers. Norway for example, reported huge discounts for biosimilar infliximab (minus 72% in 2015) [26]. Research into biosimilar tenders by regions of Italy revealed that for 191 analyzed lots referring to three off-patent biologicals (somatropin, epoetin and filgrastim) mentioned in 24 tenders performed between 2008 and 2012, the price of filgrastim and epoetin dropped considerably, whereas the price of somatropin remained steady. Somatropin had the lowest mean number of competitors (1.16), while filgrastim had the highest (2.75) [16]. Both Norway and Italy applied tenders targeted at biological and biosimilar medicines in the hospital sector. This is in line with the results of the EBE study on pricing and reimbursement policies for biological medicines, which showed that, while biological medicines are subject to tenders in several European countries, these are hospital tenders in the majority of cases [18]. Despite evidence of its effectiveness, tendering has rarely been applied for biosimilar medicines in the outpatient sector. Dutch health insurers are experienced in tendering for off-patent medicines but have traditionally refrained from including biosimilars in their tenders [42, 43]. Only recently have some Dutch insurers started launching tenders for biosimilars [25]. According to Dutch respondents, this is to be seen in the light of physician reluctance towards biosimilar medicines; while switching from the originator to a biosimilar medicine is allowed and would be appreciated by the competent authorities, it is not yet common practice.
However, pricing is only one aspect of encouraging generics and biosimilars use. Policies are also required that ensure the use of lower-priced medicines instead of higher-priced originator medicines. It is of key importance that patients trust generics and biosimilars; otherwise, they will insist on receiving the originator medicines even if they must pay more. Health professionals such as doctors and pharmacists play a key role in this respect, as their contributions in some countries such as Germany and Norway have shown [15, 44]. Health professionals must themselves understand the value of generics and biosimilars in order to communicate it to the patients. Thus, education and possibly incentives for health professionals are needed [45].
Much debate has centred around interchangeability and switching from originator medicines to biosimilars. Recent studies have been launched to prove the safety of switches [46], such as the NOR-SWITCH study, whose preliminary results suggest that a switch from originator infliximab to biosimilar infliximab is safe [47].
The Australian government announced in early 2015 that biosimilar medicines can be substituted by pharmacists based on the clinical recommendations of the Pharmaceutical Benefits Advisory Committee. The same rules that apply to generics also hold for biosimilars, and pharmacists are permitted to substitute any biosimilar medicine for an originator product, in the absence of clinical evidence to the contrary [48]. In Europe, however, biosimilar substitution has not been widely implemented, despite advanced generic medicines substitution. Countries that allow biosimilar substitution (or, at least, do not explicitly prohibit it) have been confronted with opposition by pharmacists and doctors.
It is important to note the limitations to this survey study. It concerns a new area for which data are scarce, and knowledge is limited. Since in many cases the literature does not provide conclusive information, we used primary data from members of the PPRI network, who are experts in the field of pharmaceutical pricing and reimbursement. However, we experienced a lower response rate to some of the specific questions related to biosimilar medicines. This could be a reflection of the novelty of the area for which specific policies are yet to be defined. Due to the novelty of the topic, terminology was not fully clear to all respondents, e.g. the distinction between switching by doctors and substitution by pharmacists. We addressed these challenges by providing definitions, arranging a debate of preliminary findings during a face-to-face meeting and organizing a second round of the survey based on a slightly revised questionnaire. We also aimed to validate responses using the literature, although this was not possible in several cases due to a lack of information or contradictions between sources, e.g. information on biosimilar substitution. This makes it difficult for us to discuss our findings in the light of existing literature. Further, our findings are not comprehensive. For instance, we did not survey the ‘switch climate’ or regulations for switches in different countries, as we felt that existing research had already covered these areas. The findings refer to the situation at the time of the survey (Spring 2016); in the meantime, changes in legislation might have occurred (as with the price-link policy in Austria, for instance, when in April 2017 different percentage rates for the price difference of the originator-biosimilar pair and of the originator-generic pair were introduced). Finally, this research is descriptive, and does not assess the possible impacts of policies on biosimilar prices or uptake.
Despite these limitations, we believe that our study provides interesting and updated results. Apart from the EBE study published in the same journal [18], this is the sole recent study that considers biosimilar and generic medicines policies across a large number of countries, including several non-European nations. The study also provides value by surveying both generic and biosimilar medicines, allowing a comparison between these two groups of medicines. Such a comparison has not been provided by past studies, including the above-mentioned [18], which only focused on biological and biosimilar medicines. The broad focus of our study (generic and biosimilar medicines policies) offers novel information. Due to the novelty of the topic, it is difficult to compare our findings with those of other research.
Conclusion
This study provides information about pricing policies and demand-side measures to enhance the uptake of biosimilar medicines for over 40 countries and compares them to practices applied for generics. Some aspects surveyed here have not been previously discussed in the literature.
Overall, the study shows that European countries have made good use of available policies for pricing generics and enhancing their uptake. However, with regard to biosimilar medicines, policymakers in several countries appear to be struggling to identify the most appropriate approach. Indeed, in many countries, pricing and usage-enhancing policies for biosimilars have not yet been defined. Policymakers do not always apply instruments that have been successfully implemented for generics to biosimilar medicines. The reluctance to do so might result from opposition to biosimilar medicines expressed by some stakeholder groups, such as physicians.
There is a need for further research to investigate the possible impacts of biosimilar medicines policies on prices, uptake and expenditure. Given the ongoing development of policies for biosimilar medicines, such studies need to be designed with a long-term perspective. Descriptive surveys, such as this manuscript, on poli cies and practices will help to inform such impact assessments.
For patients
Biosimilar medicines have the potential to increase patient access to medicines. Their prices are lower than those of originator medicines, which help to make biological medicines more affordable. Biosimilar medicines contribute to reduced pharmaceutical expenditure and thus free financial resources, ultimately allowing a greater number of patients to be treated. Policymakers are called upon to introduce policies for the pricing, funding and promotion of biosimilar medicines in order to take advantage of these benefits. However, successful implementation of pharmaceutical policies related to biosimilar medicines, as described in this article, requires patients’ understanding and acceptance. This research aims to contribute to patient knowledge in this area.
Country abbreviations
AL: Albania; AT: Austria; BE, Belgium; BG: Bulgaria; BY: Belarus; CA: Canada; CH: Switzerland; CY: Cyprus; CZ: Czech Republic; DE: Germany; DK: Denmark; EE: Estonia; EL: Greece; ES: Spain; FI: Finland; FR: France; HR: Croatia; HU: Hungary; IE: Ireland; IL: Israel; IS: Iceland; IT: Italy; KG: Kyrgyzstan; KZ: Kazakhstan; LT: Lithuania; LU: Luxembourg; LV: Latvia; MT: Malta; NL: The Netherlands; NO: Norway; PL: Poland; PT: Portugal; RS: Republic of Serbia; RU: Russia; RO: Romania; SE: Sweden; SI: Slovenia; SK: Slovakia; TR: Turkey; UA: Ukraine; UK: United Kingdom; ZA: South Africa.
Acknowledgements
Contributors: The authors thank their colleagues Ms Nina Zimmermann and Ms Margit Gombocz in the Pharmaceutical Pricing and Reimbursement Information (PPRI) Secretariat for their support in the data collection of the survey. Furthermore, we gratefully acknowledge the answers to the survey provided by PPRI network members.
Funders: No funding was received for writing this manuscript. The Austrian Federal Ministry of Health and Women’s Affairs financially supports the Pharmaceutical Pricing and Reimbursement Information (PPRI) Secretariat.
Prior presentations: Preliminary results related to one aspect of the presented research (pricing policies for biosimilar medicines) were presented at the ISPOR 19th Annual European Congress in Vienna, Austria, 31 October – 2 November 2016.
Funding source: The authors gratefully acknowledge the support of the Austrian Federal Ministry of Health and Women’s Affairs to the Austrian Public Health Institute for running the PPRI (Pharma ceutical Pricing and Reimbursement Information) Secretariat that is managed by the authors and colleagues. The members of the PPRI network (competent authorities for pharmaceutical pricing and reimbursement) supported this research by providing data and information for this manuscript. No funding for writing this manuscript was received.
Competing interests: None.
Provenance and peer review: Not commissioned; externally peer reviewed.
Authors
Sabine Vogler, PhD; Peter Schneider, MA
WHO Collaborating Centre for Pharmaceutical Pricing and Reimbursement Policies, Pharmacoeconomics Department, Gesundheit Österreich GmbH (GÖG/Austrian Public Health Institute), 6 Stubenring, AT-1010 Vienna, Austria.
References
1. European Commission. Carone G, Schwierz C, Xavier A. Cost-containment policies in public pharmaceutical spending in the EU. Economics and Financial Afairs, 2012 [homepage on the Internet]. [cited 2017 May 26]. Available from: http://ec.europa.eu/economy_finance/publications/economic_paper/2012/pdf/ecp_461_en.pdf
2. World Health Organization. WHO Guideline on country pharmaceutical pricing policies. Geneva: World Health Organization, 2013 [homepage on the Internet]. [cited 2017 May 26]. Available from: http://www.who.int/medicines/publications/pharm_guide_country_price_policy/en/
3. Vogler S. The impact of pharmaceutical pricing and reimbursement policies on generics uptake: implementation of policy options on generics in 29 European countries–an overview. Generics and Biosimilars Initiative (GaBI Journal). 2012;1(2):93-100. doi:10.5639/gabij.2012.0102.020
4. Dylst P, Vulto A, Simoens S. Demand-side policies to encourage the use of generic medicines: an overview. Expert Rev Pharmacoecon Outcomes Res. 2013;13(1):59-72.
5. Simoens S, De Coster S. Sustaining generic medicines markets in Europe. J Generic Med. 2006;3(4):257-68.
6. Simoens S. Sustainable provision of generic medicines in Europe. Leuven, The Netherlands: KU Leuven 2013.
7. Simoens S. A review of generic medicine pricing in Europe. Generics and Biosimilars Initiative (GaBI Journal). 2012;1(1):8-12. doi:10.5639/gabij.2012.0101.004
8. Vogler S, Zimmermann N. How do regional sickness funds encourage more rational use of medicines, including the increase of generic uptake? A case study from Austria. Generics and Biosimilars Initiative Journal (GaBI Journal). 2013;2(2):65-75. doi:10.5639/gabij.2013.0202.027
9. Dylst P, Vulto A, Simoens S. The impact of reference-pricing systems in Europe: a literature review and case studies. Expert Rev Pharmacoecon Outcomes Res. 2011;11(6):729-37.
10. Godman B, Wettermark B, Bishop I, Burkhardt T, Fürst J, Garuoliene K. European payer initiatives to reduce prescribing costs through use of generics. Generics and Biosimilars Initiative Journal (GaBI Journal). 2012;1(1):22-7. doi:10.5639/gabij.2012.0101.007
11. World Health Organization. Cameron A, Laing R. Cost savings of switching private sector consumption from originator brand medicines to generic equivalents. World Health Report, Background Paper, 35. Geneva 2010 [homepage on the Internet]. [cited 2017 May 26]. Available from: http://www.who.int/healthsystems/topics/financing/healthreport/35MedicineCostSavings.pdf
12. World Health Organization. Regional Office for Europe. Access to new medicines in Europe: technical review of policy initiatives and opportunities for collaboration and research. Copenhagen, 2015 [homepage on the Internet]. [cited 2017 May 26]. Available from: http://www.euro.who.int/en/healthtopics/Health-systems/health-technologies-and-medicines/publications/2015/access-to-new-medicines-in-europe-technical-review-of-policy-initiativesand-opportunities-for-collaboration-and-research-2015
13. Derbyshire M. Patent expiry dates for best-selling biologicals. Generics and Biosimilars Initiative Journal (GaBI Journal). 2015;4(4):178-9. doi:10.5639/gabij.2015.0404.040
14. Haustein R, de Millas C, Höer A, Häussler B. Saving money in the European healthcare systems with biosimilars. Generics and Biosimilars Initiative Journal (GaBI Journal). 2012;1(3-4):120-6. doi:10.5639/gabij.2012.0103-4.036
15. Mack A. Norway, biosimilars in different funding systems. What works? Generics and Biosimilars Initiative Journal (GaBI Journal). 2015;4(2):90-2. doi:10.5639/gabij.2015.0402.018
16. Curto S, Ghislandi S, van de Vooren K, Duranti S, Garattini L. Regional tenders on biosimilars in Italy: an empirical analysis of awarded prices. Health Policy. 2014;116(2-3):182-7.
17. European Commission. What you need to know about biosimilar medicinal products. Consensus Information Document [homepage on the Internet]. [cited 2017 May 26]. Available from: http://www.medicinesforeurope.com/wp-content/uploads/2016/03/biosimilars_report_en.pdf
18. Acha V, Allin P, Bergunde S, Bisordi F, Roediger A. What pricing and reimbursement policies to use for off-patent biologicals? – Results from the EBE 2014 biological medicines policy survey. Generics and Biosimilars Initiative Journal (GaBI Journal). 2015;4(1):17-24. doi:10.5639/gabij.2015.0401.006
19. Mestre-Ferrandiz J, Towse A, Berdud M. Biosimilars: how can payers get longterm savings? Pharmacoeconomics. 2016;34(6):609-16.
20. Renwick MJ, Smolina K, Gladstone EJ, Weymann D, Morgan SG. Postmarket policy considerations for biosimilar oncology drugs. Lancet Oncol. 2016;17(1):e31-8.
21. Mendoza C, Ionescu D, Radière G, Rèmuzat C, Young K, Toumi M. Biosimilar substitution policies: an overview. Value Health. 2015;18(7):A525.
22. Drozd M, Szkultecka-D bek M, Baran-Lewandowska I. Biosimilar drugs–automatic substitution regulations review. Polish ISPOR chapter’s Therapeutic Programs and Pharmaceutical Care (TPPC) task force report. J Health Policy Outcomes Res. 2014;1:52-7.
23. Vogler S, Leopold C, Zimmermann N, Habl C, de Joncheere K. The Pharmaceutical Pricing and Reimbursement Information (PPRI) initiative–experiences from engaging with pharmaceutical policy makers. Health Policy Technol. 2014;3(2):139-48.
24. World Health Organization. List of participating Member States [home page on the Internet]. [cited 2017 May 26]. Available from: http://www.euro.who.int/en/health-topics/disease-prevention/vaccines-and-immunization/european-immunization-week/european-immunization-week-20052015/european-immunization-week-2012/list-of-participating-member-states)
25. Gombocz M, Vogler S, Zimmermann N. Ausschreibungen für Arzneimittel: Erfahrungen aus anderen Ländern und Umsetzungsstrategien für Österreich. 2016.
26. GaBI Online – Generics and Biosimilars Initiative. Huge discount on biosimilar infliximab in Norway [www.gabionline.net]. Mol, Belgium: Pro Pharma Communications International; [cited 2017 May 26]. Available from: www.gabionline.net/Biosimilars/General/Huge-discount-on-biosimilar-infliximab-in-Norway
27. Curto A, Van de Vooren K, Garattini L, Lo Muto R, Duranti S. Regional tenders on biosimilars in Italy: potentially competitive? Generics and Biosimilars Initiative (GaBI Journal). 2013;2(3):123-7. doi:10.5639/gabij.2013.0203.036
28. World Health Organization. Collaborating Centre for Pharmaceutical Pricing and Reimbursement Policies. Leopold C, Habl C, Vogler S. Tendering of pharmaceuticals in EU Member States and EEA countries. Results from the country survey. Vienna: ÖBIG Forschungs- und Planungsgesellschaft mbH, 2008 [homepage on the Internet]. [cited 2017 May 26]. Available from: http://whocc.goeg.at/Literaturliste/Dokumente/BooksReports/Final_Report_Tendering_June_08.pdf
29. Petrou P, Talias MA. Tendering for pharmaceuticals as a reimbursement tool in the Cyprus public Health Sector. Health Policy Technol. 2014;3(3):167-75.
30. Wouters OJ, Kanavos PG. Transitioning to a national health system in Cyprus: a stakeholder analysis of pharmaceutical policy reform. Bull World Health Organ. 2015;93(9):606-13.
31. Vogler S, Habl C, Leopold C, Rosian-Schikuta I, de Joncheere K. PPRI Report. Vienna: (PPRI), 2008. Available from: https://ppri.goeg.at/Downloads/Publications/PPRI_InterimTechnicalReport.pdf
32. Habl C, Vogler S, Leopold C, Schmickl B, Fröschl B. Referenzpreissysteme in Europa. Analyse und Umsetzungsvoraussetzungen für Österreich; Wien: ÖBIG Forschungs- und Planungsgesellschaft mbH; 2008.
33. Dylst P, Vulto A, Simoens S. Tendering for outpatient prescription pharmaceuticals: what can be learned from current practices in Europe? Health Policy. 2011;101(2):146-52.
34. Thomsen E, Er S, Schüder P. PHIS Pharma Profile Denmark. Vienna: Pharmaceutical Health Information System (PHIS), 2011.
35. Leopold C, Habl C, Vogler S, Rosian-Schikuta I. Steuerung des Arzneimittelverbrauchs am Beispiel Dänemark. Vienna: Gesundheit Österreich GmbH (Austrian Health Institute), 2008.
36. Ministry of Health Denmark/Danish Health and Medicines Authority. Flowchart of the pharmaceutical system in Denmark. Poster. 3rd International PPRI Conference on Pharmaceutical Pricing and Reimbursement Policies Challenges Beyond the Financial Crisis. 12–13 October 2015; Vienna, Austria.
37. Vogler S. How large are the differences between originator and generic prices? Analysis of five molecules in 16 European countries. Farmeconomia. Health Economics and Therapeutic Pathways. 2012;13(Suppl 3):29-41.
38. Simoens S. Developing competitive and sustainable Polish generic medicines market. Croat Med J. 2009;50(5):440-8.
39. Dylst P, Simoens S. Does the market share of generic medicines influence the price level?: a European analysis. Pharmacoeconomics. 2011;29(10):875-82.
40. Aalto-Setälä V. The impact of generic substitution on price competition in Finland. Eur J Health Econ. 2008;9(2):185-91.
41. Spinks J, Chen G, Donovan L. Does generic entry lower the prices paid for pharmaceuticals in Australia? A comparison before and after the introduction of the mandatory price-reduction policy. Aust Health Rev. 2013;37(5):675-81.
42. Progenerika. Kanavos P. Tender systems for outpatient pharmaceuticals in the European Union: evidence from The Netherlands and Germany. European Medicines Information Network (EMINet), 2012 [homepage on the Internet]. [cited 2017 May 26]. Available from: http://www.progenerika.de/wp-content/uploads/2013/02/Anlage-2_Tendering-Report-EMINET-13OCT2012-FINAL.pdf
43. European Commission. Kanavos P, Seeley L, Vandoros S. Tender systems for outpatient pharmaceuticals in the European Union: evidence from The Netherlands, Germany and Belgium. European Medicines Information Network (EMINet), 2009 [homepage on the Internet]. [cited 2017 May 26]. Available from: http://ec.europa.eu/docsroom/documents/7607?locale=en
44. Flume M. Regional management of biosimilars in Germany. Generics and Biosimilars Initiative Journal (GaBI Journal). 2016;5(3):125-7. doi:10.5639/gabij.2016.0503.031
45. Towse A, Mestre-Ferrandiz J, Berdud M, Brown JD, Walson PD, Godman B, et al. Biosimilars: achieving long-term savings and competitive markets. Generics and Biosimilars Initiative Journal (GaBI Journal). 2016;5(3):103-4. doi:10.5639/gabij.2016.0503.027
46. European Commission. Report of the multi-stakeholder workshop on biosimilar medicinal products. 2016 [homepage on the Internet]. [cited 2017 May 26]. Available from: http://ec.europa.eu/DocsRoom/documents/19302
47. Goll G, Olsen I, Jorgensen K, Lorentzen M, Bolstad N, Haavardsholm E, et al. Biosimilar infliximab (CT-P13) is not inferior to originator infliximab: results from a 52-week randomized switch trial in Norway. Arthritis Rheumatol. 2016;68(Suppl 10).
48. IMS Health. Australia: substitution rules for biosimilars criticies by research based industry. IMS Pharma Pricing & Reimbursement. 2015;20(10): 306-2.
|
Author for correspondence: Sabine Vogler, PhD, Head of WHO Collaborating Centre for Pharmaceutical Pricing and Reimbursement Policies, Head of Pharmacoeconomics Department, Gesundheit Österreich GmbH (GÖG/Austrian Public Health Institute), 6 Stubenring, AT-1010 Vienna, Austria |
Disclosure of Conflict of Interest Statement is available upon request.
Copyright © 2017 Pro Pharma Communications International
Permission granted to reproduce for personal and non-commercial use only. All other reproduction, copy or reprinting of all or part of any ‘Content’ found on this website is strictly prohibited without the prior consent of the publisher. Contact the publisher to obtain permission before redistributing.