Pharmaceutical pricing policy in Saudi Arabia: findings and implications
Published on 2020/02/19
Generics and Biosimilars Initiative Journal (GaBI Journal). 2020;9(1):14-21.
|
Introduction: Many countries have introduced policies and strategies to limit pharmaceutical expenditures. These include pharmaceutical pricing policies and related strategies to control medicine prices and to ensure appropriate and stable prices. The aim of this study was to provide an overview of the current pharmaceutical pricing policy for medicines in Saudi Arabia and to provide an evaluation of the impact of this policy on medicine prices. |
Submitted: 1 October 2019; Revised: 23 January 2020; Accepted: 6 February 2020; Published online first: 19 February 2020
Introduction
Healthcare in Saudi Arabia is provided via a dual system, i.e. public and private sector. Approximately three quarters of the healthcare services are currently provided by the public sector. However, the private healthcare sector has grown in recent years and is now considered an essential component of the Saudi healthcare system [1–3]. The public sector is funded mainly by the government budget while the private sector is financed by the cooperative health insurance schemes and out-of-pocket payments [1, 2]. The total healthcare expenditure is steadily increasing from US$23.641 billion in 2011 to US$40.657 billion in 2018 (66% of the total expenditure is currently funded by the governmental budget) [4, 5]. Regarding the Saudi pharmaceutical sector, it depends heavily on imported pharmaceuticals from the US, Europe and some other the Gulf Cooperation Council (GCC) countries [1, 5]. The local pharmaceutical companies produce approximately one quarter of the pharmaceuticals in the Saudi pharmaceutical market [1]. However, more initiatives and investment have started in recent years to stimulate the pharmaceutical production as part of a strategic plan to produce at least 40% of all medicines locally on the long term [1, 5].
Pharmaceutical expenditures make up a large component of overall health expenditures [6–21], representing approximately 20%–60% of the health expenditures in low- and middle-income countries (LMICs) and approximately 18% of the health expenditures in the Organisation for Economic Co-operation and Development (OECD) countries [6]. In Saudi Arabia, pharmaceutical expenditures represented 19.4% of the total health expenditures in 2018 [5]. Moreover, similar to many other countries, this has steadily increased in recent years. For example, pharmaceutical expenditure increased from US$4.894 billion in 2011 to US$7.897 billion in 2018 [4, 5]. The rise in pharmaceutical expenditure can be attributed to many factors. These factors include the increase in the prevalence of diseases, e.g. chronic diseases, the increase in risk factors associated with these diseases, ageing populations, changes in treatment goals (i.e. stricter therapeutic targets), the introduction/early adoption of expensive medicines/new medicines, e.g. biotechnology medicines, anticancer agents, and immunomodulating agents, growth in medicine volumes, and increasing patient expectations [10, 14–18, 22–26].
In view of this tremendous increase in expenditure, which threatens the sustainability of healthcare systems, many countries have introduced strategies and policies, including pharmaceutical pricing policies, to control medicine prices, to ensure medicine affordability and accessibility, to ensure price stability, to promote innovation, and to maintain pharmaceutical production [12, 15, 22, 23, 27–31]. Globally, in terms of the pricing of medicines, several strategies are used, including external price referencing (also called international price benchmark/referencing), internal price referencing, value-based pricing, tendering and negotiations, and special pricing agreements [11, 32]. External price referencing refers to the practice of determining the price of a medicine in a given country by considering the prices of the medicine in other countries in which the product is marketed. Internal price referencing refers to the practice of determining the price of a medicine by considering the prices of its identical, similar, or therapeutically equivalent medicines within the same country. Value-based pricing is used to determine the prices of new medicines based on the therapeutic value of the medicine. This is commonly evaluated through heath technology assessment (HTA) or pharmacoeconomic evaluations, such as cost-effectiveness analysis, cost minimization, and cost–benefit analysis [11, 32]. In tendering procedures, the pharmaceutical companies and manufacturers submit quotations for a particular contract in a competitive bidding process. Then, the company or manufacturer that offers the best bid wins the tender based on the specified criteria [11, 22]. As a pricing strategy, special pricing agreements (also called innovative agreements or managed entry agreements) are adopted by some countries such as Australia, France, Germany, New Zealand, Spain, Taiwan, Thailand and the UK [11, 33]. These agreements are used to facilitate the entry and funding of some new medicines, especially when it is challenging to determine the prices of newly launched medicines because of the uncertainty of their future effectiveness and the value that the medicine will offer in real life situations, beyond just clinical trials [33].
In Saudi Arabia, historically, medicine prices have been determined by the Ministry of Health. Currently, the Saudi Food and Drug Authority (FDA), established in 2003, is the national regulatory body responsible for the registration and approval of medicines, including the pricing of medicines [1]. In Saudi Arabia, according to Pharmaceutical Institutions, and Product Law, retail pharmaceutical prices must be officially approved before marketing any pharmaceutical product [34, 35]. Currently, medicine prices are determined based on the pharmaceutical pricing policy approved by the Saudi FDA. Therefore, the aim of this article was to provide an overview of the current pharmaceutical pricing policy of medicines in Saudi Arabia and to provide an evaluation of the impact of this policy on medicine prices, including both the original brand medicines and generic medicines.
An overview of pharmaceutical pricing policy
Pharmaceutical pricing policy
Medicine prices are determined according to pharmaceutical pricing rules and policies, which were approved by the Saudi FDA and have been implemented since November 2011 [36]. The key features of this policy are as follows:
- General considerations: In general, when determining the prices of medicines, according to pharmaceutical pricing rules and policies, there are nine factors/criteria that need to be considered, see Table 1.
- Pricing of original brand medicines:
- Price deduction of the original brand medicine by 20% upon registration of the first equivalent generic medicine.
- If the original brand is manufactured locally under license agreement with the license holding company, it will be given the same registered price as the imported original brand. Once the first generic medicine is registered, there will be a 20% reduction.
- If the original brand is manufactured by a local manufacturer with license agreement with the license holding company as a second brand, it will be given the same price as the original brand during the patent protection period, and after patent expiration the second local brand will be treated as the first generic medicine in terms of pricing.
- Pricing of generic medicines:
- Generic medicines are priced by taking into consideration the criteria as stipulated in Table 1.
- The first registered generic medicines will be priced at least 35% lower than the price of original brand medicine.
- The second registered generic medicine will be priced 10% lower than the set price of the first registered generic medicine. Similarly, the third generic medicine will be priced at least 10% lower than the second registered generic medicine. This is also the case for the fourth generic medicine, where its price will be set at least 10% lower than the third registered generic medicine.
- From the fourth generic medicine onward, the price may be fixed without further reductions.
Regulating mark-ups in the pharmaceutical supply chain
Mark-ups are determined by Article 13 of the Pharmaceutical Institutions and Products Law, as summarized in Table 2 [34, 35]:
Methods
A price comparison between the original brand medicine, i.e. innovator’s product, and its generic versions was conducted. The analysis was performed for prescription medicines, i.e. active ingredients, in which their products were listed in the top 10 products sold in Saudi Arabia during the period of 2010–2015 [37]. These medicines included diclofenac (three products), amoxicillin/clavulanic acid (two products), metformin/sitagliptin, esomeprazole, atorvastatin, pantoprazole, and tadalafil. In this study, no comparison was attempted for metformin/sitagliptin since it is currently available only as an innovator’s product. To ensure the accuracy of the comparisons, only medicines of the same properties were included [38]. The products compared had to be in the same dosage form, with the same strength and the same package size. In addition, they had to be the same in terms of other characteristics that could affect the price, e.g. film coating. Also, the same package size was considered since medicines in retail community pharmacies usually sell products in their original packages. Moreover, only products that were currently marketed in the country were considered, i.e. products that were not currently marketed or that were suspended or withdrawn by the Marketing Authorisation Holder (MAH) were excluded.
Descriptive comparisons of medicine prices were made based on the official prices of medicines, i.e. retail prices for consumers. The medicine prices were obtained from Saudi FDA’s official list of registered medicines and herbal products (available at https://www.sfda.gov.sa/en/drug/search/Pages/default.aspx) [39].
The study calculated the absolute price difference between the original brand medicine and the generic medicine in Saudi Arabian Riyals (SAR) and it calculated the generics to original brand medicine price ratio. Similarly, among generic medicines, the absolute price difference between the highest price generic medicine and other generic medicines and the price ratio of the highest price generic medicine to other generic medicines of the same molecule were calculated [23].
Results
The study compared the prices of the top selling medicines in Saudi Arabia, including six different medicines. Overall, there were significant differences in the prices of the generic medicines compared to the original brand products. The price ratio of the generic medicines to the original brand medicine was 0.87–0.30. The price of the first generic medicine was close to the price of original brand medicine, where the first generic medicine to original brand medicine price ratio was 0.87–0.81. In this study, there were significant differences in the prices of the generic medicines for the same molecule. In fact, price ratio among the generic medicines for the same molecule was between 0.96 and 0.18. In addition, some generic medicines imported from high income countries were cheaper than the medicines manufactured locally or from other countries within the Middle East. The price comparisons for each medicine are presented in Figure 1.
There was only one generic version of esomeprazole during the study period. The original brand of atorvastatin is currently not marketed; hence, the prices for its generic versions were compared to the first registered generic version.
Diclofenac
As shown in Table 3, the price comparison was conducted for diclofenac sodium (50 mg tablets), available in a package size of 20 tablets. The generic medicine to original brand medicine price ratio was 0.87–0.31. In addition, there was a large variation among the prices of the generic versions of diclofenac, where the generic medicine to generic medicine price ratio was 0.9–0.35.
Amoxicillin/clavulanic acid
As shown in Table 4, the price comparison was conducted for amoxicillin/clavulanic acid (625 mg tablets), available in a package size of 20 tablets. The generic medicine to original brand medicine price ratio was 0.86–0.31. In addition, there was a large variation among the prices of the generic versions of amoxicillin/clavulanic acid, with the generic medicine to generic medicine price ratio was 0.96–0.36.
Esomeprazole
As shown in Table 5, the price comparison was conducted for esomeprazole (40 mg) gastro-resistant coated tablets, available in a package size of 28 tablets. There was only one generic version registered in the same dosage form. The price ratio of generic medicine to the original brand medicine was 0.81.
Atorvastatin
As shown in Table 6, the price comparison was conducted for atorvastatin (40 mg) film-coated tablets, available in a package size of 30 tablets. The original brand medicine (Lipitor®) is currently not marketed in Saudi Arabia. Hence, the price comparison was conducted among its generic versions that are currently registered in Saudi Arabia. There was a large variation among the prices of the generic versions of atorvastatin, with the generic medicine to generic medicine price ratio was 0.87–0.18.
Pantoprazole
As shown in Table 7, the price comparison was conducted for pantoprazole (40 mg) gastro-resistant, coated tablets, available in a package size of 30 tablets. The generic medicine to original brand medicine price ratio was 0.87–0.46. In addition, there was a large variation among the prices of the generic versions of pantoprazole, where the generic medicine to generic medicine price ratio was 0.84–0.53.
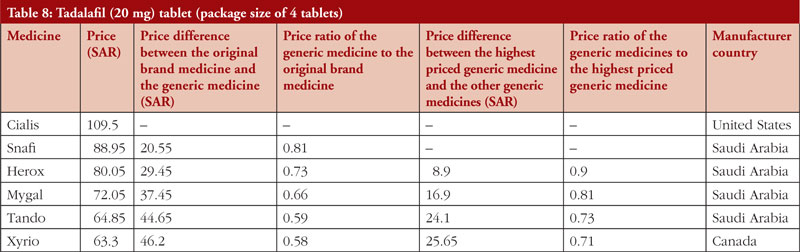
Tadalafil
As shown in Table 8, the price comparison was conducted for tadalafil (20 mg) tablets, available in a package size of four tablets. The generic medicine to original brand medicine price ratio was 0.81–0.58. In addition, there was a large variation among the prices of the generic versions of tadalafil, with the generic medicine to generic medicine price ratio was 0.9–0.71.
Discussion
The current approach adopted for medicine pricing is the ‘prescriptive pricing approach’ for both original brand medicines and generic medicines. In this approach, health authorities or regulators mandate price reductions that are necessary for the reimbursement price or for determining the price [15, 29]. In addition, a generic price link policy was adopted in Saudi Arabia. In this policy, the first generic medicine and subsequent generic medicines are priced lower than the original brand medicine. This policy has been implemented in many European countries, such as Austria, Belgium, Switzerland, Germany, Estonia, Finland, France and Ireland [32]. However, the percentage of price reductions at which generic medicines must be priced lower than the original brand medicine, as well as the additional steps, vary widely between countries [32]. Moreover, in Saudi Arabia the generic medicines are priced according to the order of registration, and the subsequent generic medicines are priced at least 10% lower than the product registered earlier.
Currently, the price reduction of the original brand medicine upon registration of the first generic medicine is 20%. The first generic medicine is priced 35% lower than the pre-patent price of the original brand medicine, with subsequent generic medicines priced at least 10% lower, until the fourth generic medicine. This approach has been adopted by several countries, with differences in the percentage of the reductions. For example, in South Korea, upon expiration of the patent, the price of the original brand medicine is reduced by 30%. The generic medicine will be priced 15% lower than the original brand medicine price after the reduction. After one year, there is an additional 10% reduction in the price of the medicine, and both the original brand medicine and the generic medicine are priced at the same level, i.e. 53.55% of the pre-patent price [12, 27, 40]. In Egypt, the original brand medicine price is fixed by selecting the lowest price in the region where the medicine is marketed. The first five generic medicines are priced 35% lower than the innovator’s medicine, while the subsequent generic medicines will be priced 40% lower than the innovator’s product. However, imported generic medicines with high technological features are priced 30%–35% lower than the innovator’s product [41]. In Australia, a 16% reduction in price is made when the first generic medicine is added to the national scheme. Subsequent reductions are made based on a price-disclosure policy. In this policy, which is based on the actual selling price, i.e. ex-factory price, a weighted, one-year average, disclosed price (WADP) is calculated. Accordingly, a price reduction is made if the current ex-factory price is higher than 10% of the WADP (i.e. the price will be reduced to the level of the WADP) [16, 27]. In Europe, particularly France, the first generic medicine is required to be at least 55% lower than the price of the original brand medicine to be reimbursed. In addition, the prices of off-patent medicines are required to be reduced by 15%–19% upon registration of the 1st generic medicine [14, 42]. In Norway, a stepped price model has been adopted. In this model, the price of the prescription medicine is reduced in a stepwise manner after patent expiration of the innovator product. The price reductions depend on the annual sales of the original brand medicine before generic competition, as well the time of generic competition establishment. In the first step (at the time of generic competition establishment), the price is reduced by 35%. This is followed by further reductions in the second (after 6 months) and third steps (18 months or more), which could be a 90% reduction in some medicines, depending on the sales [43].
The current pricing policy in Saudi Arabia has led to major differences in the prices of medicines. However, the price of the first generic medicine is relatively close to the post-patent price of the original brand medicine. In fact, the first generic medicine to original brand medicine ratio was only between 0.87–0.81. However, the price difference between the other subsequent generic versions and the original brand medicine varies widely, but generally it is considered large. The price ratio between the lowest priced medicine and its original brand medicine was 0.58–0.30. The overall generic medicine to original brand medicine price ratio was 0.87–0.30. By comparison, the study by Zeng (2013) reported that in China the difference was 0.34 and 0.98 [23]. In the current study, large variations were noted among the generic medicines of the same molecule, with a generic medicine to generic medicine price ratio of 0.96–0.18. Hence, in terms of pricing, three categories of generic medicines can be noted – the highest priced, moderately priced, and lowest priced generic medicines. In addition, the prices of some local products are higher than the imported generic medicines from some high-income countries.
In fact, in addition to the initial reductions in the prices of medicines, there are different subsequent methodological specifications, strategies, and interventions that lead to further reductions in medicine prices in many countries [16, 32, 43]. In addition, market completion can further decrease medicine prices. In Saudi Arabia, in the public healthcare sector, further reductions in medicine prices are achieved via unified procurement and the competitive tendering system. These large, centralized tenders utilize the power of group purchasing and help to provide further cost savings [1, 44]. However, the retail pharmacy prices of medicines, i.e. the prices set for the consumers at community pharmacies, are fixed, and they are printed on product packages. In addition, no discounts can be provided to medicine consumers by community pharmacies since this is legally not permitted in Saudi Arabia. Hence, to promote generic medicines, especially low-priced generic medicines, demand-side policies particularly educational activities and campaigns are needed to promote the cost-effective use of medicines. This needs to target both healthcare professionals and medicine consumers. This is particularly important since there is a general preference towards original brand medicines in Saudi Arabia [5] with negative perceptions towards generic medicines [45–48]. Moreover, only 47.9% of physicians surveyed by Salhia et al. (2015) indicated that they are familiar with the price differences [46]. In addition, this is not helped by the current myths regarding generic medicines, i.e. the association between low price and low quality and effectiveness, as reported in the literature [49–52]. Hence, educational campaigns need to highlight the facts regarding generic medicines, which are clinically interchangeable with the original brand medicines, with the same quality, safety, and effectiveness, (the only difference is their cheaper prices). In addition, the current rigorous registration system of medicines and post-marketing surveillance should be discussed in the campaigns to ensure awareness regarding this topic [1]. Additionally, incorporation of medicine prices in electronic prescribing systems, especially at private sectors, could help promote physician awareness regarding medicine prices and it could encourage cost-effective prescribing [53].
Strengths and limitations
The current study summarizes the key aspects of pharmaceutical pricing policy in Saudi Arabia, and it provides an analysis of the impact of this policy on medicine prices by studying the highest selling products in Saudi Arabia. However, this study has some limitations. The study analysed the prices of only six medicines, which is considered a small number; although, these are the top selling products. In addition, only one dosage form of these products was analysed. However, this is justified since the pricing policy takes into consideration the size and the type of dosage form when determining the price. However, we believe that the current study provides preliminary evidence that will be useful to health policymakers and researchers. Large comprehensive studies in this area are needed to further analyse the aspects of this policy for future improvement and revisions.
Conclusion
Medicine prices are strictly controlled through the pharmaceutical pricing policy in Saudi Arabia. Moreover, the current policy resulted in large price differences among medicines, including medicines of the same molecule. Due to this large difference, the cost savings will depend on the product that is prescribed or procured by the health organization.
Funding sources
None.
Competing interests: None.
Provenance and peer review: Not commissioned; externally peer reviewed.
References
1. Alrasheedy AA, Hassali MA, Wong ZY, Aljadhey H, AL-Tamimi SK, Saleem F. Pharmaceutical policy in Saudi Arabia. In: Babar ZUD, editor. Pharmaceutical Policy in Countries with Developing Healthcare Systems: Springer; 2017. p. 329-47.
2. Al Asmri M, Almalki MJ, Fitzgerald G, Clark M. The public healthcare system and primary care services in Saudi Arabia: a system in transition. East Mediterr Health J. 2019;25. doi.org/10.26719/emhj.19.049
3. Almalki M, Fitzgerald G, Clark M. Health care system in Saudi Arabia: an overview. East Mediterr Health J. 2011;17(10):784-93.
4. Business Monitor International. Saudi Arabia Pharmaceuticals & Healthcare Report Q3 2015.
5. Fitch Solutions Group. Saudi Arabia Pharmaceuticals & Healthcare Report Q3 2019.
6. World Health Organization. WHO guideline on country pharmaceutical pricing policies. 2015 [homepage on the Internet]. [cited 2020 Jan 23]. Available from: https://apps.who.int/iris/bitstream/handle/10665/153920/9789241549035_eng.pdf
7. Belloni A, Morgan D, Paris V. Pharmaceutical expenditure and policies. 2016 [homepage on the Internet]. [cited 2020 Jan 23]. Available from: https://www.oecd-ilibrary.org/social-issues-migration-health/pharmaceutical-expenditure-and-policies_5jm0q1f4cdq7-en
8. OECD. Pharmaceutical Expenditure. In: Health at a Glance 2017: OECD Indicators. London: OECD Publishing; 2017.
9. Hassali MA, Alrasheedy AA, McLachlan A, Nguyen TA, Al-Tamimi SK, Ibrahim MIM, et al. The experiences of implementing generic medicine policy in eight countries: a review and recommendations for a successful promotion of generic medicine use. Saudi Pharm J. 2014;22(6):491-503.
10. Hassali MA, Wong ZY, Alrasheedy AA, Saleem F, Yahaya AHM, Aljadhey H. Perspectives of physicians practicing in low and middle income countries towards generic medicines: a narrative review. Health Policy. 2014;117(3):297-310.
11. Verghese NR, Barrenetxea J, Bhargava Y, Agrawal S, Finkelstein EA. Government pharmaceutical pricing strategies in the Asia-Pacific region: an overview. Mark Access Health Policy. 2019;7(1):1601060.
12. Yoo K-B, Lee SG, Park S, Kim TH, Ahn J, Cho M-H, et al. Effects of drug price reduction and prescribing restrictions on expenditures and utilisation of antihypertensive drugs in Korea. BMJ Open. 2015;5(7):e006940.
13. Mousnad MA, Palaian S, Shafie AA, Ibrahim MIM. Medicine expenditure and trends in National Health Insurance Funds Sudan. J Pharmacy Pract Comm Med. 2018;4(3).
14. Godman B, Shrank W, Andersen M, Berg C, Bishop I, Burkhardt T, et al. Policies to enhance prescribing efficiency in Europe: findings and future implications. Front Pharmacol. 2011;1:141.
15. Godman B, Wettermark B, Van Woerkom M, Fraeyman J, Alvarez-Madrazo S, Berg C, et al. Multiple policies to enhance prescribing efficiency for established medicines in Europe with a particular focus on demand-side measures: findings and future implications. Front Pharmacol. 2014;5:106.
16. Vitry AI, Thai L, Roughead EE. Pharmaceutical pricing policies in Australia. In: Babar ZUD, editor. Pharmaceutical prices in the 21st century: Springer; 2015. p. 1-23.
17. Kwon H-Y, Yang B, Godman B. Key components of increased drug expenditure in South Korea: implications for the future. Value Health Reg Issues. 2015;6:14-21.
18. Pataky R, Tran DA, Coronado A, Alvi R, Boehm D, Regier DA, et al. Cancer drug expenditure in British Columbia and Saskatchewan: a trend analysis. CMAJ Open. 2018;6(3):E292-E9.
19. Schumock GT, Stubbings J, Wiest MD, Li EC, Suda KJ, Matusiak LM, et al. National trends in prescription drug expenditures and projections for 2018. Am J Health Syst Pharm. 2018;75(14):1023-38.
20. Prada SI, Soto VE, Andia TS, Vaca CP, Morales ÁA, Márquez SR, et al. Higher pharmaceutical public expenditure after direct price control: improved access or induced demand? The Colombian case. Cost Eff Resour Alloc. 2018;16(1):8.
21. Xiong Y, Cui Y, Zhang X. Pharmaceutical expenditure and total health-care expenditure in OECD countries and China: bidirectional Granger causality on the basis of health level. Expert Rev Pharmacoecon Outcomes Res. 2019:1-8.
22. Dylst P, Simoens S. Generic medicine pricing policies in Europe: current status and impact. Pharmaceuticals. 2010;3(3):471-81.
23. Zeng W. A price and use comparison of generic versus originator cardiovascular medicines: a hospital study in Chongqing, China. BMC Health Serv Res. 2013;13(1):390.
24. Godman B, Abuelkhair M, Vitry A, Abdu S, Bennie M, Bishop I. Payers endorse generics to enhance prescribing efficiency: impact and future implications, a case history approach. Generics and Biosimilars Initiative Journal. 2012;1(2):69-83. doi:10.5639/gabij.2012.0102.017
25. Lucchesi S, Marcianò I, Panagia P, Intelisano R, Randazzo MP, Sgroi C, et al. Prevalence of use and cost of biological drugs for cancer treatment: a 5-year picture from Southern Italy. Clin Drug Investig. 2018;38(3):269-78.
26. Selden TM, Abdus S, Miller GE. Decomposing changes in the growth of U.S. prescription drug use and expenditures, 1999-2016. Health Serv Res. 2019;54(4):752-63.
27. Roughead EE, Kim D-S, Ong B, Kemp-Casey A. Pricing policies for generic medicines in Australia, New Zealand, the Republic of Korea and Singapore: patent expiry and influence on atorvastatin price. WHO South East Asia J Public Health. 2018;7(2):99.
28. Godman B, Hill A, Simoens S, Kurdi A, Gulbinovič J, Martin A, et al. Pricing of oral generic cancer medicines in 25 European countries; findings and implications. Generics and Biosimilars Initiative Journal. 2019;8(2):49-70. doi:10.5639/gabij.2019.0802.007
29. Godman B, Shrank W, Wettermark B, Andersen M, Bishop I, Burkhardt T, et al. Use of generics—a critical cost containment measure for all healthcare professionals in Europe? Pharmaceuticals. 2010;3(8):2470-94.
30. Simoens S. A review of generic medicine pricing in Europe. Generics and Biosimilars Initiative Journal (GaBI Journal). 2012;1(1):8-12. doi:10.5639/gabij.2012.0101.004
31. Moodley R, Suleman F. The impact of the single exit price policy on a basket of generic medicines in South Africa, using a time series analysis from 1999 to 2014. PloS One. 2019;14(7):e0219690.
32. Vogler S, Martikainen JE. Pharmaceutical pricing in Europe. In: Babar ZUD, editor. Pharmaceutical prices in the 21st century: Springer; 2015. p. 343-70.
33. Dunlop WC, Staufer A, Levy P, Edwards GJ. Innovative pharmaceutical pricing agreements in five European markets: a survey of stakeholder attitudes and experience. Health Policy. 2018;122(5):528-32.
34. Ministry of Health of Saudi Arabia. [Executive regulations of the pharmaceutical institutions and products law 2005]. Arabic. [homepage on the Internet]. [cited 2020 Jan 23 ]. Available from: https://www.moh.gov.sa/en/Ministry/Rules/Documents/Regulation-of-Pharmaceutical-Products-and-Institutions.pdf
35. Ministry of Health of Saudi Arabia. [Pharmaceutical institutions and products law 2004]. Arabic. [homepage on the Internet]. [cited 2020 Jan 23 ]. Available from: https://www.moh.gov.sa/Ministry/Rules/Documents/008.pdf
36. Saudi Food and Drug Authority (SFDA). [The rules of pricing of medicines 2011]. Arabic. [homepage on the Internet]. [cited 2020 Jan 23 ]. Available from: https://www.sfda.gov.sa/ar/drug/drug_reg/Pages/drug_reg.aspx
37. AlKhamees OA, AlNemer KA, Maneea MWB, AlSugair FA, AlEnizi BH, Alharf AA. Top 10 most used drugs in the Kingdom of Saudi Arabia 2010–2015. Saudi Pharm J. 2018;26(2):211-6.
38. Shafie AA, Hassali MA. Price comparison between innovator and generic medicines sold by community pharmacies in the State of Penang, Malaysia. Journal of Generic Medicines. 2008;6(1):35-42.
39. Saudi Food and Drug Authority (SFDA). Official List of registered drugs and herbal products-2019 [homepage on the Internet]. [cited 2020 Jan 23]. Available from: https://www.sfda.gov.sa/en/drug/search/Pages/default.aspx.
40. Kwon H-Y, Godman B. Drug pricing in South Korea. Appl Health Econ Health Policy. 2017;15(4):447-53. doi:10.1007/s40258-017-0307-0
41. Wanis H. Pharmaceutical pricing in Egypt. In: Babar ZUD, editor. Pharmaceutical Prices in the 21st Century: Springer; 2015. p. 59-78.
42. Sermet C, Andrieu V, Godman B, Van Ganse E, Haycox A, Reynier J-P. Ongoing pharmaceutical reforms in France. Appl Health Econ Health Policy. 2010;8(1):7-24.
43. Håkonsen H, Sundell KA. Pharmaceutical pricing policies in Norway and Sweden. In: Babar ZUD, editor. Pharmaceutical Prices in the 21st Century: Springer; 2015. p. 209-27.
44. World Health Organization. Saudi Arabia pharmaceutical country profile 2012 [homepage on the Internet]. [cited 2020 Jan 23 ]. Available from: http://www.who.int/medicines/areas/coordination/Saudi_ArabiaPSCP_Narrative2012-04-18_Final.pdf
45. Albarraq AA. Consumers’ perceptions on generic medicines in Taif city, Saudi Arabia. Saudi Journal for Health Sciences. 2013;2(1):18-22.
46. Salhia HO, Ali A, Rezk NL, El Metwally A. Perception and attitude of physicians toward local generic medicines in Saudi Arabia: a questionnaire-based study. Saudi Pharm J. 2015;23(4):397-404.
47. Albadr Y, Khan TM. Factors influencing community pharmacist decision to dispense generic or branded medicines; Eastern Province, Alahsa, Saudi Arabia. Saudi Pharm J. 2015;23(2):143-6.
48. Alkhuzaee FS, Almalki HM, Attar AY, Althubiani SI, Almuallim WA, Cheema E, et al. Evaluating community pharmacists’ perspectives and practices concerning generic medicines substitution in Saudi Arabia: a cross-sectional study. Health Policy. 2016;120(12):1412-9.
49. Kaplan WA, Ritz LS, Vitello M, Wirtz VJ. Policies to promote use of generic medicines in low and middle income countries: a review of published literature, 2000–2010. Health Policy. 2012;106(3):211-24.
50. Theodorou M, Tsiantou V, Pavlakis A, Maniadakis N, Fragoulakis V, Pavi E, et al. Factors influencing prescribing behaviour of physicians in Greece and Cyprus: results from a questionnaire based survey. BMC Health Serv Res. 2009;9(1):150.
51. Jamshed SQ, Hassali MAA, Ibrahim MIM, Babar ZUD. Knowledge attitude and perception of dispensing doctors regarding generic medicines in Karachi, Pakistan: a qualitative study. J Pak Med Assoc. 2011;61(1):80-3.
52. Jamshed SQ, Ibrahim MIM, Hassali MAA, Masood I, Low BY, Shafie AA, et al. Perception and attitude of general practitioners regarding generic medicines in Karachi, Pakistan: a questionnaire based study. Southern Med Rev. 2012;5(1):22-30.
53. Wong ZY, Hassali MA, Alrasheedy AA, Saleem F, Yahaya AHM, Aljadhey H, et al. Medical specialists’ knowledge, perceptions and views about generic medicines in Malaysia: findings from a qualitative study and the implications. J Generic Med. 2015;12(2):60-73.
|
Author: Alian A Alrasheedy, BPharm(Hons), MPharm(Clin), PhD, MACP, RPh, Dean, Unaizah College of Pharmacy, Qassim University, Saudi Arabia |
Disclosure of Conflict of Interest Statement is available upon request.
Copyright © 2020 Pro Pharma Communications International
Permission granted to reproduce for personal and non-commercial use only. All other reproduction, copy or reprinting of all or part of any ‘Content’ found on this website is strictly prohibited without the prior consent of the publisher. Contact the publisher to obtain permission before redistributing.











Please change the dates as it seems in correct as typo error:
Submitted: 1 October 2020; Revised: 23 January 2020; Accepted: 6 February 2020; Published online first: 19 February 2020
Thanks
Dear Mr Mohammed Masihuddin,
Thank you for your valuable comments and insight received on 11 January 2021. We have an updated article ‘Pharmaceutical pricing policy in Saudi Arabia: findings and implications’ published in GaBI Journal, 2020, Vol 9, Issue 1 with the correct date.
We appreciate very much your kind feedback, and please continue with your valuable comments to GaBI Journal).
Thank you for your interest in GaBI. Please enjoy the quality information and content published under GaBI (GaBI Online and GaBI Journal).
GaBI Journal Editorial Office