USA and Europe differ in interchangeability of biosimilars
Published on 2017/12/14
Generics and Biosimilars Initiative Journal (GaBI Journal). 2017;6(4):183-4.
|
Abstract: |
Submitted: 5 December 2017; Revised: 7 December 2017; Accepted: 8 December 2017; Published online first: 21 December 2017
There are some major differences between USA and Europe regarding how they view interchangeability of biologicals/ biosimilars. In fact, there is a lack of harmonization around the world when it comes to how different countries or regions approach interchangeability of biosimilars [1].
USA
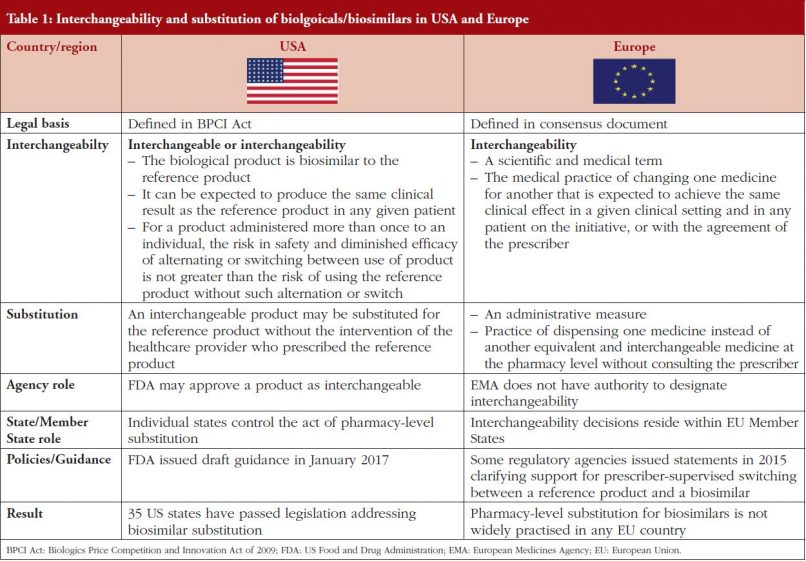
In the US, the Biologics Price Competition and Innovation Act of 2009 (BPCI Act) creates an abbreviated licensure pathway for biological products shown to be biosimilar to or interchangeable with a Food and Drug Administration (FDA) licensed reference product. Interchangeability is defined in law as part of the BPCI Act as:
‘the biological product may be substituted for the reference product without the intervention of the healthcare provider who prescribed the reference product’.
FDA defines interchangeability as:
- the biological product is biosimilar to the reference product;
- it can be expected to produce the same clinical result as the reference product in any given patient; and
- for a product that is administered more than once to an individual, the risk in terms of safety or diminished efficacy of alternating or switching between use of the product and its reference product is not greater than the risk of using the reference product without such alternation or switch.
FDA may approve a biological product as interchangeable, see Table 1. Although individual states control the act of pharmacy-level substitution. The agency issued draft guidance on interchangeability in January 2017 [2] and more recently extended the comment period on the guidance [3].
As of 1 July 2017, 35 states and Puerto Rico have passed laws allowing substitution by a pharmacist if the biosimilar is considered interchangeable and is covered under an insurer’s pharmacy benefit [4]. However, despite issuing draft guidance on interchangeability in January 2017 [2], to date FDA has yet to approve a biosimilar as interchangeable with its reference biological.
European Union
The European Commission (EC) has defined interchangeability in a consensus information document on biosimilars [5] as:
‘the medical practice of changing one medicine for another that is expected to achieve the same clinical effect in a given clinical setting and in any patient on the initiative, or with the agreement of the prescriber’.
Substitution is considered to be:
- An administrative measure
- The practice of dispensing one medicine instead of another equivalent and interchangeable medicine at the pharmacy level without consulting the prescriber
In the EU, decisions on the interchangeability or substitution of biosimilars and originator biologicals are not made by the European Medicines Agency (EMA), but at the national level, see Table 1. This is the case, despite the fact that biosimilars developed in line with EU requirements are considered by EMA to be therapeutic alternatives to their reference biologicals [6].
Automatic substitution of biosimilars is therefore not routinely practised in Europe, although in some Member States the use of biosimilars has been actively facilitated by national and local tender systems.
Competing interests: None.
Provenance and peer review: Article abstracted based on published scientific or research papers recommended by members of the Editorial Board; internally peer reviewed.
Michelle Derbyshire, PhD, GaBI Online Editor
References
1. Derbyshire M. Interchangeability of biosimilars in the US and around the world. Generics and Biosimilars Initiative Journal (GaBI Journal). 2017;6(2):97-8. doi:10.5639/gabij.2017.0602.017
2. GaBI Online – Generics and Biosimilars Initiative. FDA issues draft guidance on biosimilar interchangeability [www.gabionline.net]. Mol, Belgium: Pro Pharma Communications International; [cited 2017 Dec 7]. Available from: www.gabionline.net/Guidelines/FDA-issues-draft-guidance-on-biosimilar-interchangeability
3. GaBI Online – Generics and Biosimilars Initiative. FDA extends comment period for interchangeability guidance [www.gabionline.net]. Mol, Belgium: Pro Pharma Communications International; [cited 2017 Dec 7]. Available from: www.gabionline.net/Guidelines/FDA-extends-comment-period-for-interchange-ability-guidance
4. National Conference of State Legislatures. Cauchi R. State laws and legislation related to biologic medications and substitution of biosimilars [homepage on Internet]. [cited 2017 Dec 7]. Available from: www.ncsl.org/research/health/state-laws-and-legislation-related-tobiologic-medications-and-substitution-of-biosimilars.aspx
5. GaBI Online – Generics and Biosimilars Initiative. Integrating biosimilars into clinical practice [www.gabionline.net]. Mol, Belgium: Pro Pharma Communications International; [cited 2017 Dec 7]. Available from: www.gabionline.net/Biosimilars/Research/Integrating-biosimilars-into-clinical-practice
6. GaBI Online – Generics and Biosimilars Initiative. Biosimilar substitution in Europe [www.gabionline.net]. Mol, Belgium: Pro Pharma Communications International; [cited 2017 Dec 7 Available from: www.gabionline.net/Reports/Biosimilar-substitution-in-Europe
Disclosure of Conflict of Interest Statement is available upon request.
Copyright © 2018 Pro Pharma Communications International
Permission granted to reproduce for personal and non-commercial use only. All other reproduction, copy or reprinting of all or part of any ‘Content’ found on this website is strictly prohibited without the prior consent of the publisher. Contact the publisher to obtain permission before redistributing.



Dear Sir,
You mention a “table 1” in your article, but I didn’t find it online. Maybe I missed it.
Best regards
Stephan Garcia
Lyon Teachnig Hospital (France)
Dear Stephan Garcia,
Thank you for your valuable comments and insight received on 8 February 2018, the figures and table for the article ‘USA and Europe differ in interchangeability of biosimilars’ are now available to view online.
We appreciate very much your kind feedback, and please continue with your valuable comments to GaBI Journal.
Thank you for your interest in GaBI. Please enjoy the quality information and content published under GaBI (GaBI Online and GaBI Journal).