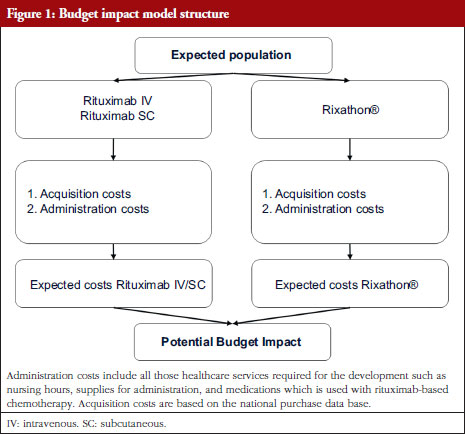
Increasing adoption of quality-assured biosimilars to address access challenges in low- and middle-income countries
Author byline as per print journal: Pritha Paul1, PhD; Rahul Kapur2*, MBBS, PhD; Shivani Mittra3, MPharm, PhD; Nimish Shah4, JD, MBA; Gopal K Rao1, MSc; Matthew E Erick4, BS, RPh; Susheel Umesh1, BPharm, MBA; Sandeep N Athalye2, MBBS, MD Abstract:Non-communicable diseases (NCDs) disproportionately affect people living in low- and middle-income countries (LMICs) compared to high-income […]