Generic clopidogrel–the medicines agency’s perspective
Published on 2012/02/10
Generics and Biosimilars Initiative Journal (GaBI Journal). 2012;1(2):89-91
|
Abstract: |
Submitted: 25 October 2011; Revised manuscript received: 5 April 2012; Accepted: 13 April 2012
Clopidogrel plays an important role as an anticoagulant drug in the prevention of atherothrombotic events such as myocardial infarction or stroke. Recently, generic clopidogrel products have gained marketing approval in Austria and are now available as alternatives to the originator product Plavix. However, scientific groups, including the eminent Austrian Society of Cardiology (Österreichische Kardiologische Gesellschaft, ÖKG), voiced concerns at the time of licensure about the safety and efficacy of the newly approved generic products [1].
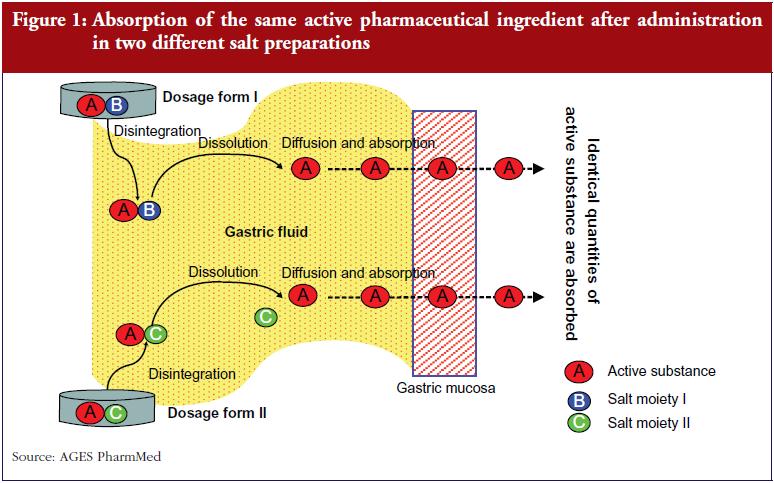
The original marketing authorisation was granted to a product containing clopidogrel as a hydrogen sulphate salt. Some generic clopidogrel products have this same salt preparation while others are compounded, for example, as clopidogrel hydrochloride, clopidogrel besylate, or clopidogrel free base. The question subsequently arose as to whether these different salt preparations act differently on their target receptors.
In support of these being equivalent products is the knowledge that the different clopidogrel salts exhibit the same pharmacodynamic properties. This is because they all dissociate in the gastrointestinal tract before reaching the blood circulation. Many active substances are formulated as salts specifically to improve their solubility in aqueous solutions and in the gastrointestinal tract. The active molecules being absorbed from the different salt preparations are always the same, see Figure 1, which results in a nearly identical pharmacodynamic profile, provided that the kinetic parameters are more or less identical. To preclude any theoretical relevant differences in pharmacokinetic parameters, the different clopidogrel salts were subjected to bioequivalence studies, as requested worldwide by medicines authorities and using acceptance limits specified in the EMA ‘Guideline on the Investigation of Bioequivalence’ [2]. This included measurement of the area under the curve (AUC) and the maximum concentration (Cmax) of the active molecules–which are the same chemically regardless of the salt formulation. If the salts show comparable pharmacokinetic behaviour in the bioequivalence studies, they can be considered pharmacologically more or less the same with regard to efficacy and safety.
Some critics have recommended that further clinical trials take place in order to substantiate the efficacy of clopidogrel generics, before regulatory approval. This is not legally required for generics medications in any of the EU Member States, however, and in general such trials have added little in terms of additional data or knowledge to support such an application. Nonetheless, pharmacodynamic data from some clinical trials do support the claim that different clopidogrel salts act in a comparable manner to inhibit platelet aggregation, so long as they have similar pharmacokinetic properties [3, 4].
EMA’s Pharmacokinetic Working Party [5], formerly named as the PK-expert group of the Efficacy Working Party (EWP), dealt comprehensively with such issues. This expert group advised on the development of generic clopidogrel products and specified the necessary testing conditions for different clopidogrel salts. They stipulated that if these conditions were fulfilled, as was the case with authorised generic clopidogrel products, then equal efficacy and safety of the originator and generic clopidogrel products can be assumed.
Bioequivalence studies have revealed differences in plasma concentrations of clopidogrel. These are permitted so long as the differences lie within defined limits [2]. Accordingly, the 90% confidence interval for the main parameters AUC and Cmax have to be contained within the acceptance interval of 0.80–1.25. Acceptance limits define a statistical range and the actual mean lies much closer to 1 (or 100%), as numerous studies have reported that the AUC and Cmax for generic products differed only by 3–4% on average from those of the originator product [6-10]. This degree of variability is negligible, particularly when considering the variation in clopidogrel plasma concentrations that occur in different people depending on whether they have just eaten. Clopidogrel is a pro-drug, and through a complex series of metabolic steps is converted to its active thiol metabolite. Initially, however, a substantial proportion of the pro-drug is converted to an inert carboxyl metabolite in the stomach, leaving only a limited amount of the parent substance available for conversion to the active metabolite [11].
Concomitant food intake reduces the stomach’s acidity through a rise in pH because clopidogrel is primarily soluble in an acidic environment this causes a delay in dissolution of clopidogrel salts: less of the inactive carboxyl metabolite is formed, and more parent substance is available to be metabolized into the active thiol metabolite. EMA’s Pharmacokinetics Working Party evaluated the relevant data and noted remarkably high AUC fluctuations, i.e. up to 500–600%, of the parent compound [12]. The originator’s summary of product characteristics however states that clopidogrel can be taken with or without food, which suggests that such food-related fluctuations happen regularly in clinical use of this drug. However, the authority has not yet received any reports of food-related clinical problems.
Plasma clopidogrel concentrations can also be influenced by a person’s genetics, particularly in relation to enzymatic genotypes. The enzyme CYP2C19 which metabolizes clopidogrel exhibits genetic polymorphism. A number of known allelic variants can lead to profound differences in plasma concentration of the active metabolite in up to 20% of patients. Therefore, differences of just a few per cent in plasma concentrations, as seen with different generic clopidogrel salts are considered clinically insignificant.
According to both the Austrian Drug Law/Medicinal Products Act and EU Directive 2001/83/EC of the European Parliament, an applicant is not required to submit additional non-clinical or clinical trial data for a generic medicinal product if it demonstrates bioequivalence [13, 14]. Furthermore, EU Directive 2001/83/EC and the European Guideline on the Investigation of Bioequivalence (CPMP/EWP/QWP/1401/98 Rev.1) state that different salts of an active substance are considered to be the same active substance [2]. If bioequivalence has been proven in terms of there being no meaningful difference in pharmacokinetic properties then different salts will be accepted as having similar efficacy and safety and authorisation will be granted [15]. Austria and other EU countries have licensed generic clopidogrel products on this basis. The concerns that different clopidogrel salts might exhibit altered pharmacodynamic effects were not substantiated from either a pharmacological or from a regulatory perspective.
Different indications
Another area of concern regarding clopidogrel generic products is the differing indications in different versions of product information. These have arisen due to patent restrictions: the reference product includes the indication ‘acute coronary syndrome’ in its product information while most of the generic products do not. While BASG will not officially recommend generic off-label use for the treatment of ‘acute coronary syndrome’, it remains important to explain that the different indications are not the result of any proven differences in efficacy or safety between the products [16-18].
Basically, there are two major reasons that may lead to the listing of different indications between originator and generic products:
- According to the European Community marketing authorisation procedures for medicinal products [19], generic products that are authorised through the mutual recognition procedure may have different, i.e. fewer indications than the originator in the same country. This will happen when the generics applicant uses a reference Member State in which the summary of product characteristics of the reference (originator) product was not harmonised so far, i.e. if there are no EU-wide harmonised indications.
- The second reason, which accounts for the situation with clopidogrel generics, is that while patent protection applies, specific indications of the originator may sometimes only be listed for the originator product, and not for the generic product.
This should allay the concerns expressed, for example, by the ÖKG and some clinicians who perform coronary stent procedures, over the safety and efficacy of clopidogrel generics, and the absence of this indication from product information. Listed below are excerpts from the two different versions of the product information leaflets:
Version 1 (all indications listed)
Clopidogrel is indicated in adults for the prevention of atherothrombotic events in:
- Patients suffering from myocardial infarction (from a few days until less than 35 days), ischaemic stroke (from 7 days until less than 6 months) or established peripheral arterial disease
- Patients suffering from acute coronary syndrome:
- Non-ST segment elevation acute coronary syndrome (unstable angina or non-Q-wave myocardial infarction), including patients undergoing a stent placement following percutaneous coronary intervention, in combination with acetylsalicylic acid
- ST segment elevation acute myocardial infarction, in combination with acetylsalicylic acid in medically treated patients eligible for thrombolytic therapy
Version 2 (fewer indications listed due to patent restriction)
Clopidogrel is indicated in adults for the prevention of atherothrombotic events in:
- Patients suffering from myocardial infarction (from a few days until less than 35 days), ischaemic stroke (from 7 days until less than 6 months) or established peripheral arterial disease
Remarkably, nearly half of all generic products authorised in Austria [20] have begun recently to obviously disregard the patent restriction by listing all the indications. Therefore, differences in the indications no longer apply to many of the generic clopidogrel products.
Maintaining quality of manufacture
The same stringent rules, for example, good manufacturing practice, apply to the manufacture of generic clopidogrel products as for any other medicinal product. However, lower quality batches may be produced on rare occasions. BASG and any other European medicines authority are therefore responsible for strictly monitoring the quality of medicinal products, irrespective of originator or generics source. These measures help to ensure that only top quality products remain in circulation.
Conclusion
The claim that generic products may be qualitatively or clinically inferior generally stems from unsubstantiated individual opinion and unverified case reports. Systematic reviews and meta-analyses have failed repeatedly to show that original products are superior [21, 22]. The predefined acceptance limits for bioequivalence also ensure that plasma concentrations vary only by 3–4% on average from those of the originator product, a range which is deemed to be clinically irrelevant.
BASG/AGES PharmMed maintains that bioequivalence studies are an internationally recognised investigative method, employed by competent authorities worldwide. They are a scientifically accepted tool for demonstrating the comparability of generic and originator products. The pharmacological principle that similar plasma concentrations reflect similar amount of the active substance present at the target sites applies to clopidogrel generics regardless of their salt preparation. They can therefore be assumed to have comparable efficacy and safety.
For patients
Several clopidogrel generic drugs were authorised recently by national and European competent authorities. All of them were thoroughly tested and represent high quality medicinal products. They offer a low-cost alternative for the prevention of atherothrombotic events with an equivalent safety- and efficacy- profile.
Competing interests: None.
Provenance and peer review: Commissioned; externally peer reviewed.
References
- Stellungnahme der Österreichische Kardiologischen Gesellschaft (ÖKG) zu Clopidogrel-Generika. Opinion of the Austrian Society of Cardiology (ÖKG) on clopidogrel generics [homepage on the Internet]. c2010 [updated2010 Mar 30; cited 2012 May 22]. German. Available from: kardiologie-gefaessmedizin.universimed.com/artikel/stellung nahme-der-%C3%B6sterrkardiologischen-gesellschaft-%C3%B6kg-zu-clo
- European Medicines Agency [homepage on the Internet]. CPMP/EWP/QWP/1401/98, Rev.1, European guideline on the investigation of bioequivalence. [cited 2012 May 22]. Available from: www.ema.europa.eu/docs/en_GB/document_library/Scientific_guideline/2010/01/WC500070039.pdf
- Kim SD, Kwang W, Lee HW, Park DJ, et al. Bioequivalence and tolerability of two clopidogrel salt preparations, besylate and bisulfate: a randomized, open-label, crossover study in healthy Korean male subjects. Clin Ther. 2009 Apr;31(4):798-803.
- Neubauer H, Krüger JC, Lask S, Endres HG, et al. Comparing the antiplatelet effect of clopidogrel hydrogensulfate and clopidogrel besylate: a crossover study. Clin Res Cardiol. 2009;98(9):533-40.
- European Medicines Agency [homepage on the Internet]. Pharmacokinetics Working Party [cited 2012 May 22]. Available from: www.ema.europa.eu/ema/index.jsp?curl=pages/contacts/CHMP/people_listing_000070.jsp&mid=WC0b01ac05802327c9&jsenabled=true
- American Medical Association. Summaries and recommendations of Council on Scientific Affairs Reports. Generic drugs (CSA Rep 6, A-02). 2002 Annual Meeting of the American Medical Association. 2002:13-4.
- Henney JE. Review of generic bioequivalence studies. JAMA. 1999;282(21):1995. doi:10.1001/jama.282.21.1995-a
- Nwakama PE, Haidar SH, Yang YS, Davit BM, Conner DP, Yu LX. CDER, FDA, Rockville, MD. Generic Drug Products Demonstrate Small Differences in Bioavailability Relative to the Brand Name Counterparts: A Review of ANDAs Approved 1996–2005. 2006 FDA Science Forum.
- Davit BM, et al. Comparing generic and innovator drugs: a review of 12 years of bioequivalence data from the United States Food and Drug Administration. Ann Pharmacother. 2009 Oct;43(10):1583-97.
- Baumgärtel C. Myths, questions, facts about generic drugs in the EU. Generics and Biosimilars Initiative Journal (GaBI Journal). 2012;1(1):34-8. doi:10.5639/gabij.2012.0101.009
- Nirogi RV, et al. Effect of food on bioavailability of a single oral dose of clopidogrel in healthy male subjects. Arzneimittelforschung. 2006;56(11):735-9.
- European Medicines Agency [homepage on the Internet].Committee for Human Medicinal Products (CHMP) EMA/618604/2008 Rev. 4, Pharmacokinetics Working Party. Questions & Answers: positions on specific questions addressed to the pharmacokinetics working party. [cited 2012 May 22]. Available from: www.ema.europa.eu/docs/en_GB/document_library/Scientific_guideline/2009/09/WC500002963.pdf
- Austrian Medicines Act – Österreichisches Arzneimittelgesetz (AMG), §10. [cited 2012 May 22]. Available from: www.basg.at/uploads/media/Arzneimittelgesetz_04.pdf
- Directive 2001/83/EC of the European Parliament. [cited 2012 May 22]. Available from://ec.europa.eu/health/files/eudralex/vol-1/dir_2001_83_cons/dir2001_83_cons_20081230_en.pdf
- Tschabitscher D, Platzer P, Baumgärtel C, Müllner M, Generika: qualität, wirksamkeit, sicherheit und austauschbarkeit. [Generic drugs: quality, efficiency and interchangeability], Wien Klin Wochenschr. 2008;120(3-4):63-9. German.
- Position of the Austrian Federal Office for Safety in Health Care on clopidogrel generics (26 March 2010). [cited 2012 May 22]. Available from: www.basg.at/uploads/media/100325_Stellungnahme__Clopidogrel_Generika_1.pdf, www.basg.at/uploads/media/100325_Stellungnahme_Clopidogrel_Generika__2.pdf
- Baumgärtel C. Clopidogrel-generics. Austrian Pharmacists Journal, ÖAZ, Ausgabe. 2010;64(11):666-8. [cited 2012 May 22]. Available from: www3.apoverlag.at/pdf/files/OAZ/OAZ-2010/OAZ-2010–11.pdf
- Baumgärtel C. Generics of clopidogrel are equivalent. Vienna Health Insurance Fund and Austrian Health Insurance, WGKK, Therapie Info. 2010;22(3). [cited 2012 May 22]. Available from: www.sozialversicherung.at/mediaDB/692427_therapieinfo_nr3_07_2010.pdf
- European Commission [homepage on the Internet]. 98/C229/03, Commission communication on the Community marketing authorisation procedures for medicinal products, 22.7.98, Official Journal of the European Communities.
- Austrian Medicines and Medical Devices Agency and Austrian Federal Office for Safety in Health Care [homepage on the Internet]. [cited 2012 May 22]. Available from: pharmaweb.ages.at/index.jsf
- Kesselheim AS, Misono AS, Lee JL, Stedman MR, et al. Clinical equivalence of generic and brandname drugs used in cardiovascular disease: a systematic review and meta-analysis. JAMA. 2008;300;2514-26.
- Kesselheim AS, Stedman MR, Bubrick EJ, Gagne JJ, et al. Seizure outcomes following the use of generic versus brand-name antiepileptic drugs: a systematic review and meta-analysis. Drugs. 2010 Mar 26; 70(5):605-21.
|
Author: Christoph Baumgärtel, MD, Department Head, Department Safety and Efficacy Assessment of Medicinal Products, Institute Marketing Authorisation of Medicinal Products & LCM, AGES PharmMed–Austrian Medicines and Medical Devices Agency, and Austrian Federal Office for Safety in Health Care, European Expert in Pharmacokinetics Working Party and Safety Working Party of EMA, Member of Austrian Prescription Commission, 9 Schnirchgasse, AT-1030 Vienna, Austria, www.basg.at |
Disclosure of Conflict of Interest Statement is available upon request.
Permission granted to reproduce for personal and non-commercial use only. All other reproduction, copy or reprinting of all or part of any ‘Content’ found on this website is strictly prohibited without the prior consent of the publisher. Contact the publisher to obtain permission before redistributing.
Related article
What lessons can be learned from the launch of generic clopidogrel?