Roundtable on registries: practical considerations for registries – making them work, London, UK, 26 January 2017
Published on 2017/03/31
Generics and Biosimilars Initiative Journal (GaBI Journal). 2017;6(3):122-34.
Author byline as per print journal: Stuart Bloom, DM, FRCP; Keith Bodger, MBChB(Honours), MD, FRCP; Fraser Cummings, BMSc(Hons), MBChB, MRCP(UK), DPhil, FRCP; Professor Sarah Garner, BPharm, PhD; Professor Kimme Hyrich, MD, PhD, FRCPC; Clare Jacklin; Professor John G Williams, CBE, FRCP
|
Introduction: Registries collect clinical data to evaluate specified outcomes for a population defined by a particular disease, condition or exposure. They can be used to describe the natural history of a condition or disease, determine the effectiveness or cost-effectiveness of products, measure or monitor safety and harm, and measure quality of care. To do this, registries must overcome a series of significant challenges including effective data entry, data security and long-term, sustainable funding. |
Submitted: 26 May 2017; Revised: 19 July 2017; Accepted: 26 July 2017; Published online first: 8 August 2017
Introduction
Registries play a central role in many aspects of patient care. A registry is ‘an organized system that uses observational study methods to collect uniform data (clinical and other) to evaluate specified outcomes for a population defined by a particular disease, condition, or exposure, and that serves a predetermined scientific, clinical, or policy purpose(s)’ [1]. Depending on their scope, registries can describe the natural history of a condition or disease, determine the effectiveness or cost-effectiveness of products, measure or monitor safety and harm, and measure quality of care [2]. Patient registries address a defined population over a period of time, and they may have been selected for a specific disease, condition or exposure. Disease registries focus on a population with a given disease, assessing all of the treatments used and the clinical and patient reported outcomes. Product registries collect information about the use of a single treatment, whether that is a medicine, a class of medicines, a device or a medical intervention [1].
The potential benefits of registries are clear, but registries face many challenges. It is important that patient data are kept secure while at the same time accessible; ideally data need to be entered as part of the routine clinical process and data certainly should not be entered more than once.
Physicians, nurses and patients need to be able to upload data efficiently in challenging settings; different data collections need to be linked to get the most out of the data. The registry also has to manage different levels of access, as some stakeholders require (or may only be allowed) to enter or edit certain types of data. One of the greatest challenges is to set up a funding model that is sustainable in the long term and not dependent on commercial considerations.
A diverse group of speakers and participants, including representatives from the National Health Service (NHS), pharmaceutical industry, healthcare organizations, universities, hospitals, clinics, and patient groups, met to discuss the benefits offered and the challenges faced by registries.
Parallel discussions formed a major part of the meeting and centred on the arrangement for data entry and a requirement for this to avoid duplication and to follow normal clinical care.
The meeting was chaired by Dr Stuart Bloom, a consultant gastroenterologist at University College Hospital, London, UK who is chair of the UK inflammatory bowel disease (IBD) registry.
Methods
On 26 January 2017, GaBI and the Association of the British Pharmaceutical Industry (ABPI) hosted a Roundtable on patient and disease registries in London, UK, with participation by clinicians, nurses, academics, pharmacists, patient groups, regulators, and industry representatives in the UK – participants were selected based on their experience with, or responsibility for, registries in the hospital setting.
The event offered speaker presentations and discussion sessions to provide participants with current views on disease and patient registries in the UK and worldwide.
Results
Registry: how to set up spontaneous reporting – practical challenges, information sharing, multi-source environment, limitation of registry data
Dr Fraser Cummings, Consultant Gastroenterologist at University Hospital Southampton NHS Foundation Trust, UK, outlined the purpose of a registry, see Box 1. Registries increase our understanding of the long-term outcomes of patient care, leading to improvements in patient care. The central tenet of a registry is to input data once, and then use it for many different purposes. Data from different sources are linked while remaining secure.
|
A registry is a collection of information about individuals, usually focussed around a specific diagnosis or condition (NIH 2016). A registry is an organized system that uses observational study methods to collect uniform data (clinical and other) to evaluate specified outcomes for a population defined by a particular disease, condition, or exposure, and that serves one or more predetermined scientific, or policy purposes [1]. The purposes of a registry are:
The potential benefits of a registry are:
The potential weaknesses of UK registries are:
|
At the care delivery and coordination level, registries could provide real time feedback, generate patient level reports and reminders, and can send relevant notifications to providers and patients. Information can be shared with patients and with other providers, and can be linked to relevant patient education.
At the population level, registries provide population level reports (standardized measures; benchmarking; and different reports for different levels of user); they enable ad hoc reports, provide utilities to manage populations or subgroups, generate dashboards, and facilitate third-party quality reporting.
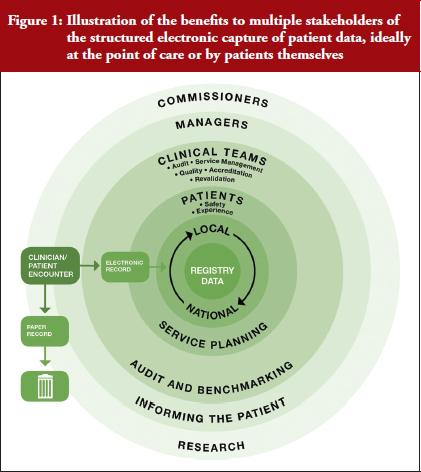
Currently, most clinical data are captured with pen and paper and saved in a patient’s case notes, which provide limited utility given they are generally recorded in an unstructured manner. If data is recorded in a structured way at the point of care, a host of benefits will roll out, see Figure 1.
UK Inflammatory Bowel Disease Registry (IBDR)
Dr Cummings is clinical lead for the UK Inflammatory Bowel Disease (IBD) Registry, which will provide the first repository of pseudonymized IBD adult and paediatric patient data for prospective quality assurance, audit and research purposes in the UK. Bringing this data together will, he said: drive continuous improvement in patient care and access to care across the UK; inform commissioning and service design; improve our understanding of long-term outcomes; provide local, regional and national data in order to better define the pattern of ulcerative colitis and Crohn’s disease; and support IBD research.
The IBD Registry aims to bring about a cultural change in the setting of IBD by maximizing the benefit of data and information to patients, their clinicians and their teams. Key to this is avoiding double data entry and encouraging as far as possible point of care data entry. Data that are added to a registry can subsequently be used for multiple purposes. Standardized data comes under the headings: demographics, phenotype, medications, disease activity scores and patient reported outcome measures (PROM), and are uploaded to NHS Digital via a secure portal.
NHS Digital (formerly Health and Social Care Information Centre) is the national provider of information, data and IT systems for commissioners, analysts and clinicians in health and social care. By using NHS Digital as a data safe haven, the registry can link to multiple other NHS England databases thus facilitating population level analysis of data.
The rewards are clear, but the obstacles are considerable. Data governance is vital, and in light of media headlines of the sharing of patient data with commercial organizations, patients need to be reassured that patient confidentiality is protected and patient data are being used appropriately and for legitimate purposes such as research and public health benefit.
Dr Cummings made the case for pseudonymized data which, unlike fully anonymized data, means that patients can be identified for future research projects; however, key to this is a robust information governance framework and patient consent. Patients must give consent to have their data included in the registry to be linked to their NHS data. Data can be used for both non-commercial research projects and for commercial partners (with clear information governance oversight and patient consent) for research and pharmacovigilance. Patients in the registry will be contacted for future research projects.
It is not clear who should fund registries – the NHS, specialist societies, industry, individual NHS trusts, charities or research funders. The most likely long-term sustainable model is to involve funding from a combination of all the stakeholders. Registries offer numerous collaborative opportunities for UK IBD Biologics audit; UK Quality Improvement Program; Academic (Crohn’s & Colitis UK/Dr Keith Bodger); and industry (pharmacovigilance projects, real-world evidence, specific projects – VEST (the UK Vedolizumab real life experience study in inflammatory bowel disease), and Anaemia service evaluation).
There are currently 23,000 patients included in the IBD Registry with 40 sites actively uploading data and 80 set-ups to upload data. There have been 150 expressions of interest to participate in UK biologicals audit. In 2017, the registry plans to focus on patients treated with biologicals; to work on its funding model (subscription model, pharma projects/support); reporting; data visualization; and pharmacovigilance systems.
Practical experience with a pharmacovigilance registry for biologicals/biosimilars – the BSRBR-RA, a Manchester case study
The British Society for Rheumatology Biologics Registries – Rheumatoid Arthritis (BSRBR-RA) was formed in 2001 and acted as a model for the IBD Registry (outlined above) when it was set up. Kimme Hyrich, professor of epidemiology at the University of Manchester’s Centre for Musculoskeletal Research, UK, told delegates how BSRBR-RA had started as a study of original anti-TNF therapies but had expanded to include rituximab, certolizumab, tocilizumab and most recently biosimilars.
Rheumatoid arthritis affects an estimated 1% of the British population1. The original clinical trials of Remicade and Enbrel included only a few hundred patients so there was some concern about the effectiveness and long-term safety of biologicals when used more widely over prolonged periods of time. Since it is known that spontaneous pharmacovigilance captures only a small per cent of all serious adverse events (SAEs), more systematic data capture of a large cohort of treated patients was needed to assess the true occurrence of SAEs.
¹Symmons D, Turner G, Webb R, et al. The prevalence of rheumatoid arthritis in the United Kingdom: new estimates for a new century. Rheumatology (Oxford) 2002;41:793-800. https://pubmed.ncbi.nlm.nih.gov/12096230/
Professor Hyrich, who is also an Honorary Rheumatology Consultant at Central Manchester Foundation Trust, opened her talk by discussing how patient exposure to a drug progresses from early human studies, where just a few cases are recorded, through to clinical trials and eventually – where the level of exposure is exponentially greater – to post-licensing use. Spontaneous pharmacovigilance has occurred traditionally in this phase, and this is where observational patient registries are now being set up.
Although observational studies in the ‘real world’ have significant advantages over randomized clinical trials in ideal, designed settings, Professor Hyrich urged caution. While observational studies have increased sample sizes, a wider variety of patients and longer follow-up, treatment decisions are no longer randomized, and careful consideration must be taken if comparing outcomes between treatments.
The BSRBR-RA offered a new way of adding to post-licensing data. According to the structure of the BSRBR-RA, an independent academic institution would gather safety data independently of the drug manufacturers and share anonymous safety data with manufacturers as part of a risk management plan. The aim of the registry is not to capture information on every patient with RA, but to capture data on specific groups of patients who are beginning treatment on specific biologicals. Recruitment has been a success, supported in the early years by National Institute for Health and Care Excellence (NICE), and the registry now holds data on over 25,000 patients.
Industry is accustomed to collecting and managing data, but collecting data from an independent source, and sharing it, is key to the success of registries like BSRBR-RA. The BSRBR-RA is overseen by the British Society of Rheumatology, which conducts negotiations with manufacturers and allows academics to analyse the data independent of manufacturers to address questions about safety and effectiveness both for individual products and across products. It is supported financially almost exclusively by participating pharmaceutical companies.
For pharmacovigilance, the registry is required to report SAEs without patient or doctor identifiers, along with six monthly aggregated reports to manufacturers to help them monitor the safety of their products.
A large amount of clinical and patient data are collected six monthly and the registry is linked to NHS Digital for death and cancer outcomes. There are plans to link data with Hospital Episode Statistics (HES) and other databases, but the BSRBR-RA is currently navigating the governance issues and permissions that need to be in place to facilitate these linkages in the absence of explicit patient consent.
The study in 2001 pre-dated the widespread use of online data capture and, since there is currently no universal rheumatology electronic medical record (EMR) and no national database of biologicals prescribing, all the data in the registry (currently 21,000 patients including 40,000 different treatment courses) are collected by direct report. Although originally data were collected via paper forms completed in the hospitals, a web-portal to capture data online is in development.
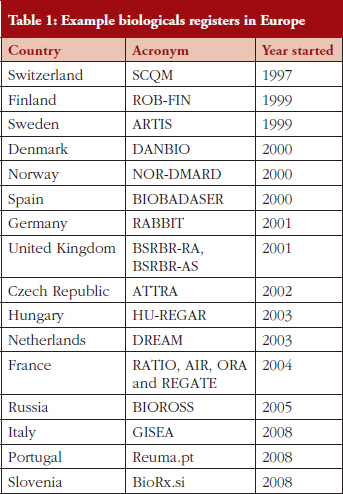
The BSRBR-RA is not the only rheumatology register in Europe capturing outcome data on biological drugs, see Table 1. The structure and design of each register differs but is designed to fit with the local healthcare system. Traditional cohort models, such as the BSRBR-RA, have the advantage of collecting extensive patient-level data but are hard work at the local level. Registries in Denmark, Sweden and Switzerland (similar to the IBD Registry) are embedded in EMRs, which have the advantage of potentially larger sample sizes.
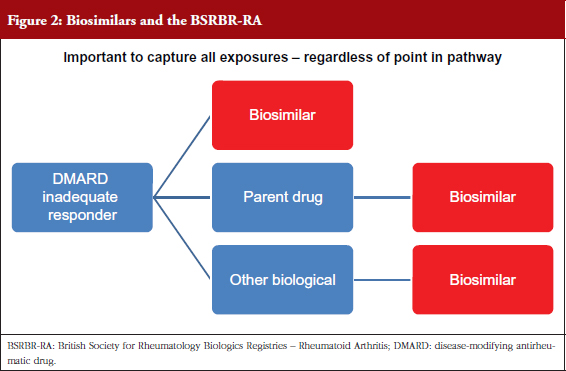
Biosimilars pose a new challenge, e.g. data collection, switching, to the sustainability of healthcare systems [2], and BSRBR-RA aims to capture exposure to biosimilar drugs as well as to biological originators, see Figure 2. It is not known how many patients will switch to a biosimilar, or at what stage of their treatment [3]. The information is vital in case of adverse drug reactions (ADRs) or loss of efficacy after a switch has taken place, as it would allow accurate identification of the products – whether the product is an originator biological or a particular biosimilar [4]. Ideally, a registry would include the exact date of switch (which may not be recorded in the medical record), which would allow greatest accuracy about the suspected products. It is not possible for a register like the BSRBR-RA to capture batch number of the product.
Assessment of patients’ disease activity may not always be assessed by a healthcare professional at the point of switch, so arthritis disease activity measures may not be recorded. Biosimilar switching is happening at many UK centres and there is a view that this is involving considerable numbers of patients. Capturing data from every patient who switches poses a challenge to a register such as the BSRBR-RA alongside the NHS providers who record data for the register, but it is hoped that, in the future, electronic data capture will help facilitate this.
Registries and Health Technology Assessment: a view from NICE
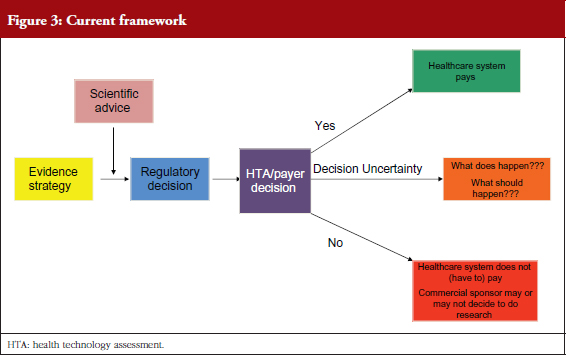
Professor Sarah Garner, Associate Director of Science Policy and Research at NICE, UK, explained how NICE uses health technology assessment (HTA) to provide guidance to the NHS, see Figure 3. HTA is a multidisciplinary process that summarizes information about the medical, social, economic and ethical issues related to the use of a health technology in a systematic, transparent, unbiased and robust manner.
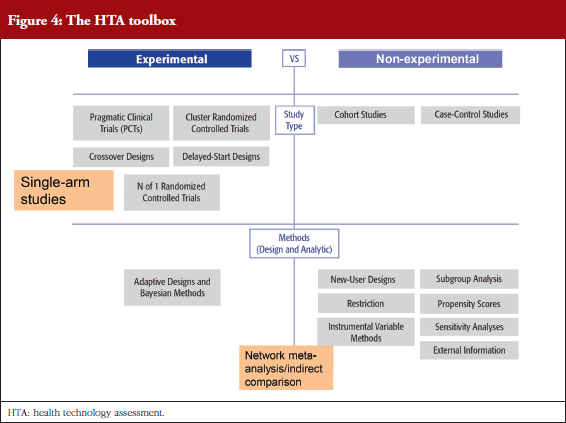
The benefits of randomized clinical trials (RCTs) decrease with a move towards personalized medicine and treatment for rare diseases where patient numbers are very small. This underlines the need for alternative tools in the HTA toolbox, see Figure 4. It is illogical, said Professor Garner, that the considerable data resource provided by registries cannot be used to inform clinical practice. Professor Garner is calling for investment in registries.
Professor Garner is currently working with the Innovative Medicines Initiative (IMI), a European Commission funding route in which academics, policy stakeholders and industry work together to find solutions to science policy issues. NICE plays a significant role, benefitting from the large patient numbers compared with those in the UK alone, and Professor Garner hopes that it will not be affected by Brexit. Projects include GetReal, which aims to show how robust new methods of real-world evidence collection and synthesis could be adopted earlier in pharmaceutical R & D and the healthcare decision-making process (https://www.imi-getreal.eu/), and ‘Big data for better outcomes’ which seeks stakeholders’ input on the scope and desired impact of a proposed IMI2 programme focused on maximizing the potential of big data in health care.
Professor Garner concluded by noting the call made for a UK-wide strategy for disease registers back in 2000, and that, although things were moving in that direction, there needed to be much better coordination and strategy as this capability is one of the UK’s best assets in terms of global competitiveness for research investment.
Linkage of registry and routine administrative datasets for pharmacoeconomic research
Dr Keith Bodger, Research Chair for the UK IBD Registry and a consultant gastroenterologist in Liverpool, UK, used IBD to describe the potential for combining selected data from a chronic disease register with information extracted from routine hospital administrative data to generate real-world data.
He began by quoting Sir Michael Rawlins, who has said that ‘RCTs, long regarded as the “gold standard” … have been put on an undeserved pedestal. They should be replaced by a diversity of approaches that involve analysing the totality of the evidence base’.
Professor Rawlins has previously noted that observational studies are useful and, with care in the interpretation of results, can provide an important source of evidence about both the benefits and harms of therapeutic interventions. The ABPI has published guidance on demonstrating value with real-world data (www.abpi.org.uk/our-work/library/guidelines/Pages/real-world-data.aspx).
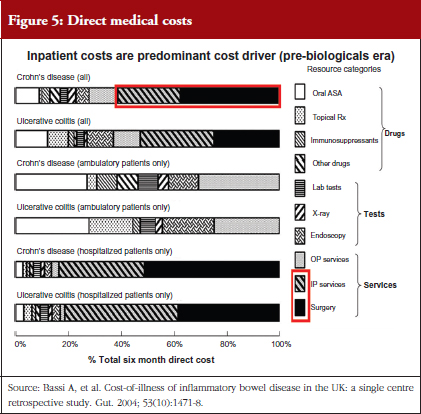
Research has shown that patients enrolled in RCTs do not adequately represent the IBD patient population encountered in routine settings – many IBD patients would not qualify for biologicals trials and yet they are treated successfully with these agents in practice. Surprisingly little is known about the cost of treatment in the UK. Traditionally, unplanned hospital events and emergency admissions have been the most expensive component of IBD treatment. Dr Bodger published a study in 2004 (Bassi A, et al. Gut. 2004;53(10):1471-8) in which his team recorded the direct medical costs of treating IBD over six months at his hospital. Although a single centre study, it is cited regularly and the work has provided cost inputs for independent and industry-led health economic modelling studies as part of NICE appraisals of biological agents in IBD, see Figure 5. He highlighted the need for larger scale and more up-to-date studies, particularly given the growth in use of biologicals over the past decade. Unlike traditional drug treatments for IBD, the annual cost of a biological drug is comparable to that of a hospital admission for major surgery. Economic evaluations in IBD are currently limited by a lack of real-world data both on resource use and longer-term outcomes.
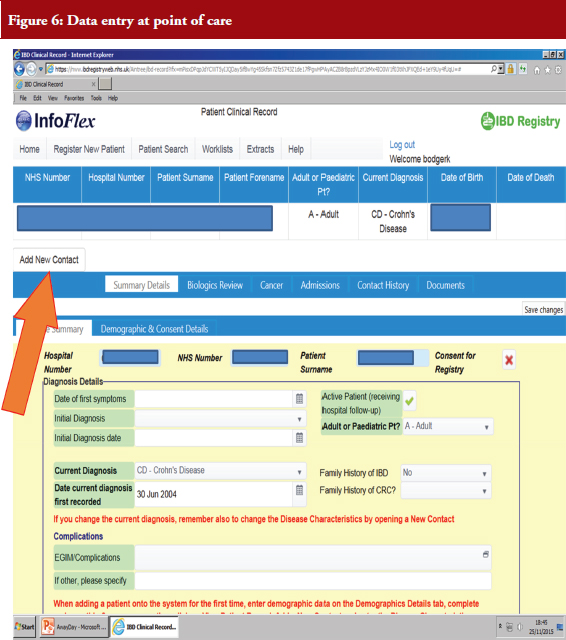
Dr Bodger hopes to combine routinely, ubiquitously collected simple data, like HES, and selective collection of data items in the IBD Registry. His team looked at early data from the IBD Registry and found considerable outpatient and medication data but no systematic recording of hospitalization events in the system, see Figure 6.
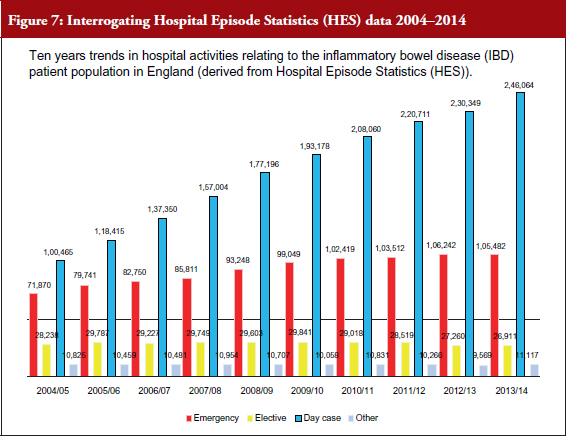
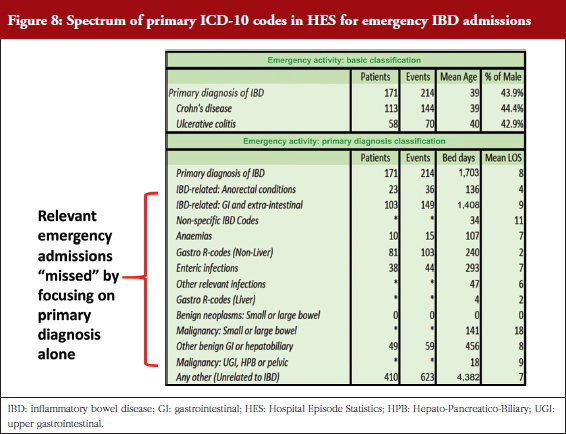
Dr Bodger’s team has been supported by funding from Crohn’s & Colitis UK to try to get clinical input into all stages of interrogation of routinely collected data – harnessing clinical expertise to extract best value from the data. Working with the IBD Registry, the team has analysed 10 years of anonymized HES data, examining all-cause inpatient episodes relating to patients coded with IBD. Local-level reports for individual hospitals were produced and shared with clinical teams, who have responded positively to the value of the data and provided advice on improving the way HES data is interrogated, see Figure 7. The pattern of primary diagnoses recorded for emergency admissions among IBD patients is quite varied and complex. Algorithms have been developed to identify admissions for IBD care where non-specific codes or disease complications were coded rather than the limited set of IBD-specific diagnosis codes, see Figure 8.
Sharing data that had been interrogated in this way with clinicians enabled a discussion about what clinicians found helpful and not helpful, and where to go next. His team is now looking at readmission rates and identifying key clinical and economic events such as hospitalization and surgery with the primary purpose of providing metrics to support local care and service evaluation.
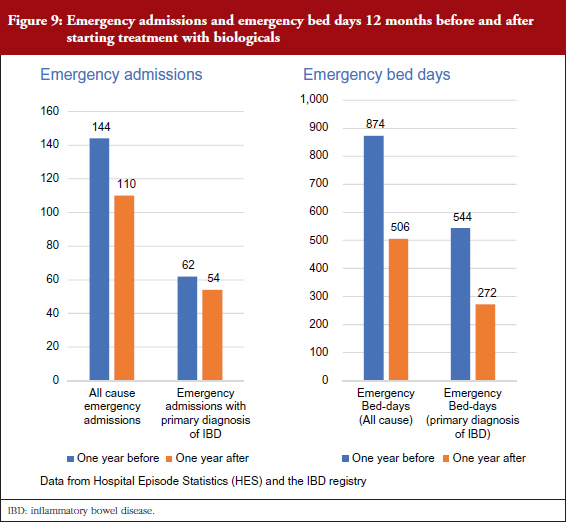
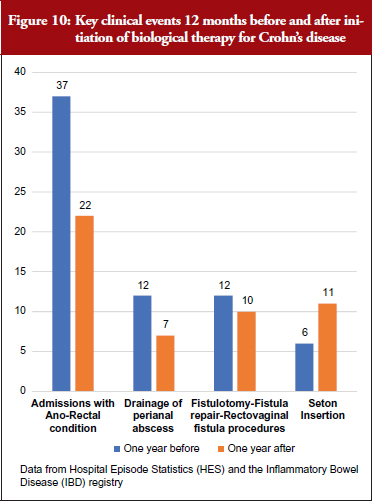
Supported by anonymized linkages undertaken by NHS Digital, the project has been able to explore the value of combining data from the IBD Registry with metrics derived from HES data. For a cohort of biologicals-treated cases, they identified the drug start date from IBD Registry data and used this time-point to interrogate HES events for the 12 months before and after starting treatment. They started by looking at emergency admissions and emergency bed days, see Figure 9, and drilled down to key clinical events over that time, see Figure 10.
Evaluating the cost-effectiveness of high-cost biological therapies for chronic diseases has been limited by a lack of long-term data on real-world costs and outcomes. Models have often relied on simplistic assumptions, sparse empirical data or expert opinion when simulating downstream events beyond the timescale of clinical trials. The UK IBD Registry is set up to capture much of the data required, and this combined with information extracted from routine hospital administrative data will generate real-world data that supports local service evaluation and quality assurance with potential to inform future pharmacoeconomic research.
Enhancing the role of routinely collected clinical data in a registry setting, and to support pharmacovigilance
The successful operation of a disease registry poses many challenges, not least the collection of accurate clinical data in sufficient depth to be valuable, said Professor John G Williams, Professor of Health Services Research at Swansea University, and Director of the Royal College of Physicians’ Health Informatics Unit. Professor Williams started by defining a registry, and analysing the strengths and weaknesses, see Box 1.
Professor Williams noted that the rigour paid to data quality and pharmacovigilance during clinical trials is ‘non-existent’ for data collected for other purposes [5]. Registries need to be converged with data collected at the point of care. Clinical data collection in the UK could be improved.
There is no single ideal pharmacovigilance system. Different approaches – from the Yellow Card Scheme (the UK system for collecting information on suspected ADRs) to post-marketing surveillance, to registries – currently complement each other. The Yellow Card Scheme is an excellent method to raise signals, but not good for testing hypotheses of association or estimating incidences (risks); analytical studies are good for testing hypotheses of association but less good at raising signals.
Dedicated data collection for registries is not sustainable in the long term and the Royal College of Physicians has been addressing the issues facing clinicians as they strive to maintain many and disparate data collections in a busy clinical environment. There are at least 250 registries in the UK currently, and the number continues to grow for an ever-increasing number of diseases and interventions. A set of national standards for the structure and content of records and communications is being developed, alongside the use of new technologies to simplify human–computer interaction by both professionals and patients.
The aim is to enable use of data recorded once at the point of care to inform both individual patient management, and aggregate analysis for other purposes, including pharmacovigilance, said Professor Williams.
Professor Williams divided analysable patient data into three categories: operational data captured at the point of care; routine data collected continuously as a by-product of care, e.g. HES; and designed data, which is bespoke for audit, research or other purposes. These need to be aligned.
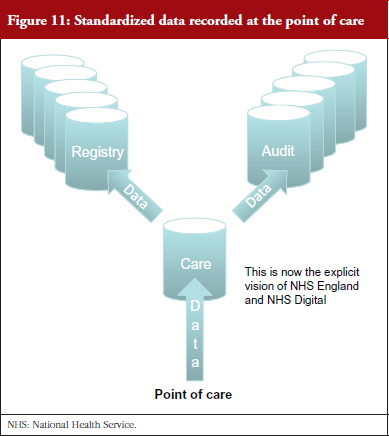
Professor Williams explained that data recorded in patient records at the point of care need to feed into any of these categories. This is now the vision of NHS England and NHS Digital, see Figure 11. Achievement of this vision depends upon the widespread adoption of national standards for the structure and content of records, including the headings in the record, and clinical terms used. Currently, clinical data from hospitals are not standardized, and lack sufficient breadth, depth and quality. They are not timely and they do not meet good practice requirements applicable to research systems.
Standardized data recorded at the point of care in electronic patient records will make data collection sustainable, said Professor Williams. It is important that what is being done now is ‘future proofed’ by taking a standards-based approach so that the vision that is now spelt out by NHS Digital and NHS England takes root, see Figure 11.
Registries must conform to standards when they are developed. This means technical standards for the systems, information standards for the data, and professional standards for the structure and content of the record. The UK has these professional standards, and these have been endorsed by the Academy of Medical Royal Colleges, the Professional Record Standards Body and NHS Digital, and the requirement to use them is explicit in national policy and NHS contracts. Standards for clinical terms are embodied in SNOMED-CT. The economic benefits of using streamlined data processes are underlined in a report published in 2014 by Volterra (http://volterra.co.uk/wp-content/uploads/2014/09/Final-EMC-Volterra-Healthcare-report-web-version.pdf).
‘Registries are a challenging solution to the need for comprehensive data, including for pharmacovigilance’, concluded Professor Williams. Until ‘point of care’ data recording improves there is a need for registries to be populated by parallel data collection, but this is inefficient, and unsustainable in the long term.
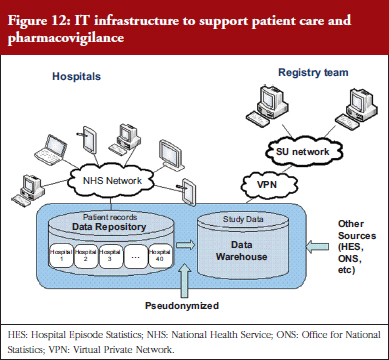
Professor Williams illustrated the direction in which pharmacovigilance is heading, and how registries should evolve, with the example of the Comparison Of iNfliximab and ciclosporin in STeroid Resistant Ulcerative Colitis: a Trial (CONSTRUCT) (an RCT comparing infliximab and ciclosporin in steroid resistant ulcerative colitis; http://bmjopen.bmj.com/content/4/4/e005091). The IT infrastructure for this trial enabled point of care recording to feed the designed data requirements for the trial. Practitioners collected data that went into a central data repository. Data could only be identified by the hospitals that entered that data (not by the research team or by other hospitals). Pseudoanonymized data were saved to a data warehouse, which the research team could access, see Figure 12.
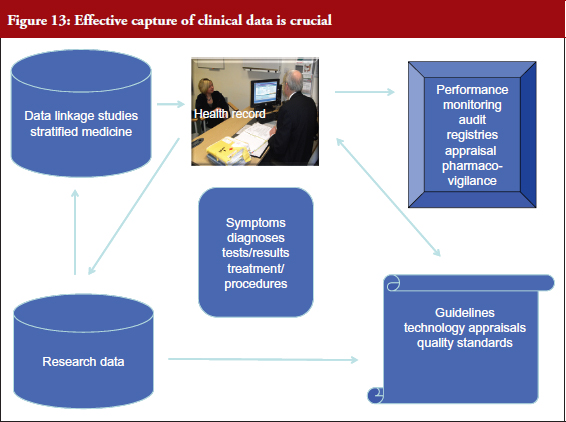
Professor Williams called on delegates to incorporate national standards for clinical data structure and content (particularly the clinical headings and terms) in their registries now to ensure transition to the future. In that way, standardized data recorded at the point of care will not only be able to feed into national registries, but also provide comparable data for research using linked data and enable the development and implementation of stratified medicine. They will also feed into NICE guidelines. The terms used in guidelines should be the same as the terms found in the records, see Figure 13.
Patient perspective on biosimilars safety data and other concerns
Clare Jacklin, Director of External Affairs at the National Rheumatoid Arthritis Society (NRAS), UK, reviewed her members’ perspective on biosimilars and the role played by BSRBR-RA, discussed earlier by Professor Kimme Hyrich.
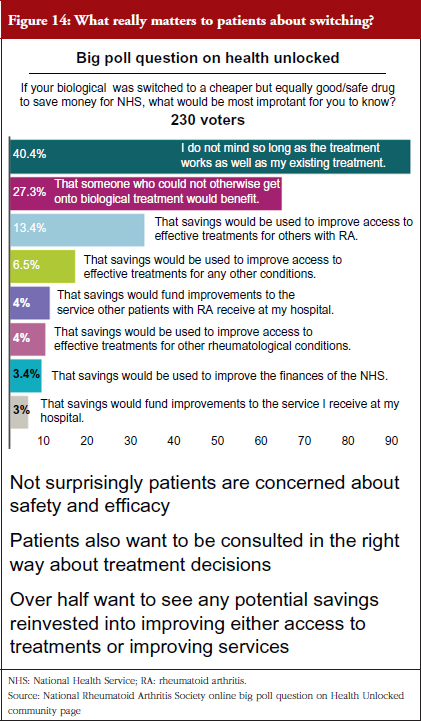
NRAS believes that all stakeholders – patients, clinicians, funders and industry – require robust and reliable data to accurately measure the true impact of biologicals, including biosimilars. All stakeholders, patients in particular, want the reassurance that only longitudinal data can accurately give. They want the reassurance that their long- and short-term health is not being (and has no potential to be) compromised for the sake of cost-saving initiatives, such as switching to a less expensive biosimilar, see Figure 14.
NRAS, and those people with RA who the charity represents, would be concerned if biosimilars were not recorded in registries in the same way that their biological innovators are today. There are concerns that potential savings gained by the introduction of biosimilars might not be invested in research into RA and improving patient care. Despite the robust regulatory pathway for biosimilars in Europe to demonstrate quality, safety and efficacy, some patients continue to harbour concerns that there may be a difference in efficacy between products. Above all, NRAS contacts want to be involved in decision-making in this area, particularly on the decision to switch between biosimilars produced by different companies.
NRAS, whose position on biosimilars is available online at www.nras.org.uk/biosimilars, is collaborating with BSRBR-RA on the production of patient information to encourage patients to sign up to the register. NRAS has updated its position on biosimilars twice since 2014 to reflect current data on safety and efficacy and pragmatism about what is happening on the ground.
NRAS highlights four key areas that need to be addressed: (1) patients must be properly informed through robust shared decision-making mechanisms about being switched; (2) manufacturers must agree to long-term safety data collection through BSRBR-RA; (3) financial savings must benefit rheumatology services and patients in an equitable way; and finally; (4) both NRAS and British Society for Rheumatology (BSR) would like to see NICE review the health economic model as they believe that, with the drop in prices, there is a potential to reduce the threshold to access biological treatment for some patients with specific disease profiles allowing them to access such treatment at an earlier stage of their disease progression.
Summary of discussion about the speakers presentations and panel discussion
The challenges of setting up a registry
Healthcare professionals need guidance – particularly from NHS England – on data collection and the importance of registries. It was agreed that all prescribing information should be recorded, i.e. not just noting that a medicine was a ‘biosimilar’ and that patients should never receive different versions of a medicine without their or their physician’s knowledge.
It was suggested that NICE should mandate data collection before approving a new medicine. However, since NICE does not currently have a national mandate for this, nor the funds to carry out such a role, this is currently not possible. There is also an ethical question; is it reasonable to say that – without a data collection system set up – a patient cannot be treated with the preferred drug? Those keen to press the issue of mandatory data collection in the UK should contact the Chair of NHS England to outline the changes needed.
Setting up a new national registry is currently unlikely because it would need to be funded in perpetuity and the UK is currently unable to fund such an undertaking. Nordic countries have succeeded where the UK is failing because they have a different tax system, more money and a smaller population. They also have a different relationship with the state in relation to data capture.
All national registries will need to agree on a shared set of headings under which data is collected. This must be standardized across all registries to ensure quality of data is maintained.
A major problem holding back data collection in the UK is the fragmentation of national infrastructure. There was a view shared that multiple organizations including the Department of Health (DH), NHS England, NICE and NHS Digital are not working together on a coordinated strategy. More data collection cannot be mandated until NHS Digital and local funders collaborate to provide the means for that at the point of care.
Delegates agreed that a link between registries and primary care data would be welcome, but primary data obtained through the Clinical Practice Research Datalink (CPRD) is currently prohibitively expensive and requires training before it can be accessed.
Data collection
There was agreement that clinicians, particularly those on hospital wards, are slow to move away from recording information with pen and paper. Hospital wards might be particularly behind on data collection because, while general practitioners (GPs) are paid for every item of service, secondary care is relatively insulated from payment. There is much less of a driver to record information. Nevertheless, the failure to collect data electronically is surprising, given the ubiquity of electronic data entry around us, notably in shopping transactions and banking.
Although the problem is not unique to the UK, there are examples of countries – notably Sweden – where electronic data entry is in place and is saving physicians time and improving patient care. The Swedish example discussed had taken 15 years to set up, so the UK cannot expect to change overnight without proper investment in IT infrastructure and training.
The arrival of a new generation of clinicians and patients who will only be familiar with electronic data entry, and not have grown up with pens and paper, is sure to fuel the move to electronic data capture. In addition, the NHS has the ambition to go paperless by 2020 which will advance electronic data capture.
The digitalization of services is extending across health and social care as outlined in the National Information Board objectives, which will enable electronic data capture. Pharmacists are already beginning to adopt electronic prescribing systems and they can play an important role data collection from pharmacy services.
Financial incentives
The question of whether the study presented by Dr Keith Bodger, which revealed the cost of patient care before and after the introduction of biologicals, see Figures 11 and 12, could be used to support future funding applications was raised. Dr Bodger said that this should be possible but there are currently no data at all about patient-level costing. There are data on drug costs, but no data on nursing or clinic costs. This can make it look as though more expensive drugs mean more expensive care. Delegates agreed that the costs of care needed to be understood.
The benefits of switching to the most cost-effective biological are not always shared with all stakeholders meaning that many are immediately disenfranchised.
Dr Fraser Cummings shared his experience with a gain-share agreement, where improved performance is rewarded with financial gain shared between different stakeholders. Dr Cummings’ gain-share experience goes back five years to his original biologicals service at Southampton where his team demonstrated through audit that they had not been screening patients properly. There were patients who had been on infliximab for six years and who had never seen a clinician in that time. This represented a huge clinical need in terms of patient safety and effectiveness. By systematically moving patients onto the most cost-effective infliximab product (which was a biosimilar) and putting larger numbers of patients into research, the biologicals bill was cut by GBP 300,000 a year, this sum was shared between commissioners and the hospital trust, but a certain amount went directly to the clinical service provider to facilitate data entry and patient education. Despite this success, the concept that making an investment can end up saving money is not widely appreciated.
Data linkage
Data linkage is central to public health practice. Linking together the data from numerous sources – GPs, hospitals, clinics and more – on a national scale involves numerous separate databases. In 2013, the UK Medical Research Council invested GBP 20 million to establish The Farr Institute of Health Informatics Research – a UK health informatics research institute.
The Farr Institute incorporates 21 academic institutions and health partners in England, Scotland and Wales. It does not own or control data but analyses data to better understand the health of patients and populations. By connecting diverse molecular, phenotypic, health and non-health datasets at scale in this way, the core activities of the UK’s health and medical bioinformatics research community can apply cutting-edge data science approaches to address major UK-wide challenges.
There is currently no single package in the programme of work at the Farr Institute that relates to data provenance. Responsibility must be taken for the quality of the data being linked and the provenance of that data. This is important because the Farr Institute is a UK-wide research collaboration that extends beyond even the Department of Health, NHS England and Trusts.
The Farr Institute is not the only setting in the UK where data linkage takes place. NHS Digital, Public Health England (PHE) and the Clinical Practice Research Datalink (CPRD) are also involved in data linkage on a national level, and at a regional level through organizations such as NorthWest EHealth and Connected Health Cities.
In addition to linkage between data, there is a growing need for linkage, or collaboration, within the clinical community at large. Professional bodies, as well as the clinical academic community, have a major role to play in leadership.
Health technology assessment
Helping clinicians gather data for ‘real world research’ is not straightforward. Professor Sarah Garner at NICE called on delegates for help in unpicking the statutory responsibilities of the different NHS organizations. There is often resistance to supplying data to NICE. NICE needs to know who to ask, and Professor Garner called on clinicians to publicize what NICE is trying to achieve. NICE gets a lot of negative reports in the academic press, and this prevents productive interaction. Different organizations need to pull together, rather than travel in different directions.
Patient engagement
It is important that patients know the value of their data that has been collected. Knowing where best to publicize these findings is ongoing – from leaflets in patients’ waiting rooms to improved, accessible websites. There is no one solution; different patients find information in different settings. Improving communications is key – for example, with Facebook pages – to encourage ongoing patient engagement.
Group discussions (summaries from moderators of the parallel discussion work groups)
Participants were divided into four discussion groups, each of which reported back a summary of their findings to the meeting.
Sarah Critchley of Pennine MSK Partnership Ltd chaired a group that talked about the challenge of limited resources, manpower and busy clinics when inputting data to a registry. The question of who inputs the data was discussed, focussing on specialist nurses and, where Ms Critchley works, healthcare assistants and apprentice healthcare assistants. Dedicated data clerks were discussed, alongside the practical challenge of patients inputting their own data.
In a perfect world, a perfect system would talk to all other perfect systems, but currently some systems talk to no other systems. For example, the system used by Ms Sarah Critchley does not talk to the GP system, which does not talk to the hospital system. Greater IT systems development and interoperability measures are needed to fix this so that systems do communicate, while maintaining data security and patient consent.
It was agreed that pharmacovigilance was poorly understood, not just by patients, but also clinicians and nurses. Industry and regulators have a key role to play here, to explain pharmacovigilance relating to drug safety and public health, and for pharmaceutical companies to meet regulatory requirements.
While people can access registry data, it was not well understood how this works. Local units, not just major organizations, can access data for their studies.
Finally, this discussion group agreed that registries benefit patients by improving safety and reassuring patients that their data is of value. Patients are reassured that their data will enable registries to describe the natural history of a condition or disease, determine the effectiveness or cost-effectiveness of products, measure or monitor safety and harm, and measure quality of care. It is important to think carefully about how the use of registry data is fed back to patients. Registries will also help on the issue of switching; there is currently very little switching data in the UK, but switching data could be accessed from other countries with more experience of switching.
Ms Critchley’s group produced three action points: (1) finding ways to link data systems and registries; (2) determining who would input the data and how; and (3) improving patient understanding of registries.
Professor Chris Probert from the University of Liverpool chaired a group that looked at the challenges from the patients’ perspective. The group discussed how patients with multiple sclerosis (MS), rheumatology or IBD could input their own data, and how apps and related technology would be better than filling in forms on paper. Integrating this information with information from clinicians would be key. Likewise, linking this data with HES data was clearly valuable. The group discussed how registries were funded in different ways – the BSRBR-RA is funded by industry, whereas the MS Registry is funded by the MS Society – and how NHS England has made certain drugs available only if a patient is on a registry (which has advantages but comes with problems associated with patient consent).
The benefits to patients of registries were discussed – with some patient groups (particularly MS and IBD) signing up for altruistic reasons so other patients can also benefit, and other patients with rare diseases who find that registries give them a sense of belonging, they are no longer alone or forgotten.
Dr Nick Kennedy of the Royal Devon and Exeter NHS Foundation Trust chaired a group that focussed on funding. BSRBR-RA and BADBIR (the British Association of Dermatologists Biologic Interventions Register) are both industry funded and have fixed costs – covering the operation of the registry – and variable costs – per patient on a particular drug. The IBD Registry is different, funded by pharmaceutical manufacturers and by private donors, charities and the British Society of Gastroenterology. The group recognized that funding models will change as contracts come to an end, and noted that the arrival of biologicals and now biosimilars will see this evolve further.
Registries are an important tool to gather data for pharmacovigilance, quality improvement and research. However, there are also great opportunities to link registry data with other sources of data. Linking with HES, cancer and mortality data allows access to important outcome data which may be missed, particularly as patients move around the country. Patient-reported data will pick up, for example, infections that might not make it into the clinical record.
Dr Kennedy’s group also looked at health economics. Registries can be used to understand the cost of managing different diseases in the real world. They provide an opportunity to link data sources with individual patient consent, and also to fill in information that is not easily attained from existing prescribing and hospital episode data.
The sustainability of registries is important. Less is more – not necessarily capturing everything, but thinking about the information needed. Ideally, data capture should be embedded into clinical practice, and the number of questions asked therefore needed to be kept manageable.
Ann Jacklin of NHS Improvement chaired a discussion group which concluded that registries cannot and should not be used in isolation for health economics. The datasets are too big and registries need to be linked to other point of care systems.
It is important that registries standardize their data fields and that data is collected at the point of care. To improve pharmacovigilance it is important to decide what needs to be collected in a standardized way.
Ms Ann Jacklin’s team was not optimistic that the current funding problems would be overcome soon. It was agreed that registries are not good at defining who should be funding them. ‘Everyone thinks they’re great, but nobody thinks they are their responsibility’. Registries need a ‘clear sell’. Is it legitimate to say they all have different purposes? Work must be done to standardize data in registries so they align with information held by NHS Digital.
Conclusions of the Roundtable meeting
A diverse group of stakeholders attending this Roundtable on registries presented and discussed their views on registries now and in the future, which raised a number of issues. This GaBI meeting was applauded for offering a rare opportunity to network between different specialists and disease areas. There is little time available for such multidisciplinary meetings, although clearly different groups share common challenges.
There is considerable variation between registries and how they are managed and it will be important in future to have a standardized and sustainable model. The diversity of registry models impedes funding; since it is not always clear which organization a registry should apply to for funding. Agreeing on exactly which data should be collected – and under which headings – will make it possible to link data between different registries and hospital records and increase the likelihood of one day being able to enter all data into NHS Digital (or its equivalent) in 10 or 20 years’ time.
Ensuring that patient data are secure is key to the success of registries, but this must not be at the expense of an effective system of pharmacovigilance. Consent is often brought up in arguments against data sharing, which is key to effective pharmacovigilance and how effectiveness and cost-effectiveness are measured.
The Royal College of Physicians’ view for the future, as presented by Professor John Williams, was roundly welcomed. Registries, as they exist today, are not sustainable. Multiple patient registries and collecting data at the point of care are the ideal. It could take over 10 years to achieve this, and will require a lot of thought before it can fulfil the need of the multiple stakeholders.
How registries will perform in the future was central to discussions. ‘People working on registries now need to work with the people who are working with the visions for the future to ensure that what we have now will not be lost and that it can streamline into future data capture’, concluded Professor Kimme Hyrich of BSRBR-RA. ‘What we do have is an incredible natural history of these diseases in the era of biologicals that should never be forgotten’.
Key findings of the Roundtable meeting
The conclusions of the Roundtable meeting can be summarized in the following priorities:
- We need clearer alignment on the vision of registries in the future. The NHS and Departments of Health could play an instrumental role in setting out a clear policy and strategy for registries in the UK.
- Best practice would be facilitated by establishing a good model for the setting up, funding and sustainability of registries. Linking registries to frontline services to provide value to patients and their healthcare professionals is key. A partnership approach is needed to link registries with primary care data.
- Collaboration amongst stakeholders is necessary for the sustainability of registries, as they are resource-intensive (in terms of time and money) assets for health care.
- Improvement of data collection and standards in registries is needed. The new UK Institute for Health and Biomedical Informatics Research could play a central role in this effort.
- Long-term success for registries depends on effectively outlining the value and benefits of patient registries for stakeholders, i.e. patients, clinicians, regulators, HTA bodies, and industry.
- To make progress in the UK on registries, we need practical next steps. A multi-stakeholder task and finish group may be a constructive way to support the development of an NHS policy on registries, particularly as they relate to initiatives to improve patient outcomes and medicines optimization.
Speaker Faculty, Moderators and Participants
Speakers
Stuart Bloom, DM, FRCP, UK
Keith Bodger, MBChB(Honours), MD, FRCP, UK
Fraser Cummings, BMSc(Hons), MBChB, MRCP(UK), DPhil, FRCP, UK
Professor Sarah Garner, BPharm, PhD, UK
Professor Kimme Hyrich, MD, PhD, FRCPC, UK
Clare Jacklin, UK
Professor John G Williams, CBE, FRCP, UK
Moderators and Co-moderators
Sarah Critchley
Associate Professor Susanne Cruickshank, PhD
Julie Duncan, MSc
Richard Gardner, MA
Sagair Hussain, BSc (Hons), MSc, MBA, PhD
Ann Jacklin, BPharm
Nick Kennedy, MBBS, FRACP
Professor Chris Probert, MD
Participants
Catherine Akers, Amgen Ltd
Caroline Boulliat Moulle, PharmD, Amgen Ltd
Sarah Critchley, Pennine MSK Partnership Ltd
Associate Professor Susanne Cruickshank, PhD, Royal College of Nursing
Sally Dickinson, National Ankylosing Spondylitis Society (NASS)
Julie Duncan, MSc, Guy’s and St Thomas NHS Foundation Trust
Aileen Fraser, MSc, University Hospital Bristol
Shelley Gandhi, MSc, NDA Group AB
Richard Gardner, MA, British Society of Gastroenterology
Dr Sagair Hussain, MBA, British Association of Dermatologists
Ann Jacklin, BPharm, NHS Improvement
Gareth Jones, BSc (Hons), MScEcon, PhD, University of Aberdeen
Nick Kennedy, MBBS, FRACP, Royal Devon and Exeter NHS Foundation Trust
James Kent, Southend Hospital NHS Foundation Trust
Alastair Kent, OBE, Genetic Alliance UK
Professor Jayne Lawrence, Royal Pharmaceutical Society
Katie Marchbank, PhD, AbbVie Ltd
Kathleen McElhone, PhD, British Association of Dermatologists
Dr Stoyan Minchev, MBA, Amgen Ltd
Claire Munro, MA (Hons), IBD (inflammatory bowel disease) Registry UK
Professor Chris Probert, MD, University of Liverpool
Alan Roach, PhD, British Society for Rheumatology
Imogen Scott Plummer, MSc, MS Society
Sunayana Shah, Association of the British Pharmaceutical Industry (ABPI)
Alison Shaw, PhD, Medicines and Healthcare Products Regulatory Agency (MHRA)
Martin Sheppard, MPharm, MRPharmS, Southend Hospital NHS Foundation Trust
Helen Terry, Crohn’s & Colitis UK
Belinda Theis, University College London Hospitals NHS Foundation Trust
Hannah Yarrow, Guy’s and St Thomas’ NHS Foundation Trust
Acknowledgement
The Generics and Biosimilars Initiative (GaBI) wishes to thank Dr Stuart Bloom for his strong support through the offering of advice and information during the preparation of this Roundtable meeting.
The authors would like to acknowledge the help of all the Roundtable speaker faculty and participants, each of whom contributed to the success of the meeting and the content of this report as well as the support of the moderators: Ms Sarah Critchley, Associate Professor Susanne Cruickshank, Mr Richard Gardner, Dr Sagair Hussain, Ms Ann Jacklin, Dr Nick Kennedy, Professor Chris Probert, in facilitating meaningful discussion during the parallel group discussions, and presented the discussion findings at the meeting.
The authors wish to thank Dr Bea Perks, GaBI Journal Editor, in preparing this meeting report manuscript and providing English editing support on the group summaries and for finalizing this manuscript.
Competing interests: The Roundtable meeting was sponsored by an unrestricted educational grant to GaBI from Amgen Inc.
Provenance and peer review: Not commissioned; externally peer reviewed.
Authors
Stuart Bloom, DM, FRCP
Keith Bodger, MBChB(Honours), MD, FRCP
Fraser Cummings, BMSc(Hons), MBChB, MRCP(UK), DPhil, FRCP
Professor Sarah Garner, BPharm, PhD
Professor Kimme Hyrich, MD, PhD, FRCPC
Clare Jacklin
Professor John G Williams, CBE, FRCP
References
1. Gliklich R, Dreyer N, Leavy M, eds. Registries for evaluating patient outcomes: a user’s guide. 3rd ed. Agency for Healthcare Research and Quality (AHRQ). 2010.
2. European Medicines Agency. Patient Registries Workshop, 28 October 2016: Observations and recommendations arising from the workshop. EMA/69716/2017. 13 February 2017 [homepage on the Internet]. [cited 2017 Jul 19]. Available from: http://www.ema.europa.eu/docs/en_GB/document_library/Report/2017/02/WC500221618.pdf
3. GaBI Online – Generics and Biosimilars Initiative. Biosimilars: management of clinical issues [www.gabionline.net]. Mol, Belgium: Pro Pharma Communications International; [cited 2017 Jul 19]. Available from: www.gabionline.net/Biosimilars/Research/Biosimilars-management-of-clinical-issues
4. Ebbers HC, Chamberlain P. Interchangeability. An insurmountable fifth hurdle? Generics and Biosimilars Initiative Journal (GaBI Journal). 2014;3(2):88-93. doi:10.5639/gabij.2014.0302.022
5. Claus BOM, Somers A, Bauters T. Pharmacovigilance of biosimilars and other biologicals within the hospital: current practices and future challenges. Generics and Biosimilars Initiative Journal (GaBI Journal). 2017;6(1):24-6. doi:10.5639/gabij.2017.0601.005
6. Royal College of Physicians. Standards for the clinical structure and content of patient records. 2013 [homepage on the Internet]. [cited 2017 Jul 19]. Available from: http://www.aomrc.org.uk/publications/reports-guidance/standards-for-the-clinical-structure-and-content-of-patient-records-0713/
Disclosure of Conflict of Interest Statement is available upon request.
Copyright © 2017 Pro Pharma Communications International
Permission granted to reproduce for personal and non-commercial use only. All other reproduction, copy or reprinting of all or part of any ‘Content’ found on this website is strictly prohibited without the prior consent of the publisher. Contact the publisher to obtain permission before redistributing.