Local policies on biosimilars: are they designed to optimize use of liberated resources?
Published on 2020/08/03
Generics and Biosimilars Initiative Journal (GaBI Journal). 2020;9(4):163-70.
Author byline as per print journal: Arianna Bertolani, PhD; Claudio Jommi, MS
|
Study Objectives: Different policies have been implemented to enhance uptake of biosimilars. Regarding policies focussing on the demand-side, the literature has mainly concentrated on interchangeability and substitutability recommendations, issued by national or regional policymakers. Information on actions taken by healthcare organisations (HCOs) regarding prescribing behaviour is limited. Furthermore, there is no evidence on whether local authorities implemented a policy framework aimed to appropriately reallocate resources gained through patent expiration. This paper aims to fill these gaps, investigating policies on biosimilars implemented at the local level in the Italian National Health Service. |
Introduction
The European Medicines Agency (EMA) defines a biosimilar as a biological medicine highly similar to another biological medicine already approved in the EU (reference medicine) in terms of structure, biological activity and its efficacy, safety and immunogenicity profile. The first biosimilar was approved by EMA in 2006 [1]. Since then, 54 biosimilars have been approved in Europe, nine authorizations were withdrawn after approval, and two applications were refused (up to April 2020). The approved biosimilars include growth factors (epoetins, filgrastim, pegfilgrastim), hormones (follitropin-α, insulin glargine and lispro, somatropin, teriparatide), low molecular weight heparins (enoxaparin sodium), monoclonal antibodies (adalimumab, infliximab, rituximab, bevacizumab, trastuzumab) and fusion proteins (etanercept) [2].
Due to the natural variability and more complex manufacturing of biological medicines, a biosimilar is not considered to be a generic of a biological medicine. This has ignited a debate regarding the interchangeability of biosimilars, which is defined as ‘the possibility of exchanging one medicine for another that is expected to have the same clinical effect’. This can refer to replacing a reference medicine with a biosimilar (or vice versa) or replacing one biosimilar with another. Replacement can be done by: (i) switching, which is when the prescriber decides to exchange one medicine for another with the same therapeutic intent; (ii) automatic substitution, which is the practice of dispensing one medicine instead of another interchangeable medicine at the pharmacy level without consulting the prescriber [1].
Akin to generics, biosimilars have the opportunity to create competition, offer less expensive alternatives to existing medicines, and/or push alternative medicines to lower their prices due to biosimilar competition [3, 4]. This increases the availability of financial resources, which is important in an era of restricted healthcare budgets [5–7]. Savings from biosimilars can be used to: (i) fund new and costly medicines required for unmet needs, or that provide an added therapeutic value to existing therapies; (ii) fund other healthcare services/initiatives; (iii) generate savings; or (iv) increase the number of patients treated due to lower treatment costs. Several budget-impact studies have estimated savings from biosimilar introduction and/or the related number of additional patients that can potentially be treated with these liberated resources in different European countries [8–10]. Two studies assessed the savings achievable by using biosimilar filgrastim for the treatment of chemotherapy-induced febrile neutropenia and biosimilar follitropin-α for the treatment of anaemia, simulating their reinvestment to increase the number of patients treated with new targeted antineoplastic drugs [11, 12].
Diversified policies have been implemented in Europe, both at the national, regional and local level, to increase the use of biosimilars in clinical practice. These have exhibited differences, in terms of uptake of biosimilars and related savings, even when implemented in the same country [3]. Policies developed by national policymakers are related to the supply and demand of biosimilars. On the supply-side, these include pricing and reimbursement procedures [13], e.g. internal reference pricing (IRP), external reference pricing (ERP); and recommendations on tendering practices that might influence pricing strategies of the industry at regional and local levels. On the demand-side, these include interchangeability and substitutability [14] recommendations, possibly supported by post-marketing evidence on the switch from originators to biosimilars [15]. However, most of the demand-side policies are implemented by regional and local payers, including educational programmes/information campaigns, pharmaceutical prescription budgets, prescription quotas, monitoring of prescriptions patterns, financial incentives or penalties. These payers are also mostly responsible for procurement procedures [16].
Both supply- and demand-side measures are important to achieve savings from biosimilars and regulatory authorities should find a balance between the implementation of these different policies. Only introducing price reductions, without including demand-side measures, typically limits the use of biosimilars [17]. However, appreciable price reductions may play an important role in countries where access to biologicals is limited due to the high prices of originators and high co-payments, such as many of those in Central and Eastern Europe [18].
Policies on biosimilars have been investigated in the recent literature. We performed a literature search on MEDLINE (PubMed) and Web of Science with the purpose of identifying the most important peer reviewed, original research articles published, in English, on policies on biosimilars implemented at local level. The literature search was performed using the following key terms: ‘biosimilar medicines’, ‘demand- and supply-side measures’, ‘local policies’, ‘prescribing targets’, ‘tender’ and ‘educational programmes’. Articles were selected for further analysis based on title and abstract screening. The search was supplemented by a manual review of the reference lists of identified articles.
Some studies focused on nationwide policies, e.g. recommendations on interchangeability, automatic substitutability, tendering procedures [19, 20], while others, using literature review and/or interview/survey methods, investigated the adoption of both national, regional and local policies, generally comparing the regulatory landscape of different countries [3, 21-23]. Very few studies have empirically investigated the impact of these policies. The impact of incentive schemes to encourage biosimilar uptake was assessed by Rémuzat et al. across several European countries [22]. The study found a correlation between incentive policies and the uptake of biosimilars, but the presence of incentive schemes was surveyed at the national level, and heterogeneity of the local measures adopted was disregarded. Moorkens et al. appraised the effect of biosimilar policies and initiatives on market dynamics for infliximab and etanercept among regions in Sweden [24, 25]. Variations in the market share of biosimilars between the Swedish regions, especially regarding infliximab, were found to be largely explained by the discounted price difference between the biosimilar and the originator, thus depending more on supply-side than demand-side policies. Another study by Curto et al. investigated regional policies on tenders in Italy and found important differences in actual prices charged in different regions [26, 27].
In most cases, the reported evidence does not account for the heterogeneity of the policies implemented at the local level. Furthermore, no study has investigated whether healthcare organisations (HCOs): (i) have implemented systematic, prospective and/or retrospective impact evaluation of biosimilars; and (ii) planned in advance how to invest resources made available by treatment cost reduction. As outlined above, these resources can be used to increase patient access to these treatments, fund new and innovative medicines and other healthcare services, and/or simply accumulate savings.
This paper aims to address the information gaps identified. It will investigate local biosimilars policies and uncover whether they are driven by prospective and/or retrospective impact evaluations of biosimilars; and if there have been structured analyses of how to invest resources made available by treatment cost reduction.
The analysis was carried out in Italy. Here, the Italian National Health Service (INHS) represents an interesting case study due to its fragmentation. Variations across HCOs can be attributed to both regional and intra-regional differences.
Italian policies on biosimilars
Italy has a decentralized healthcare system and biosimilar policies have been developed by national, regional and local policymakers. The Italian Medicines Agency (Agenzia Italiana del Farmaco, AIFA) issued two position papers on biosimilars in 2013 and 2018, respectively [28, 29]. In both documents, AIFA notes that the comparability exercise performed by EMA is sufficient to define biosimilars as interchangeable with the related originators both for naïve and non-naïve patients and, as specified in the more recently released version, in terms of quality, safety and efficacy aspects, even in the case of extrapolation of therapeutic indications. However, AIFA advised that the final decision on interchangeability should be left to the prescriber. Regulation for tenders that involve off-patent biological medicines was enacted in 2016 (Law 232/2016). According to this regulation: (i) one lot cannot contain chemically different active principles, even if they are authorized for the same therapeutic indication; (ii) framework agreements must be used if more than three biological medicines based on the same active ingredient are present on the market; and (iii) clinicians should choose between one of the first three drugs identified according to the lowest price or the lowest bid criterion in the framework agreement.
In Italy, Regional Health Authorities (RHAs) are responsible for planning healthcare services, allocating financial resources to healthcare providers and promoting actions to influence the prescribing behaviour, including educational programmes and, moreover, pharmaceutical tendering. Local health authorities are responsible for implementing national and regional policies at a local level, but they are also able to introduce new actions or integrate existing ones [30].
For example, RHAs are free to set or recommend prescriptions targets to HCOs, with related incentives and/or sanctions for clinicians, and/or usage guidelines for biosimilars. Local health authorities can decide to implement these policies as they have been developed at the regional level or to integrate them with further directives. Additionally, local payers might also introduce these policies autonomously, if such recommendations are not made at the regional level. In general, these policies might be implemented to increase savings in those regions affected by mandated Recovery Plans, i.e. regions with important healthcare deficits who must adopt expenditure containment measures.
In 2018, prescriptions of biosimilars accounted for 19% and 25% of the total market for off-patent medicines with at least one available biosimilar, in terms of expenditure and consumption, respectively. The biosimilar market share (expressed as consumption incidence) ranges from 79.4% for epoetin, to 2% for adalimumab. There are also differences in biosimilar market share across regions [31].
Materials and methods
A literature search of local policies on biosimilars was carried out as described above. Data were collected on local policies using a survey distributed to HCOs. A web-based questionnaire, consisting of 40 closed and open-ended questions, was delivered to the general managers of all 199 Italian public HCOs, which includes local health authority and independent hospitals. The questionnaire was sent by email to the target respondents for completion between October 2019 and January 2020. Three reminder email messages were sent after sending the original questionnaire. The individual respondents were directly contacted in case of incomplete surveys.
The questionnaire related to all commercially available biosimilars and was divided into five sections. The first section focused on educational/information programmes on biosimilars, that are important both for physicians and patients to prevent the ‘nocebo’ effect [32]. Investigation included examination of their implementation by year, frequency, main topic(s) of the programme, i.e. biosimilar pipelines, comparative exercise and marketing authorization process, market access pathway for biosimilars, information on the results of tenders; and key target audiences, i.e. clinicians, hospital pharmacists, patients, administrative staff.
The second section aimed to collect information on the prospective and retrospective impact evaluations of biosimilars. The systematic implementation of these analyses by HCOs was investigated along with their subject, i.e. total savings, price reduction, prescribing shift from off-patent (with biosimilars) to patent-protected molecules for the same or similar indication; increase in the number of treated patients due to the lower treatment cost, time horizon and update rate.
The third section investigated whether HCOs estimate the proportion of the target population indicated for biological treatments who may not receive treatments due to budget constraints.
The fourth section explored the implementation of prescription targets and related incentive or sanction schemes, e.g. possibility to reallocate resources gained for new and expensive medicines or for other healthcare services, and withdrawal of the authorization to prescribe. More specifically, the study distinguishes between prescription targets for the same molecule (larger market share for biosimilars or the cheapest products) and those for different molecules sharing the same or similar indications (larger market share for the cheapest molecules in the same therapeutic class). We scrutinized incentive schemes, i.e. rewarding physicians who reach the prescription targets, and sanction schemes, i.e. sanctions for physicians who do not reach the prescription targets. Incentive schemes could be financial, e.g. bonus, or non-financial, e.g. earned autonomy, and intended to change the behaviour of prescribers (direct incentives, e.g. bonus) or patient’s choice of providers (indirect incentives, e.g. cost differentials for patients) [33]. The focus was on direct incentives including financial incentives (bonus), or non-financial incentives, e.g. including allowing prescribers to use free-up resources for innovative but expensive medicines to treat more patients; or to reallocate these resources for other healthcare services, e.g. diagnostics. Sanctions may range from monetary sanctions to a withdrawal of the authorization to prescribe.
The fifth section of the questionnaire aimed to collect evidence on the participation of HCOs in post-marketing studies on biosimilars. More specifically, it surveyed the study type, i.e. effectiveness and safety profile studies, patient compliance, impact evaluation of policies on prescribing behaviour, perceptual surveys targeting patients and healthcare professionals; the year of the study and the availability of grey and/or peer-reviewed references.
The structured questionnaire was validated by two potential respondents before it was administrated to the final target population. The questionnaire was written in Italian. All the collected data are related to 2019 (or last available year), except for educational/information programmes for which the adhesion rate since 2016 was investigated. Missing values were not countered for in the relevant questions.
The next section discusses the sample size. The representativeness of this sample is measured in terms of: (i) the number of respondents; and (ii) the dimension of the relevant HCOs, using HCO total reimbursement funding as an indicator of dimension. Since the 199 HCOs include both local health authorities and independent hospital trusts, other indicators, e.g. number of beds or the relevant population, were not feasible. The descriptive analysis presented in the next section were performed using Microsoft excel.
Results
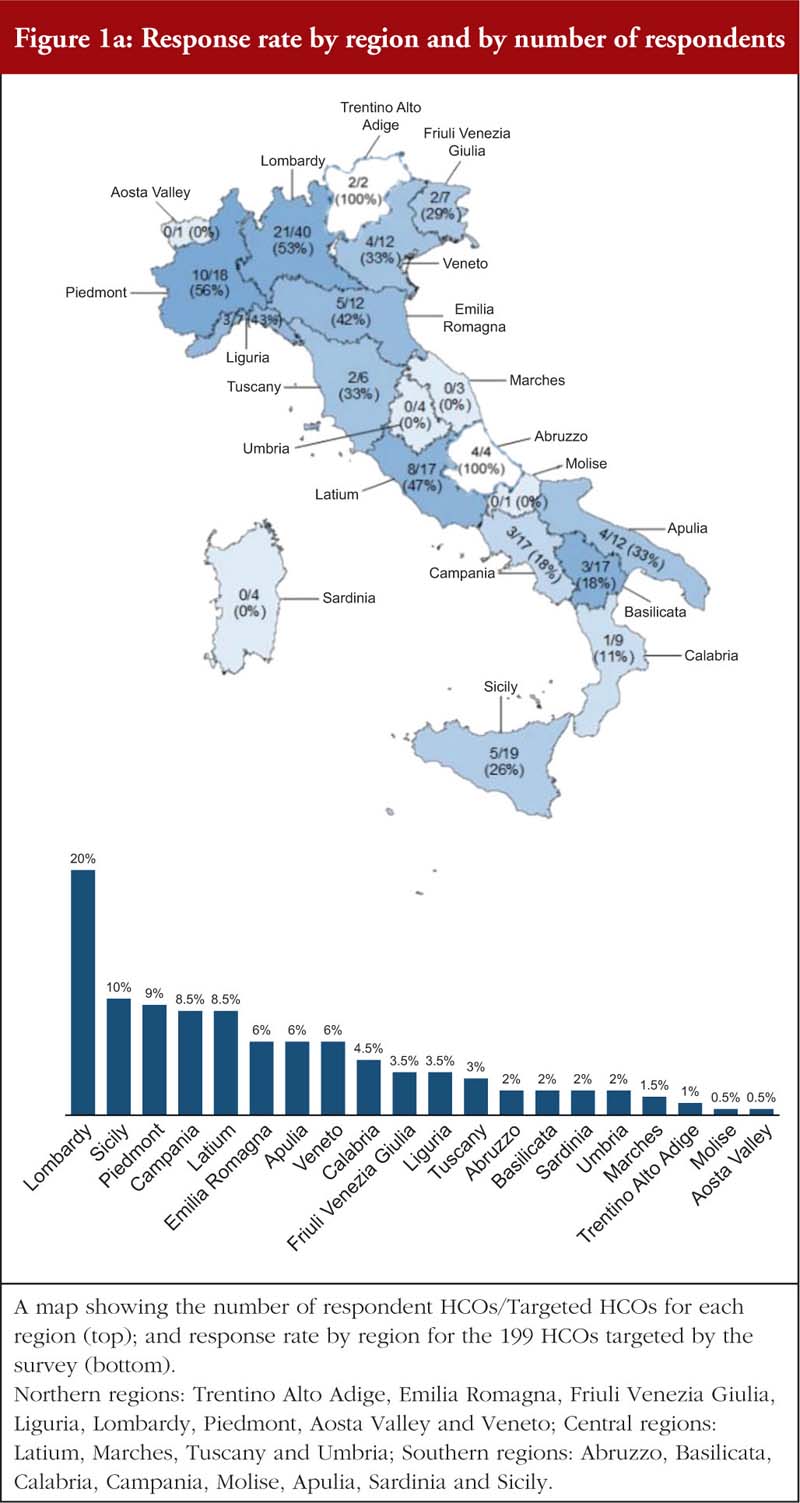
The HCOs survey response rate was 38% nationwide (39% in terms of total HCO total reimbursement funding, as a proxy of the dimension of the HCO), and 47%, 33% and 27% (47%, 26%, 35% in terms of total reimbursement funding) in the Northern, Central and Southern regions, respectively. The majority of all regions (16 out of the 21 regions, accounting for 93% of the Italian population [34]) are represented with a region-specific response rate ranging from 11% to 100% of HCOs, see Figure 1a and 1b. The HCOs that did not complete the questionnaire are located in small regions (between 1% to 2% in terms of total HCO total reimbursement funding) in which there are few local health authorities (in some cases, only one). Most respondents (80%) completed more than 90% of the questionnaire, and only 5% answered less than 70% of questions.
Educational programmes on biosimilars have been carried out in almost all HCOs, with an increasing trend in recent years (from 20% of HCOs in 2016, to 89% in 2019). These programmes have mainly focused on the market access pathway for biosimilars in Italy (83% of the HCOs) and the relevant region (88%), followed by information on the results of tenders (60%), comparability exercises to obtain marketing approval and marketing authorization processes (26%), and biosimilar pipelines (19%). Clinicians and hospital pharmacists are the main targets of these programmes (97% and 77% of the initiatives, respectively). Patients were involved in educational/information programmes on biosimilars in only 22% of the HCOs. These figures are very similar across respondent regions.
We investigated whether HCOs systematically predict and/or ex post evaluate the impact of patent expiration of biological medicines and the availability of biosimilars. It seems that this activity is quite common. Prospective and retrospective impact evaluations are carried out by 75% of the HCOs, 18% of the HCOs systematically evaluate only ex post effects, and 7% do not conduct any analysis. The impact of patent expiration can be prospectively or retrospectively measured in terms of total savings, price reduction, prescribing shift from off-patent (and with biosimilars) to patent-protected molecules for the same or similar indication and increase of treated patients due to a price reduction. On average, HCOs are more interested in investigating total savings and prescribing shift from off-patent to patent-protected medicines. To a lesser extent, prospective analyses are carried out by regions subject to Recovery Plans, however these regions should be more engaged in these analyses as they are required to cover healthcare spending deficits. The same regions pay more attention, both prospectively and retrospectively, to the prescribing shift. It seems that these regions are more concerned with potential inappropriateness of this shift from off-patent to patent-protected molecules, see Table 1.
Only 21% of the HCOs assessed the untreated proportion of the target population for biological treatments. Coverage improvement due to lower costs derived from patent expiration, is estimated in half of these regions.
The objective of increasing the proportion of biosimilar prescriptions (or the cheapest products) is communicated to prescribers by 75% of HCOs. A lower proportion of HCOs (62%) provide physicians with prescription targets for off-patent medicines within the same therapeutic class, e.g. encouraging physicians to prescribe cheaper antitumour necrosis factor-alpha (anti-TNF-α). Among the HCOs providing prescription targets, 68% and 24% rely on incentives and sanctions, respectively. Regions affected by Recovery Plans have implemented these policies in similar proportions to the other regions, but they rely less than others on incentives and more on sanctions.
Rewards include monetary incentives or allowing prescribers to reallocate resources gained for new and expensive medicines, to either treat new patients or to enable physicians to use these resources for other healthcare services, e.g. diagnostics. Monetary incentives account for 56% of reward policies and prevail in the Northern regions; reallocation to new medicines is more frequent in the Central and Southern regions of Italy, see Figure 2A. Similar results are found for incentives and sanctions linked to broader prescription targets within therapeutic classes. In very few cases, savings are redirected to other healthcare services, see Figure 2B. Where sanctions are applied, they are mostly monetary; in a single case, authorization to prescribe was withdrawn.
Finally, 25% of the HCOs reported participation in post-marketing studies on biosimilars. These studies are focused on the safety profile (31% of total cases reported) and the impact of policies on prescribing behaviour (31%), followed by biosimilar effectiveness (16%). Perceptual surveys, targeting patients and healthcare professionals were less frequent (9% and 3%, respectively). Respondents did not provide any grey or peer-reviewed references resulting from these studies.
Supplementary Material (questionnaire and complete set of results) is available on request.
Discussion
Patent expiration and price-competition generated by biosimilar medicines give healthcare systems an opportunity to liberate and redirect resources. These resources can be invested in new medicines approved for unmet needs and severe diseases, or those that bring added therapeutic value to existing alternatives; or they can be used to improve access to medicines as the treatment cost reduces. HCOs may also utilize these resources for other healthcare services or simply as savings. Both demand- and supply-side measures implemented by the regulatory authorities are important to enhance the cost savings achieved by the introduction of biosimilars [21].
Most of the studies on biosimilar policies have focused either on supply-side measures, e.g. price regulation, or on recommendations issued by central authorities on interchangeability and/or substitutability issues. Little empirical evidence on the role played by local payers has been published thus far, in particular with reference to actions aimed at governing prescribing behaviour [28, 29]. Furthermore, the published evidence has not highlighted the heterogeneity of adopted solutions at the local level and the existence of a general framework for policy design and implementation. This paper is intended to fill this gap, using Italy as a case study as its decentralized health system provides interesting insight into the convergence and divergence of local policies.
The study has revealed that there has been huge investment in biosimilar policies by respondent HCOs. Educational programmes on biosimilars have been carried out in almost all HCOs. Some 93% of HCOs conduct retrospective and/or prospective assessment of the impact of patent expirations for biological drugs. Prescribers have been urged to increase prescriptions of biosimilars (or the cheapest product) within the same therapeutic class by 75% and 62% of HCOs, respectively. Incentive schemes mostly rely on monetary transfers and were applied by 68% of those HCOs that have introduced prescription targets, whereas penalties for prescribers who do not reach target levels are much less diffused.
However, it is clear that local initiatives still lack a general framework for policy design and implementation. For example, only 21% of the HCOs systematically estimate the proportion of the target population that is not receiving biological treatment. This information is useful to understand whether resources gained from biosimilar substitutions should be used to treat more patients and exploit the lower prices. Furthermore, in the face of widespread retrospective evaluation of the market impact of biosimilars, only 25% of HCOs declare having participated in post-marketing studies on biosimilars, and only 30% of these HCOs have carried out specific impact analyses of biosimilar policies, with no published evidence or references reported.
This current study has some limitations. Firstly, the sample accounts for 38% of public HCOs. It can be assumed that the HCOs which were more active in implementing policies were also those which were more likely to respond, generating a possible selection bias. Despite almost all regions (and all major regions) being represented, the proportion of respondents do vary across regions. Hence, we cannot state that the sample represents all HCOs in Italy. We also decided to include only public HCOs, since private accredited hospitals are very different across regions, ranging from small hospitals where biological treatments are not used at all, to highly specialized hospitals where biological treatments may represent a high proportion of the drug budget. Secondly, the study relied on the information provided by the respondents. Information on the HCO websites was limited, and cross-check analysis could not be performed. Thirdly, the impact of these policies on biosimilar penetration rates was not estimated. This has compromised the comparability of this research to the few other empirical studies which have investigated this topic through cross-country [22] and cross-regional [24, 25] comparisons. This research question was beyond the scope of this study and other policies may have affected this penetration rate, e.g. tenders, and actions on prescribing behaviour were not necessarily aimed at increasing biosimilar penetration rates, but on maximizing the impact of patent expiration.
Conclusions
Despite its limitations, this study provides broad and empirical information on the biosimilar policies implemented by HCOs that are part of the Italian National Health Service. These policies include educational and information programmes, prospective and retrospective analysis of the impact of patent expiration and biosimilars launch, prescription targets and incentive/sanction schemes, implementation of post-marketing studies on biosimilars, and estimates of patients not able to access biological medicines due to high treatment costs (the untreated) who could benefit from the availability of cheaper drugs. The findings have some important policy implications.
Firstly, variability in implemented policies was uncovered. However, variation across HCOs is not necessarily an issue as different policies are needed to solve different problems. For example, Southern regions have focused their prospective evaluation on prescription shifts from off-patent to patent protected molecules. This could be attributed to a higher perceived risk of an inappropriate prescription shift, whereas Northern regions were more focused on the impact on prices.
In addition, in Southern regions, where HCOs were more focused on prescription shifts in their prospective analysis, there has been a less pronounced introduction of prescription targets per therapeutic class. However, targets are provided exactly to avoid inappropriate prescription shifts. This highlights the importance of ensuring the internal consistency of policies.
Lastly, the study shows that in most HCOs a general framework to decide how to reallocate resources recovered as a result of biosimilar introduction is still lacking. This framework implies a systematic prospective and retrospective analysis of the impact of biosimilars and an evaluation of possible areas of under-treatment with biologicals. For example, a more diffused awareness of the extent of untreated patients is important to understand whether such regained resources should be prioritized for untreated patients or earmarked for other purposes, e.g. coverage of new molecules launched on the market.
Acknowledgements
The authors would like to thank Paolo Bordon (General Manager of Azienda Provinciale per i Servizi Sanitari – Trento) and Angelo Tanese (General Manager of Azienda Sanitaria Locale Roma 1) for having validated the questionnaire, and all participants in the survey. The authors thank Helen Banks (CERGAS, SDA Bocconi) and Lisa Pirrie (ApotheCom) for revision of the English text.
Funding sources
The present study was funded by Sandoz SpA through an unrestricted grant to CERGAS, SDA Bocconi School of Management. No interferences occurred in carrying out the research project and in writing the manuscript for which the authors are solely responsible.
Priori presentations: These results have not been presented before.
Competing interests: Arianna Bertolani and Claudio Jommi received an unrestricted grant from Sandoz, but no interferences occurred in carrying out the research project and in writing the manuscript for which the authors have the sole responsibility.
Provenance and peer review: Not commissioned; externally peer reviewed.
Authors
Arianna Bertolani, PhD, Junior Lecturer – Government, Health and Non-Profit Division
Claudio Jommi, MS
Centre for Research on Health and Social Care Management (CERGAS), SDA Bocconi School of Management, Bocconi University, 10 Via Sarfatti, IT-20136 Milan, Italy
References
1. European Medicines Agency and the European Commission. Biosimilars in the EU: information guide for healthcare professionals. 2019 [homepage on the Internet]. [cited 2020 Sep 25]. Available from:https://www.ema.europa.eu/en/documents/leaflet/biosimilars-eu-information-guide-ealthcare-professionals_en.pdf
2. European Medicines Agency. European public assessment reports (EPARs) for human medicines [homepage on the Internet]. [cited 2020 Sep 25]. Available from: https://www.ema.europa.eu/en/medicines/download-medicine-data
3. Moorkens E, Vulto AG, Huys I, Dylst P, Godman B, Keuerleber S, et al. Policies for biosimilar uptake in Europe: an overview. PLoS One. 2017;12(12):e0190147.
4. Troein P, Newton M, Patel J, Scott K. The impact of biosimilar competition in Europe. IQVIA Report 2019 [homepage on the Internet]. [cited 2020 Sep 25]. Available from: https://ec.europa.eu/docsroom/documents/38461
5. Farfan-Portet MI, Gerkens S, Lepage-Nefkens I, Vinck I, Hulstaert F. Are biosimilars the next tool to guarantee cost-containment for pharmaceutical expenditures? Eur J Health Econ. 2014;15(3):223-8.
6. Dutta B, Huys I, Vulto AG, Simoens S. Identifying key benefits in European off-patent biologics and biosimilar markets: it is not only about price! BioDrugs. 2020;34:159-70.
7. Godman B, Allocati E, Moorkens E. Ever-changing landscape of biosimilars in Canada; findings and implications from a global perspective. Generics and Biosimilars Initiative Journal (GaBI Journal). 2019;8(3):93-7. doi:10.5639/gabij.2019.0803.012.
8. Gulácsi L, Brodszky V, Baji P, Rencz F, Péntek M. The rituximab biosimilar CT-P10 in rheumatology and cancer: a budget impact analysis in 28 European countries. Adv Ther. 2017;34(5):1128-44.
9. Jha A, Upton A, Dunlop WCN, Akehurst R. The budget impact of biosimilar infliximab (Remsima®) for the treatment of autoimmune diseases in five European countries. Adv Ther. 2015;32(8):742-56.
10. Rognoni C, Bertolani A, Jommi C. Budget impact analysis of rituximab biosimilar in Italy from the hospital and payer perspectives. Global Reg Health Technol Assess. 2018. doi:10.1177/2284240318784289.
11. Sun D, Andayani TM, Altyar A, MacDonald K, Abraham I. Potential cost savings from chemotherapy-induced febrile neutropenia with biosimilar filgrastim and expanded access to targeted antineoplastic treatment across the European Union G5 countries: a simulation study. Clin Ther. 2015;37(4):842-57.
12. Abraham I, Han L, Sun D, MacDonald K, Aapro M. Cost savings from anemia management with biosimilar epoetin alfa and increased access to targeted antineoplastic treatment: a simulation for the EU G5 countries. Future Oncol. 2014;10(9):1599-609.
13. Vogler S, Zimmermann N, Haasis MA. PPRI Report 2018. Pharmaceutical pricing and reimbursement policies in 47 PPRI network member countries. WHO Collaborating Centre for Pricing and Reimbursement Policies, Gesundheit Österreich GmbH (GÖG/Austrian National Public Health Institute). 2019 [homepage on the Internet]. [cited 2020 Sep 25]. Available from: https://ppri.goeg.at/sites/ppri.goeg.at/files/inline-files/PPRI%20Report2018_final.pdf
14. Vogler S, Schneider P. Do pricing and usage-enhancing policies differ between biosimilars and generics? Findings from an international survey. Generics and Biosimilars Initiative Journal (GaBI Journal). 2017;6: 79–88. doi:10.5639/gabij. 2017.0602.015.
15. Jørgensen KK, Olsen IC, Goll GL, Lorentzen M, Bolstad N, Haavardsholm EA, et al. Switching from originator infliximab to biosimilar CT-P13 compared with maintained treatment with originator infliximab (NOR-SWITCH): a 52-week, randomised, double-blind, non-inferiority trial. Erratum. Lancet. 2017;389(10086):2304-16.
16. WHO Collaborating Centre for Pharmaceutical Pricing and Reimbursement Policies. Glossary [homepage on the Internet]. [cited 2020 Sep 25]. Available from: https://ppri.goeg.at/ppri-glossary
17. Kim Y, Kwon HY, Godman B, Moorkens E, Simoens S, Bae S. Uptake of biosimilar infliximab in the UK, France, Japan and Korea. Budget savings or market expansion across countries? Front Pharmacol. 2020;11:970.
18. Baumgart DC, Misery L, Naeyaert S, Taylor PC. Biological therapies in immune-mediated inflammatory diseases: can biosimilars reduce access inequities? Front Pharmacol. 2019;10:279.
19. O’Callaghan J, Barry SP, Bermingham M, Morris JM, Griffin BT. Regulation of biosimilar medicines and current perspectives on interchangeability and policy. Eur J Clin Pharmacol. 2019;75(1):1-11.
20. Trifirò G, Marcianò I, Ingrasciotta Y. Interchangeability of biosimilar and biological reference product: updated regulatory positions and pre- and post-marketing evidence. Expert Opin Biol Ther. 2018;18(3):309-15.
21. Renwick MJ, Smolina K, Gladstone EJ, Weymann D, Morgan SG. Postmarket policy considerations for biosimilar oncology drugs. Lancet Oncol. 2016;17(1):e31-8.
22. Rémuzat C, Kapuśniak A, Caban A, Ionescu D, Radière G, Mendoza C, et al. Supply-side and demand-side policies for biosimilars: an overview in 10 European member states. J Mark Access Health Policy. 2017;5(1):1307315.
23. Acha V, Allin P, Bergunde S, Bisordi F, Roediger A. What pricing and reimbursement policies to use for off-patent biologicals in Europe? – Results from the second EBE 2014 biological medicines policy survey. Generics and Biosimilars Initiative Journal (GaBI Journal). 2015;4(1):17-24. doi:10.5639/gabij. 2015.0401.006.
24. Moorkens E, Simoens S, Troein P, Declerck P, Vulto AG, Huys I. Different policy measures and practices between Swedish counties influence market dynamics: Part 1-biosimilar and originator infliximab in the hospital setting. BioDrugs. 2019;33(3):285-97.
25. Moorkens E, Simoens S, Troein P, Declerck P, Vulto AG, Huys I. Different policy measures and practices between Swedish counties influence market dynamics: Part 2-biosimilar and originator etanercept in the outpatient setting. BioDrugs. 2019;33(3):299-306.
26. Curto A, Van der Vooren K, Garattini L, Lo Muto R, Duranti S. Regional tenders on biosimilars in Italy: potentially competitive? Generics and Biosimilars Initiative Journal (GaBI Journal). 2013;2(3):123-9. doi:10.5639/gabij.2013.0203.036.
27. Curto S, Ghislandi S, van de Vooren K, Duranti S, Garattini L. Regional tenders on biosimilars in Italy: an empirical analysis of awarded prices. Health Policy. 2014;116(2-3):182-7.
28. Agenzia Italiana del Farmaco (AIFA) – Position Paper. I farmaci biosimilari. 2013 [homepage on the Internet]. [cited 2020 Sep 25]. Available from: https://www.aifa.gov.it/documents/20142/0/AIFA_POSITION_PAPER_FARMACI_BIOSIMILARI.pdf/22e2c111-dbbd-5b96-fd9c-e29c057d8204
29. Agenzia Italiana del Farmaco (AIFA) – Secondo Position Paper AIFA sui farmaci biosimilari. 2018 [homepage on the Internet]. [cited 2020 Sep 25]. Available from: https://www.aifa.gov.it/sites/default/files/pp_biosimilari_27.03.2018.pdf
30. Jommi C, Costa E, Michelon A, Pisacane M, Scroccaro G. Multi-tier drugs assessment in a decentralised health care system. The Italian case-study. Health Policy. 2013;112(3):241-7. doi:10.1016/j.healthpol.2013.06.004
31. Agenzia Italiana del Farmaco (AIFA) – Osservatorio Nazionale sull’impiego dei Medicinali. L’uso dei farmaci in Italia. Rapporto Nazionale Anno 2018 [homepage on the Internet]. [cited 2020 Sep 25]. Available from: https://www.aifa.gov.it/documents/20142/0/Rapporto_OsMed_2018.pdf/c9eb79f9-b791-2759-4a9e-e56e1348a976
32. Pouillon L, Socha M, Demore B, Thilly N, Abitbol V, Danese S, et al. The nocebo effect: a clinical challenge in the era of biosimilars. Expert Rev Clin Immunol. 2018;14(9):739-49.
33. Custers T, Hurley J, Klazinga NS, Brown AD. Selecting effective incentive structures in health care: a decision framework to support health care purchasers in finding the right incentives to drive performance. BMC Health Serv Res. 2008;8:66.
34. I.STAT Database – Popolazione residente al 1° gennaio 2019 [homepage on the Internet]. [cited 2020 Sep 25]. Available from: http://dati.istat.it/Index.aspx?DataSetCode=DCIS_POPRES1#
|
Author for correspondence: Arianna Bertolani, PhD, Centre for Research on Health and Social Care Management (CERGAS), SDA Bocconi School of Management, Bocconi University, 10 Via Sarfatti, IT-20136 Milan, Italy |
Disclosure of Conflict of Interest Statement is available upon request.
Copyright © 2020 Pro Pharma Communications International
Permission granted to reproduce for personal and non-commercial use only. All other reproduction, copy or reprinting of all or part of any ‘Content’ found on this website is strictly prohibited without the prior consent of the publisher. Contact the publisher to obtain permission before redistributing.