Abstract: In December 2023, the University of Helsinki, Helsinki University Hospital (HUS), and University Pharmacy (Yliopiston Apteekki) Helsinki collaborated to present the online symposium titled ‘Current Trends in Biosimilar Uptake and Research with Special Focus on Automatic Substitution’. This provided an overview of global trends in biosimilar use and a systematic examination of the implications…
Author(s):
Professor Alan Lyles, BSPharm, MPH, ScD, PhD (h.c.), Heinonen E, MD, PhD, Tolonen HM, MSc (Pharm), PhD, Linden K, MSc (Pharm), MSc (Econ), PhD, Sihvo S, PhD, Sarnola K, MSc (Pharm), MSc (Econ), PhD, Airaksinen M, MSc (Pharm), PhD
affordable medicines, automatic substitution, biological medicines, biosimilar, community pharmacy, Finland
DOI: 10.5639/gabij.2025.1402.009
 Interview
Interview
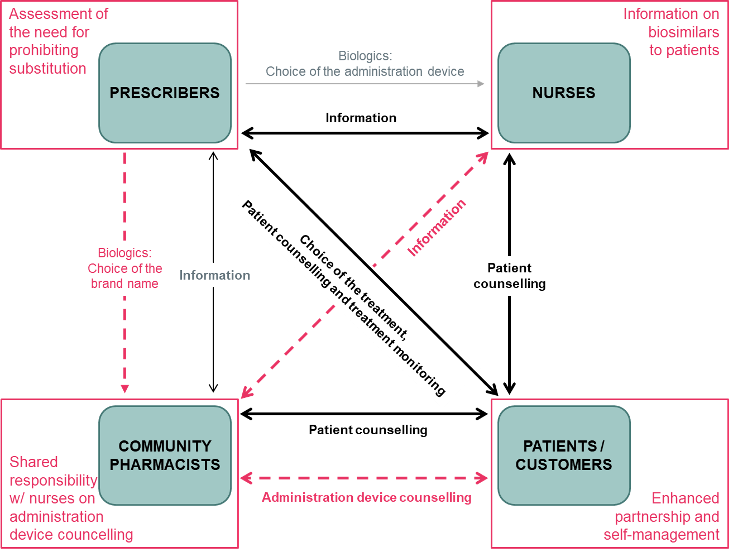 Meeting Report
Meeting Report
 Editor's Letter
Editor's Letter
 Original Research
Original Research
 Meeting Report
Meeting Report
 Letters to the Editor
Letters to the Editor
 Letters to the Editor
Letters to the Editor
 Editor's Letter
Editor's Letter
 Review Article
Review Article


