 Editor's Letter
Editor's Letter
Published on 11 July 2024
First 2024 GaBI Journal issue highlights
Author(s): Robin Thorpe, PhD, FRCPath
DOI: 10.5639/gabij.2024.1301.001
1.103 views
 Editor's Letter
Editor's Letter
Published on 11 July 2024
Author(s): Robin Thorpe, PhD, FRCPath
DOI: 10.5639/gabij.2024.1301.001
1.103 views
Published on 11 July 2024
Author(s): Irene Krämer, PhD, Judith Thiesen, PhD
anticancer drugs, cytotoxic, in-use stability, parenteral administration
DOI: 10.5639/gabij.2024.1302.010
4.546 views
Published on 11 July 2024
biosimilarity, interchangeability, misinformation, regulation, uptake
DOI: 10.5639/gabij.2024.1302.009
3.193 views
Published on 30 July 2024
Author(s): Shivani Mittra, MPharm, PhD, Shylashree Baraskar, MBBS, Elena Wolff-Holz, MD, Sandeep N Athalye, MBBS, MD
biological, biosimilars, carbon footprint, clinical trials, ESG (Environmental Social Governance), sustainability
DOI: 10.5639/gabij.2024.1303.033
2.234 views
Published on 10 May 2024
Author(s): Michael S Reilly, Esq
formulary, interchangeable biosimilar, market uptake, pharmacy benefit managers
DOI: 10.5639/gabij.2024.1301.006
2.493 views
Published on 07 May 2024
affordability, biosimilars, low- and middle-income countries (LMICs), market access, non-communicable diseases, regulatory framework
DOI: 10.5639/gabij.2024.1302.008
6.748 views
Published on 19 April 2024
Author(s): Adjunct Professor Pekka Kurki, MD, PhD
biosimilars, competition, controlled efficacy studies, harmonization
DOI: 10.5639/gabij.2024.1301.004
2.724 views
 Table Contents
Table Contents
Published on 15 April 2024
57 views
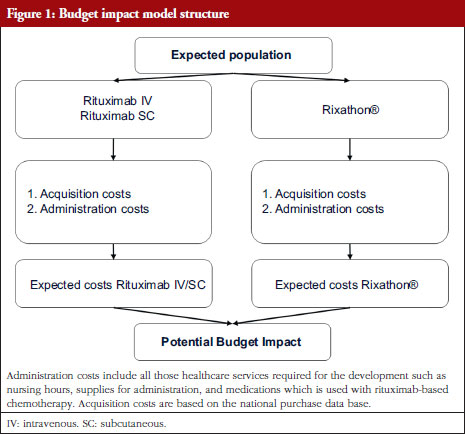 Original Research
Original Research
Published on 21 March 2024
biosimilars, budget impact analysis, Chile, health system perspective, non-Hodgkin lymphoma, rituximab
DOI: 10.5639/gabij.2024.1301.002
3.221 views
Published on 06 March 2024
biosimilar, education, oncology, regulatory
DOI: 10.5639/gabij.2024.1301.005
1.729 views
Published on 25 January 2024
DOI: 10.5639/gabij.2023.1203.017
1.178 views
Published on 11 January 2024
Author(s): Sarah Schrieber, PharmD
DOI: 10.5639/gabij.2023.1203.016
4.408 views